Lecture 11 Key notes
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Phases are at equilibrium if they have te same
temperature, pressure, chemical potential
Metastable
sold phases are in contact for a long time but dont reach equilibrium (supersaturated); arent in equilibrium but chemical potential is not enough to force it
Interstitial solid solutions typically have solute solubility limits of
<10%
Substitution solid solubility limits depend on
atomic size, crystal structure, electronegativity, valence electrons
Isomorphous systems
completely soluble across all compositions NO MATTER what ratio
Liquidis Line
freezing point, liquid goes to solid, the top upper line
Solidis Line
melting point, solid to liquid, seperates Liquid from mixed solids,
Equlibrium cooling
slow cooling that follows liquidis (top line)
slow cooling = more time for diffusion = homogeneous crystal composition
Non equilibrium cooling
faster cooling, not enough time for diffusion to make nice homogeneous crystal
cored structure (rings of growth like an onion)
segregation (different rings have different compositions of solid)
Homogenization Het treatment
hold metal just below solidious temperature (melting point) to allow for diffusion that eliminates segregation/coring problems in non-equilibrium cooling
Alloys have __________ dependent solid solution hardening
concentration; results in loss of ductility
Laminar Structure
zebra like structure
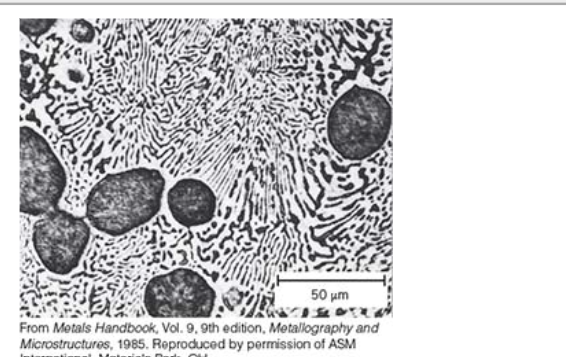
Eutectic Structure
nucleation happens. As nucleation sites grow they bump into each other, expand, and form lines
more nucleation = finer lines
Eutectic Point
Liquid → solid 1 + solid 2
Peritectic
Liquid + solid → solid
Eutectoid
solid 1 → solid 2 + solid 3
Peritectoid
solid 1 + solid 2 → solid 3
Phase transformation helps
Start of Word: Eu = one transforms in two things; Peri = two things converge into one
End of Word: ic = liquids, oid = solids
Intermatallic Compounds
compounds of 2 distinct metals w/ distinct chemical formula
**SOLID VERTICAL LINE on phase diagrams
Congruent Phase Transformation
no change in composition of phases involved (allotropic transformations and melting pure materials)
Incongruent Transformation
at least 1 phase changes composition (eutectic or eutectoid)
Questions → hw 8,
question 2c (actual 2c)
question 6 (Why not just alpha - co / alpha - eutectoid)
question 5 (how is it that the point is a peritectic, phase transformations just questions)
need to complete → 5 and 6