SRS Unit 1.2 ATAR Physics - Ionising Radiation & Nuclear Reactionc
1/45
Earn XP
Description and Tags
WIP - til Decay Series
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
Bohr’s Atomic Model
The nuclear model where protons and neutrons are contained in a nucleus and electrons are orbiting it in shells
Proton
Charge:
+ 1.602×10−19 𝐶
+1 𝑒
Location:
Nucleus
Mass:
1.6726 ×10−27 𝑘𝑔
1.007276 𝑎𝑚𝑢
Neutron
Charge:
0C
0 e
Location:
Nucleus
Mass:
1.6749 ×10−27 𝑘𝑔
1.008665 𝑎𝑚𝑢
Electron
Charge:
−1.602×10−19 𝐶
−1 𝑒
Location:
Outside nucleus
Mass:
9.1094 ×10−31 𝑘𝑔
0.000549 𝑎𝑚𝑢
Nucleons
A collective term for protons and neutrons found in the nucleus.
Nuclides
The range of atomic nuclei associated with a particular atom, which is defined by the atomic number and the various isotopes of that atom as identified by the mass number.
Isotopes
Two or more types of atoms that have the same atomic number and position in the periodic table but different number of neutrons.
Atomic Number (Z)
The number of protons that defines the element
Atomic Mass Number
The total mass of protons (Z) and neutrons (N) of an atom in Atomic Mass Units (amu)
Neutron Number (N)
Atomic Mass Number (rounded) - Atomic Number
Electrostatic Force (EF)
An attractive and repulsive force between particles caused by their electric charges
Like charges repel, opposites attract
Also referred to as Coulomb’s force
Strong Nuclear Force (SNF)
The “glue” holding protons and electrons together in the nucleus together
Exists within quarks, which form protons and neutrons
SNF > EF
If the Strong Nuclear Force is stronger than the Electrostatic Force, nuclei will be stable
However, if this delicate balance is interfered with, nuclei will become unstable and decay
Stable nuclide
A nuclide that will never (or close to never) decay
Unstable nuclide
A nuclide that will decay
It will keep decaying until the balance of protons and neutrons is stable
Radioactive decay
The process by which an unstable atomic nucleus loses energy by radiation and the emission of subatomic particles
Radioisotope
A radioactive isotope
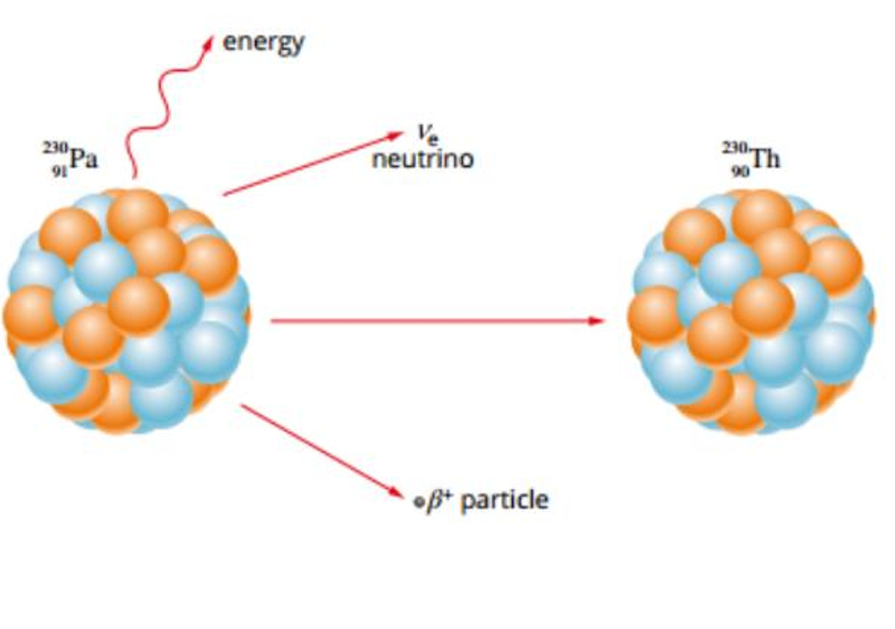
Beta Positive/Plus Decay (β+)
Occurs when the nucleus has too many protons
Therefore the Electrostatic Force overpowers the Strong Nuclear Force
The proton decays into a neutron to release excess EF and stabilise the nucleus
Due to this, the nucleus emits a positron (anti-electron) known as a Beta Positive Particle along with a neutrino
Beta Negative/Minus Decay (β-)
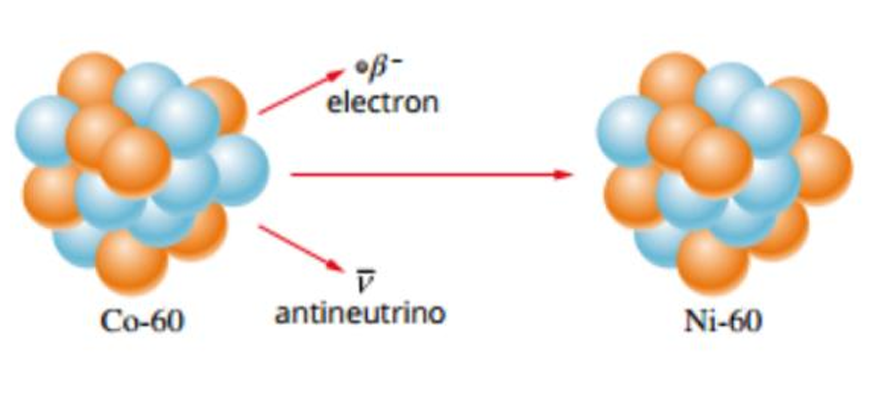
Occurs when the nucleus has too many neutrons
Therefore the Strong Nuclear Force overpowers the Electrostatic Force
The neutron decays into a proton to gain additional EF and stabilise the nucleus
Due to this, the nucleus emits an electron known as a Beta Negative Particle along with an anti-neutrino
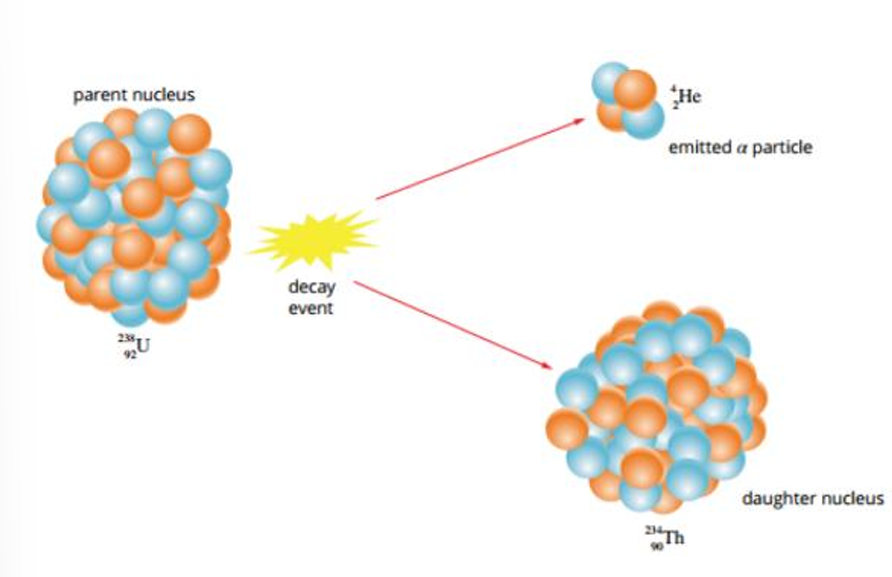
Alpha Decay (α)
Occurs when the nucleus is too heavy to naturally be stable (every element above Bismuth)
The nucleus releases two protons and two neutrons at the same time - an Alpha Particle
An Alpha Particle is the same as a Helium-4 nucleus
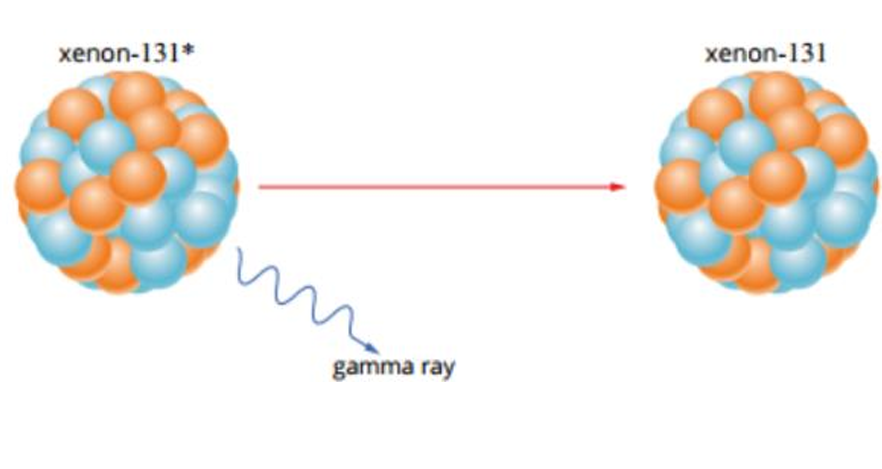
Gamma Decay (γ)
Occurs when nucleus has excess energy but the correct ratio of protons to neutrons
The nucleus emits energy Gamma Radiation - gamma waves
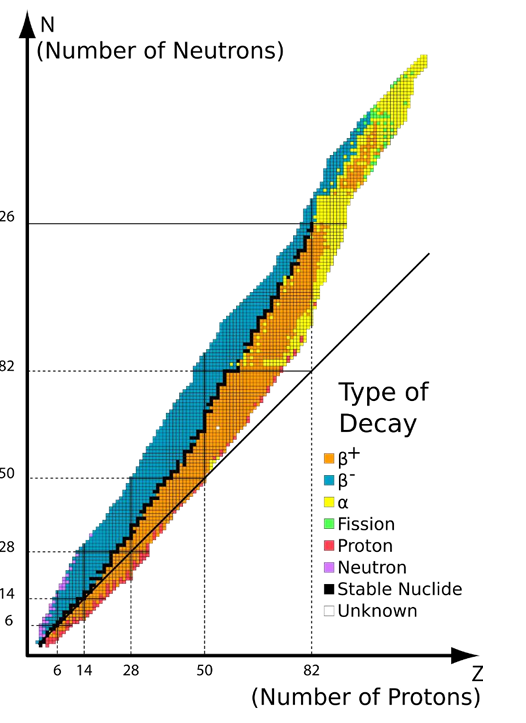
Segre Plot
A graph/visualisation of the stability in nuclei showing all known isotopes of each element and their radioactivity/stability
y axis is number of neutrons (N), x axis is number of protons (Z)
Beyond element 82 (Lead) there are no more stable isotopes
The number of radioactive isotopes for each element exceeds the number of stable ones
Penetrating ability
How well radiation can pierce through matter
Ionisation ability
How well radiation can ionise atoms
Ionisation is the process by which an atom loses or gains an electron, thus resulting in a net positive or negative charge
Alpha Particle (α)
Mass:
4.001 506 u
Charge:
+2e
Penetrating Ability:
Low
Ionisation Ability:
High
Beta Positive/Plus Particle (positron) (β+)
Mass:
0.000549 u
Charge:
+1e
Penetrating Ability:
Medium
Ionisation Ability:
Medium
Beta Negative/Minus Particle (electron) (β-)
Mass:
0.000549 u
Charge:
-1e
Penetrating Ability:
Medium
Ionisation Ability:
Medium
Gamma Particle (γ)
High energy light/electromagnetic wave
Mass:
0 u
Charge:
0e
Penetrating Ability:
High
Ionisation Ability:
Low
Can only ionise atoms if it strikes an electron directly and knocks it loose
Nuclear Equation formats
LHS → RHS
Reactants → Products
Parent Nucleus → Daughter Nucleus + decay particles + energy
Alpha Decay Equation
PN → DN + α + energy
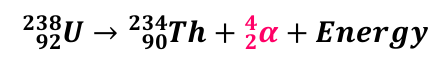
Beta Positive/Plus Decay Equation
PN → DN + β+ + ve + energy
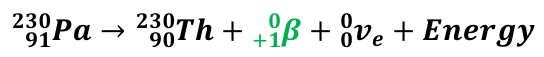
Beta Negative/Minus Decay Equation
PN → DN + β- + ve + energy
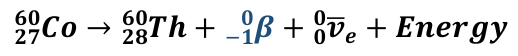
Gamma Decay Equation
PN* → PN + γ + energy
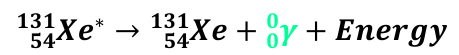
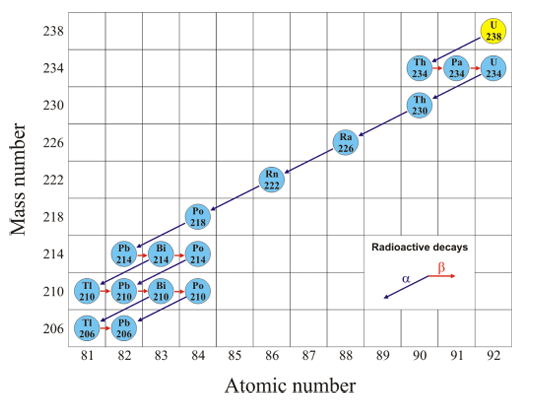
Decay Series
A graph visualising the different decays an isotope undergoes to achieve stability
Each isotope has their own “signature” sequence
y-axis: mass number
x-axis: atomic number
Diagonal line → alpha decay
Horizontal line → beta decay
Series stops at Lead-206
Half-life (t1/2)
The time required for a substance to decay to half its initial size
Graphically, its trend line is mathematical exponential decay
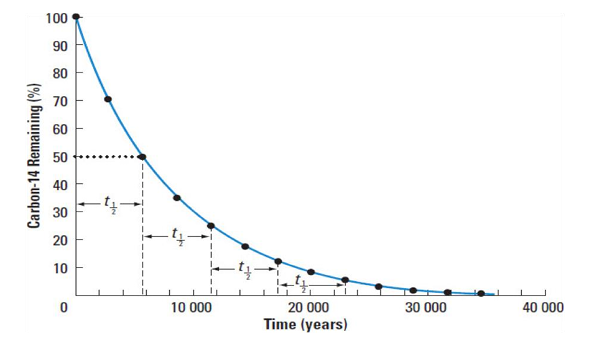
Equation for half-life
N = N0 × (1/2)n
N = Final amount of substance / no. of remaining atoms
N0 = Initial amount of substance / no. of remaining atoms
n is the number of half-lives that have elapsed
n = (time elapsed)/(time of half-life) = t/t1/2
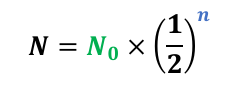
Equation for number of half-lives elapsed
n = t/t1/2
n = number of half-lives elapsed
t = time elapsed
t1/2 = time of half-life
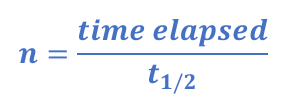
Atomic Mass Unit
Defined as 1/12 of the mass of a Carbon-12 nucleus
1u = 1.66054 × 10-27kg
Electron volts
The preferred units of Energy on the atomic scale
Equal to the energy gained by an electron when passing a potential difference of 1 Volt
1eV = 1.602 × 10-19J
1J = 6.242 × 1018eV
1 megaelectron volt is 1,000,000 eV
Equation for Mass-Energy Equivalence
E = mc2
E = energy (J or eV)
m = mass (kg or u)
c = the speed of light (299 792 458 m/s)
1u = 931.6 MeV
Mass Defect
Mass of nucleons together < mass of nucleons apart
Mass defect (∆m) is usually measured in atomic mass units (u)
∆m = mass of nucleons − total mass of individual nucleons
This mass is converted into binding energy
Binding Energy
The mass of individual nucleons that is converted in energy to keep them together
Binding Energy = ∆m × 931.6
Calculating energy released from Alpha Decay
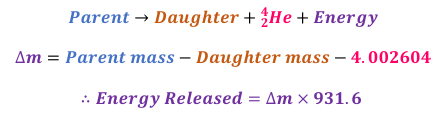
Calculating energy released from Beta Plus Decay
The value of 0.0010972 is twice the mass of an electron and is due to the mass of the positron released by the parent nucleus and the one fewer electron in the daughter isotope
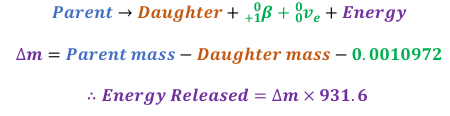
Calculating energy released from Beta Minus Decay
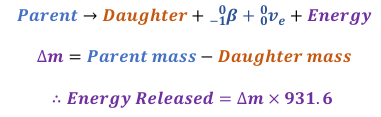
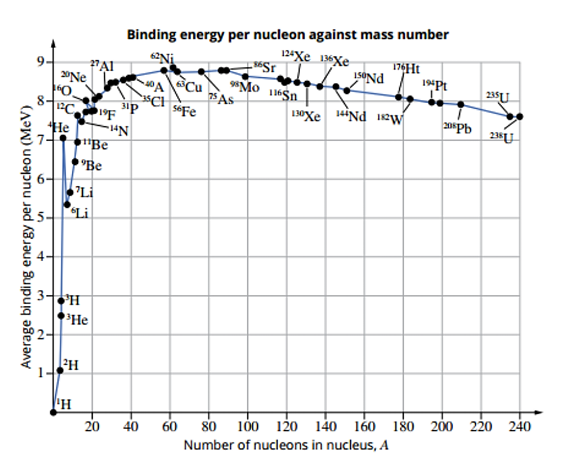
Binding Energy Per Nucleon
A measurement of stability in the nucleus
The higher the value, the more stable the nucleus will be