Material Science ME313 Final Practice
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
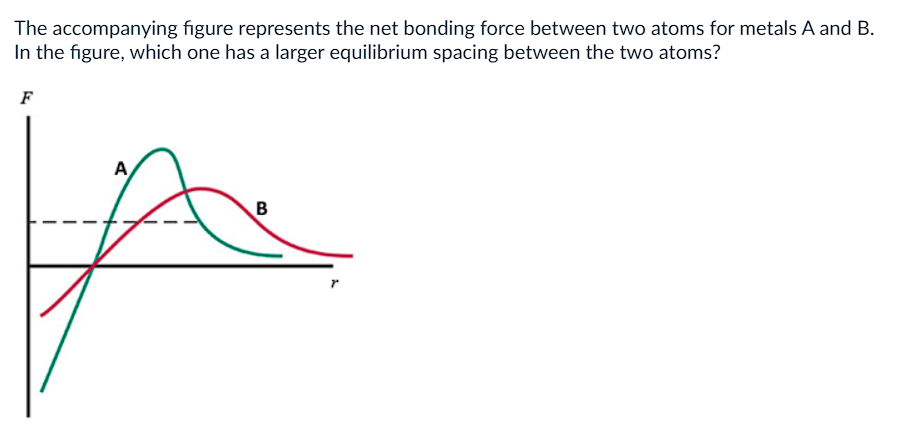
a) A
b) B
c) They are Equal
d) Not enough info
c) They are Equal
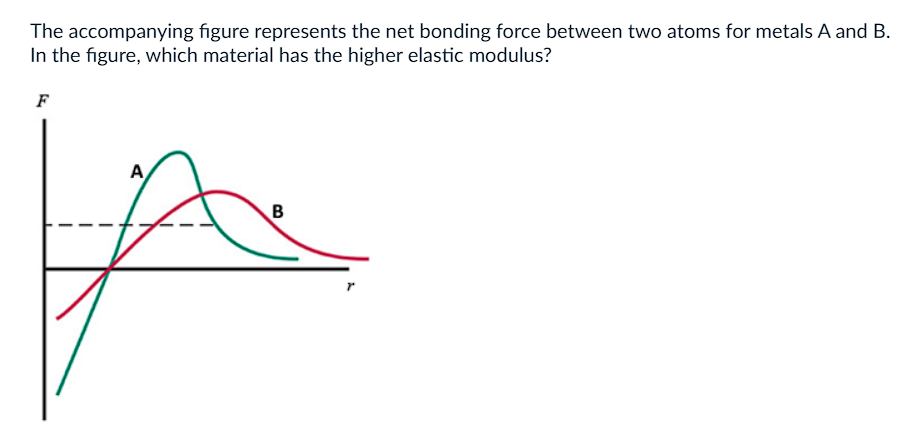
a) A
b) B
c) They are Equal
d) Not enough info
a) A
Which class of material is generally associated with the highest density values at room temperature?
a) ceramics
b) metals
c) polymers
d) composites
b) metals
The unit cell of a face centered cubic (FCC) crystal structure contains:
a) 1 atom
b) 2 atoms
c) 4 atoms
d) 14 atoms
c) 4 atoms
![<p>a) BCC [1 0 0]</p><p>b) BCC [1 1 0]</p><p>c) BCC [1 1 1]</p><p>d) FCC [1 0 0]</p><p>e) FCC[1 1 0]</p><p>f) FCC [1 1 1]</p>](https://assets.knowt.com/user-attachments/d03b02ff-a0fc-46c3-a741-b3b005214faa.png)
a) BCC [1 0 0]
b) BCC [1 1 0]
c) BCC [1 1 1]
d) FCC [1 0 0]
e) FCC[1 1 0]
f) FCC [1 1 1]
b) BCC [1 1 0]
Allotropy is:
a) the process by which an element transitions from a solid state directly into a gaseous state without becoming a liquid.
b) financial support ordered by a court following divorce.
c) a story that contains a hidden meaning, typically a moral one.
d) when an elemental solid can have more than one crystal structure; also known as polymorphism.
d) when an elemental solid can have more than one crystal structure; also known as polymorphism.
What is the atomic packing factor (APF) for body-centered cubic (BCC)?
a) 0.74
b) 0.82
c) 0.68
d) 0.52
c) 0.68
Which atomic bonding is characterized by the sharing of valence electrons between adjacent atoms?
a) Metallic bond
b) Hydrogen bond
c) Covalent bond
d) Dipole-dipole bond
e) Ionic bond
c) Covalent bond
Which of the following are considered linear defects in crystals (Choose all that apply)?
a) a kink defect
b) a vacancy
c) an interstitial atom
d) an edge dislocation
c) a screw dislocation
both d and c
The atomic mass of an atom may be expressed as the sum of the masses of (Choose all that apply):
a) electrons
b) xenons
c) neutrons
d) protons
d) neurons
both c and d
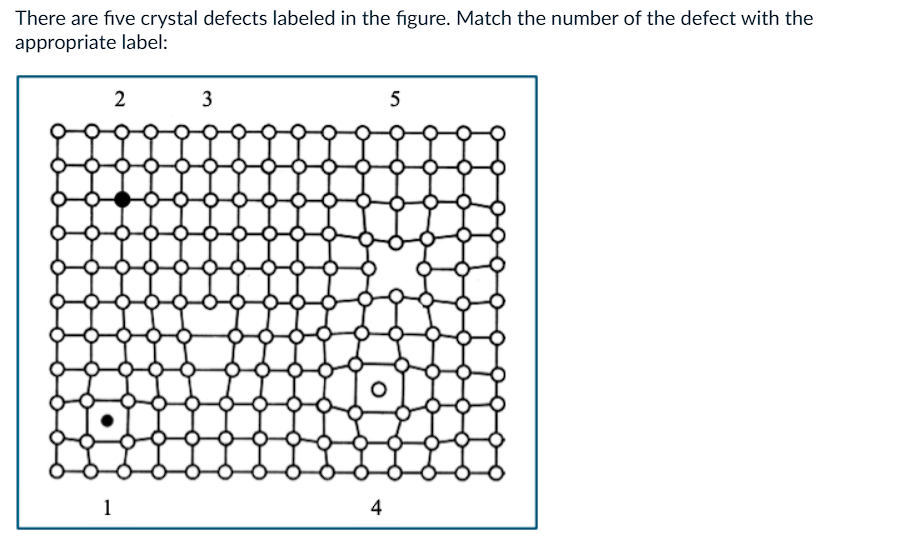
What is 1?
a) edge dislocation
b) self interstitial
c) substitutional atom
d) vacancy
e) impurity interstitial
e) impurity interstitial
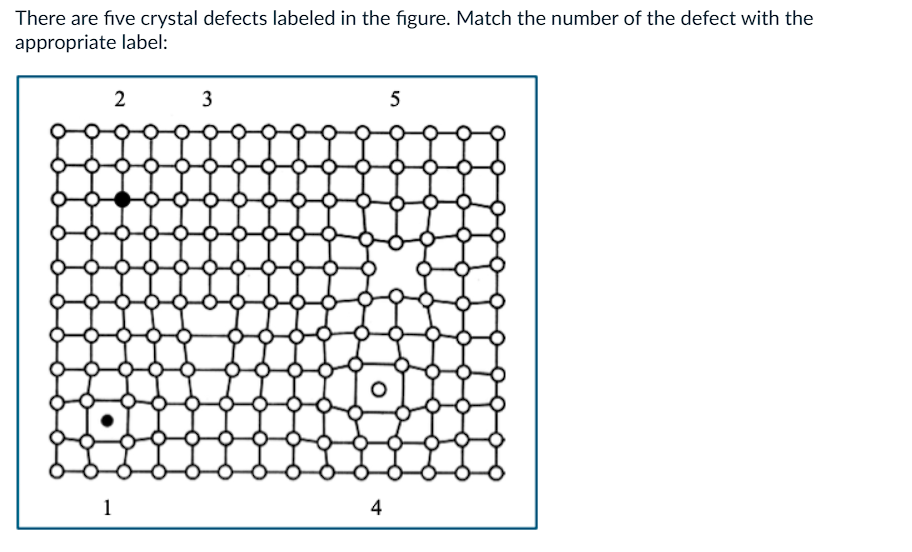
What is 2?
a) edge dislocation
b) self interstitial
c) substitutional atom
d) vacancy
e) impurity interstitial
c) substitutional atom
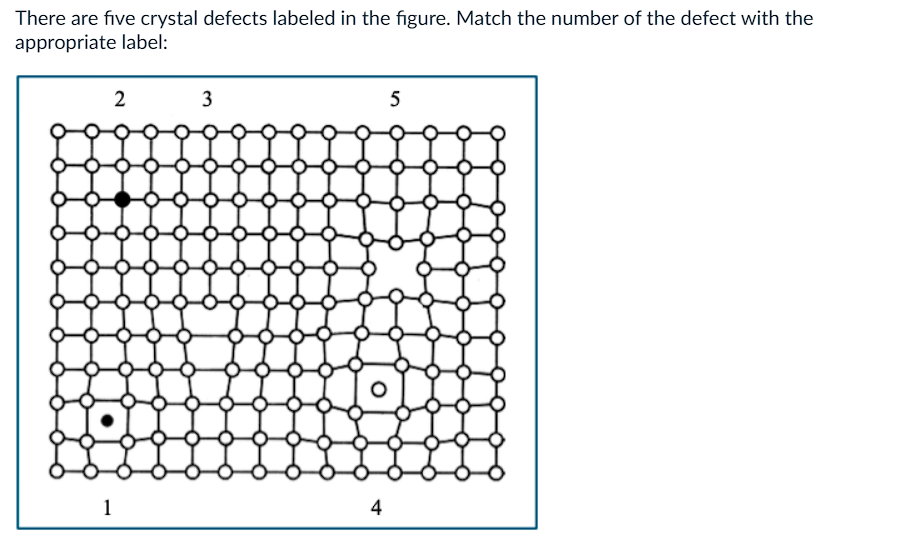
What is 3?
a) edge dislocation
b) self interstitial
c) substitutional atom
d) vacancy
e) impurity interstitial
a) edge dislocation
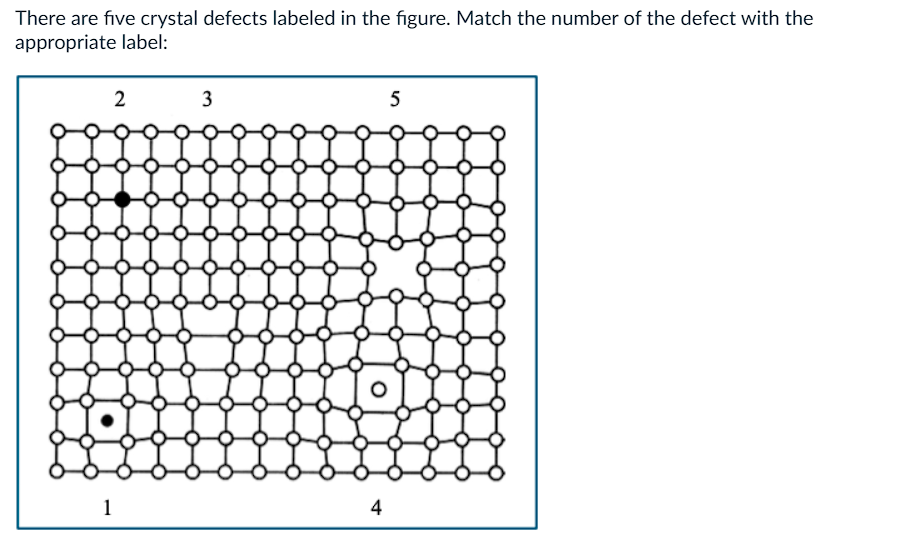
What is 4?
a) edge dislocation
b) self interstitial
c) substitutional atom
d) vacancy
e) impurity interstitial
b) self interstitial
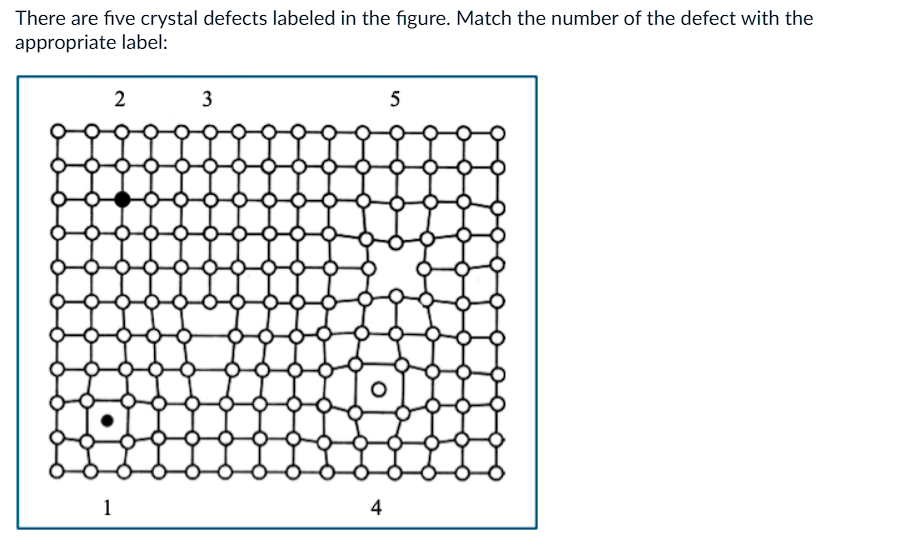
What is 5?
a) edge dislocation
b) self interstitial
c) substitutional atom
d) vacancy
e) impurity interstitial
d) vacancy
The performance of a material is independent of its internal structure and is only determined by its processing.
a) True
b) False
b) False
Noncrystalline (amorphous) materials possess a long-range periodic order of atoms throughout their entire volume.
a) True
b) False
b) False
Grain boundaries are considered material defects because they represent the interface where two crystals of different orientations meet.
a) True
b) False
a) True
In a cubic unit cell, the [110] direction is perpendicular to the (110) plane.
a) True
b) False
a) True
As the temperature of a metal increases, the vacancy concentration increases.
a) True
b) False
a) True
One "Hume-Rothery Rule" states that for two metals to have total solubility, their atomic radii must differ by no more than 5%.
a) True
b) False
b) False
For a given material, a higher bonding energy typically correlates to a lower melting temperature.
a) True
b) False
b) False
Secondary bonds (Van der Waals bonds) are generally stronger than primary bonds (ionic, covalent, and metallic) because they rely on dipole interactions.
a) True
b) False
b) False
Dislocations motion is the mechanism by which plastic deformation occurs.
a) True
b) False
a) True
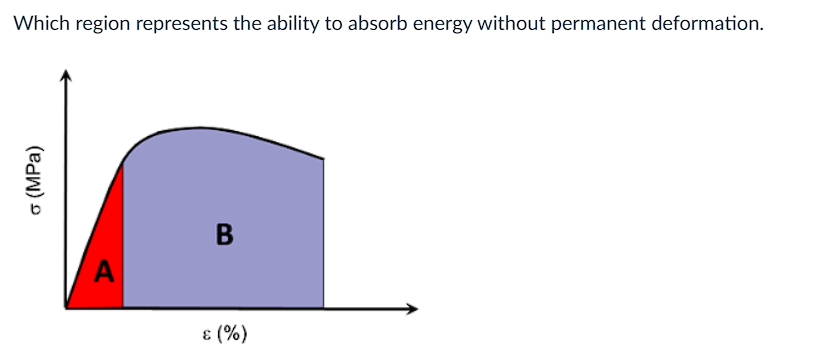
a) A
b) B
c) both
d) none
a) A
How does grain size influence the strength of a polycrystalline material?
a) fine < course
b) fine = course
c) fine > course
c) fine > course
On a microscopic level, the yield stress is defined as the minimum stress to:
a) break all the bonds in the crystal structure
b) increase distance between two atoms in a crystal
c) cause dislocation motion
d) increase vacancy concentration
d) none of the above
c) cause dislocation motion

all answers are in cm²/s:
a) D = 4E-19
b) D = 4E-17
c) D = 4E-15
d) D = 4E-13
e) D = 4E-11
b) D = 4E-17
Which of the following will NOT extend fatigue life?
a) imposing surface residual compressive stress by shot peening
b) decreasing the mean stress level
c) creating sharp surface discontinuities
d) improving surface finish
e) case hardening using carburizing or nitriding
c) creating sharp surface discontinuities
A plate of an alloy steel has a plane-strain fracture toughness of 50 MPa-m1/2. If it is known that the largest surface crack is 0.5 mm long, and that the value of Y is 1.1, which of the following can be said about this plate when a tensile stress of 1200 MPa is applied?
a) the plate will definitely fracture
b) the plate will definitely not fracture
c) It is not possible to determine whether or not the plate will fracture
a) the plate will definitely fracture
What is the correct order of changes that occur for annealing of a metal?
a) Cold work, recovery, crystallization, grain growth
b) Cold work, recuperation, recrystallization, grain growth
c) Recovery, cold work,, grain growth, recrystallization
d) Cold work, recovery, recrystallization, grain growth
e) Grain growth, recrystallization, recovery, cold work
d) Cold work, recovery, recrystallization, grain growth
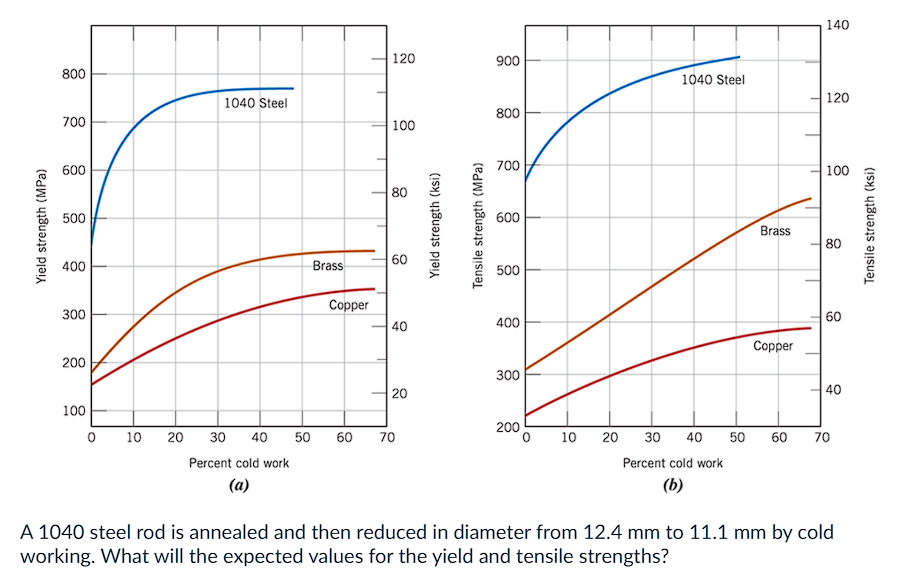
a) YS = 694 MPa; UTS = 787 MPa
b) YS = 787 MPa; UTS = 694 MPa
c) YS = 746 MPa; UTS = 834 MPa
d) YS = 450 MPa; UTS = 675 MPa
c) YS = 746 MPa; UTS = 834 MPa
How does the theoretical strength of a solid material compare with its experimental strength?
a) theoretical < experimental
b) theoretical = experimental
c) theoretical > experimental
c) theoretical > experimental