Spectroscopy
1/35
Earn XP
Description and Tags
UV and visible, Infrared, Mass Spec and NMR
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
what does NMR spectroscopy stand for?
Nuclear magnetic resonance spectroscopy
What are the two types of NMR spectroscopy?
carbon 13 NMR spectroscopy
proton NMR spectroscopy
Scientists (dating back to the 1500s) identified elements by using the flame test. Now scientists use spectroscopy. give 3 advantages of spectroscopy than flame testing
sped up identification
only small amounts of the sample is needed for analysis
results are more accurate
Why cant NMR spectroscopy be used for all compounds/atoms?
only compounds with an nuclei with an odd mass number/odd atomic number will posses spin.
a nucleus without spin cannot be detected by NMR spectroscopy
Q1.Which amine has only three peaks in its proton NMR spectrum? Methylamine, Trimethylamine , Diethylamine , Propylamine
Diethylamine
The non-toxic, inert substance TMS is used as a standard in n.m.r. spectra. (i) Give two other reasons why TMS is used as a standard in recording n.m.r. spectra.(2 marks)
low boiling point-can be distilled off and used again
wont interfere with main signals
can easily be removed
produce a single peak
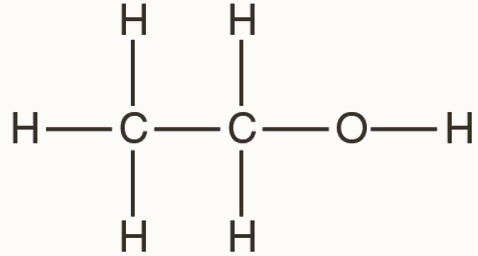
draw the displayed formula of Ethanol, how many peaks will Ethanol have in a carbon 13 NMR spectroscopy
2 peaks
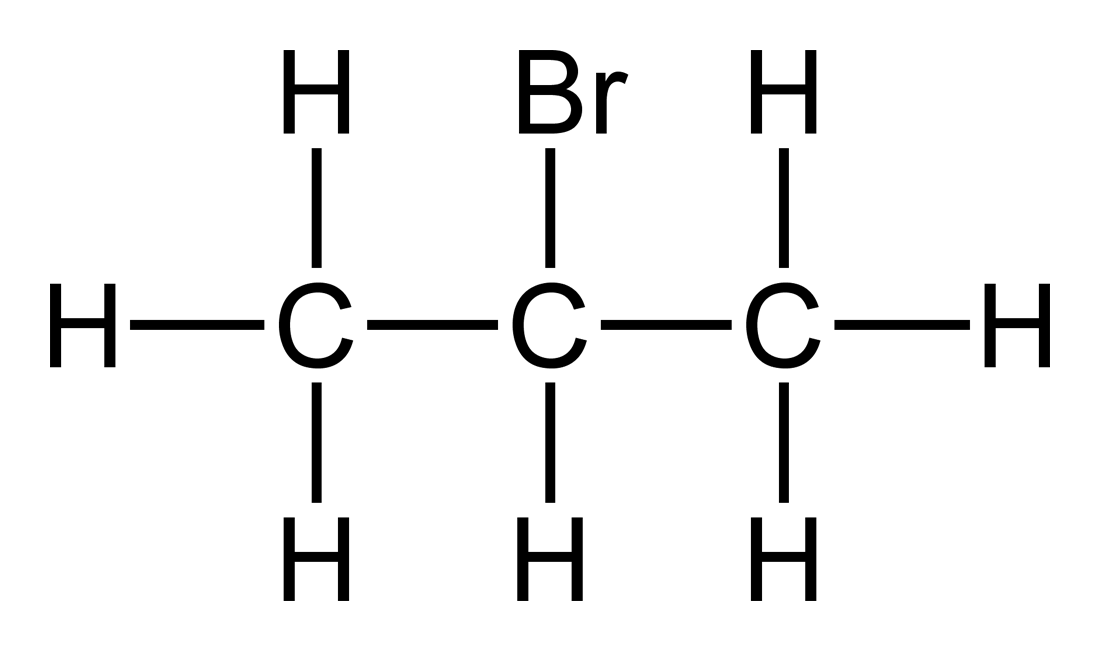
draw the displayed structure of bromopropane and how many peaks will it have in a carbon 13 NMR spectroscopy
2 peaks
Top Tips! when working out how many peaks a compound has in carbon NMR
draw it displayed and follow each carbon atom through
draw the structural formula of TMS. (1 mark)
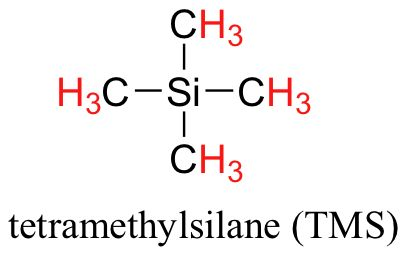
give the molecular formula of TMS
C4H12Si
Give the name of the compound used as a reference used in carbon 13 MNR spectroscopy
Tertramethylsilane
give the number of peaks you would expect for the carbon 13 NMR spectra of pentane
3
give the number of peaks you would expect for the carbon 13 NMR spectra of dimethylpropane
2
give the number of peaks you would expect for the carbon 13 NMR spectra of butanone
4
give the number of peaks you would expect for the carbon 13 NMR spectra of cyclohexane
1
which type of NMR spectroscopy does peak heigh matter?
proton NMR spectroscopy
why would CCl4 and CDCL3 good solvents for NMR?
Inert
They both have no protons so they will not produce peaks in a proton spectra
produce one singlet peak in a carbon spectra that doesn’t interfere with other peaks
CCl4 and CDCL3 are used in NMR spectroscopy.
In terms of structure and polarity why is CCl4 and CDCL3 good solvents
CCl4 has a tetrahedral shape and is symmetrical. therefore is non-polar and can dissolve non-polar substances
CDCL3 has a tetrahedral shape and is non-symmetrical. therefore is polar and can dissolve polar substances
In proton NMR spectroscopy the number of signals/peaks tells you…
the number of different proton environments
In proton NMR spectroscopy the chemical shift tells you…
the general environment of the protons
In proton NMR spectroscopy the peak area/height tell you…
how many protons are in each environment
In proton NMR spectroscopy multiplicity tells you…
how many protons are on neighbouring carbon atoms.
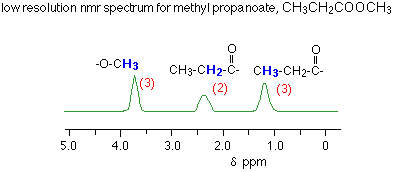
In low resolution proton NMR spectra what is shown?
give the number of protons for each environmental group
represented as 1 peak
In high resolution proton NMR spectra what is shown?
multiplicity/splitting can happen for each peak.
each peak indicated the number of protons on adjacent carbon atoms
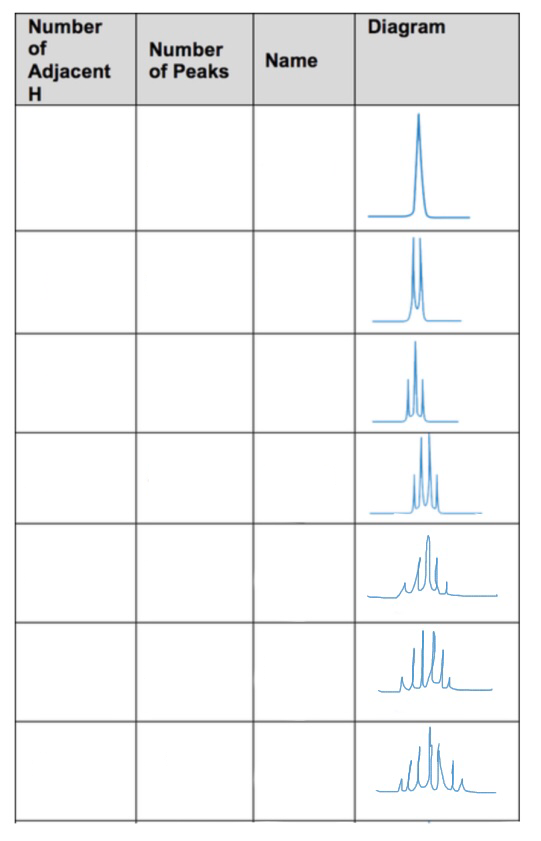
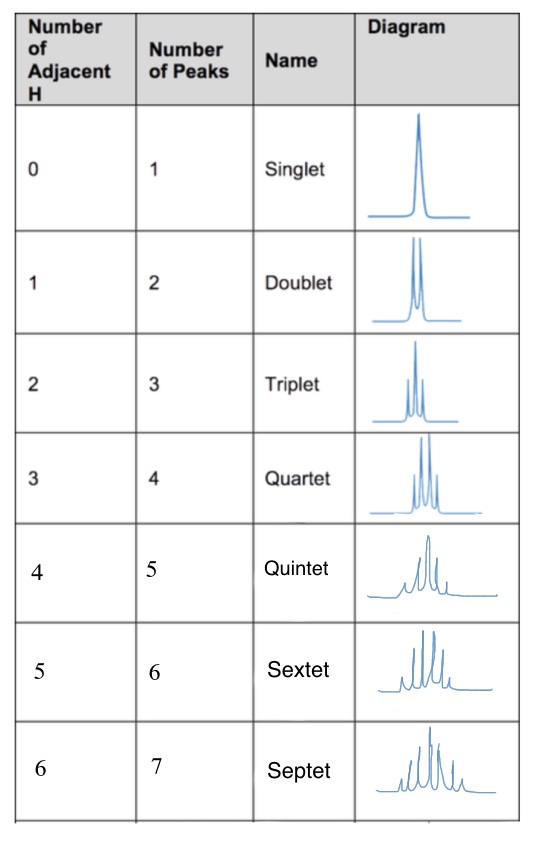
1 peaks is
a singlet
so zero neighbouring H
2 peaks is
a doublet
So 1 neighbouring H
3 peaks is
a triplet
so 2 neighbouring H
4 peaks is
a quartet
so 3 neighbouring H
5 peaks is
a quintet
4 neighbouring H
6 peaks
a sextet
5 neighbouring H
7 peaks
a septet
6 neighbouring H
What do you do if an exam question asks you to work out the number of hydrogen atoms for each peak in a proton NMR spectra?
measure the distance between the to and bottom lines for each peak.
then compare the heights and make them into the simple ratio
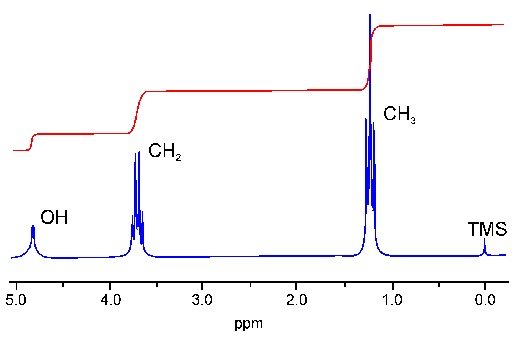
Work out the ratio of hydrogen atoms present in each peak
5:10:15
so 1:2:3
how many peaks will Cl-CH2-CH2-Cl produce?
because the protons are in chemically equivalent environments, so no splitting occurs and a
1 singlet peak will form