Organic chem: PART 1
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What are organic compounds?
Compounds that contain carbon atoms (except carbon dioxide, carbon monoxide and carbonate salts)
What are the simplest organic compounds?
Molecules that only contain carbon and hydrogen atoms bonded together
What are hydrocarbons?
Molecules that only contain carbon and hydrogen atoms bonded together
How are carbon and hydrogen atoms held together in hydrocarbons?
Through covalent bonding
What is covalent bonding?
The electrostatic force of attraction between the positive nuclei of two non-metal atoms and the negative shared pair of electrons
What is a displayed formula?
A 2D formula that shows how all the atoms in an organic molecule are bonded together
When would hydrocarbons be described as saturated?
If they contain ONLY carbon-carbon SINGLE bonds, so the molecule holds the max possible number of hydrogen atoms for a carbon chain
What are alkanes?
Non-cyclic hydrocarbons that contain only SINGLE c-c bonds
What are cycloalkanes?
Cyclic hydrocarbons that contain only SINGLE c-c bonds
What is the shape of the alkane display?
Straight, branched
What is the shape of the cycloalkane display?
Cyclic, ring
What is the alkanes general formula?
Cn H2n+2
What is the cycloalkanes general formula?
Cn H2n
What is the name of CH4?
Methane
What is C2H6?
Ethane
What is C3H8?
Propane
What is C4H10?
Butane
What is C5H12?
Pentane
What is C6H14?
Hexane
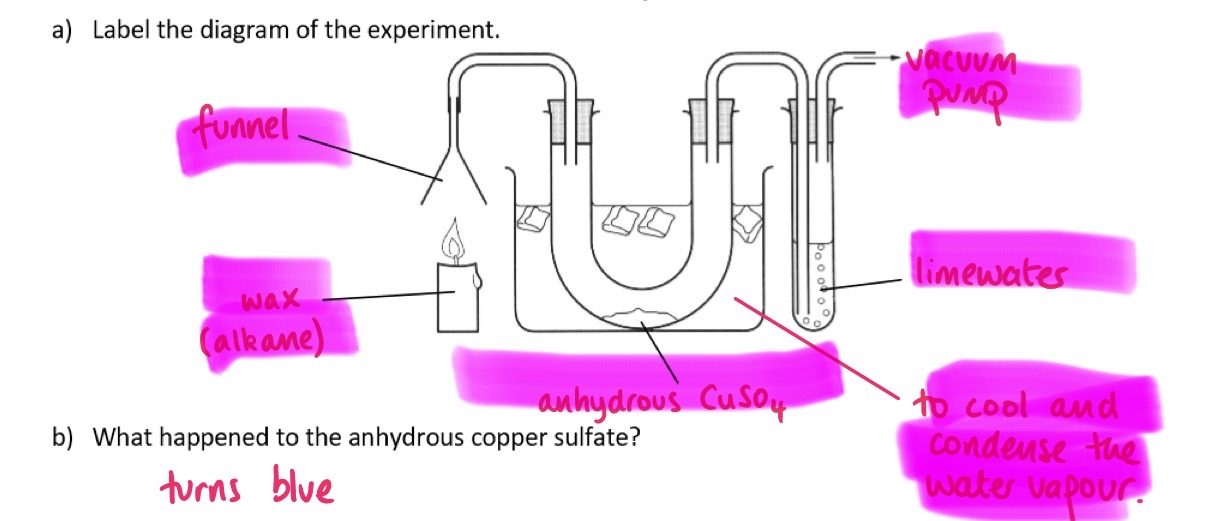
What equipment is required for a hydrogen combustion?
Funnel, wax (alkane), Anhydrous CuSO4, Vacuum pump, Limewater, cold water
Why is the combustion experiment set up in a cold water bath?
To cool and condense the water vapour
What type of reaction is a combustion reaction an example of?
An oxidation reaction - the C and H atoms from the hydrocarbon bond with O atoms
What is a structural formula?
A formula used to show how all the atoms in an organic molecule are bonded together

Explain why methane has a very low boiling point of -162 degrees C?
Methane has a simple molecular structure
There are forces of attraction between the molecules which are weak
Little energy needed to overcome them
Why are hexane and 3-methylpentane not easy to separate?
The boiling points are very similar
What can fractional distillation be used for?
Fractional distillation can be used to separate a mixture of hydrocarbons which have different boiling points
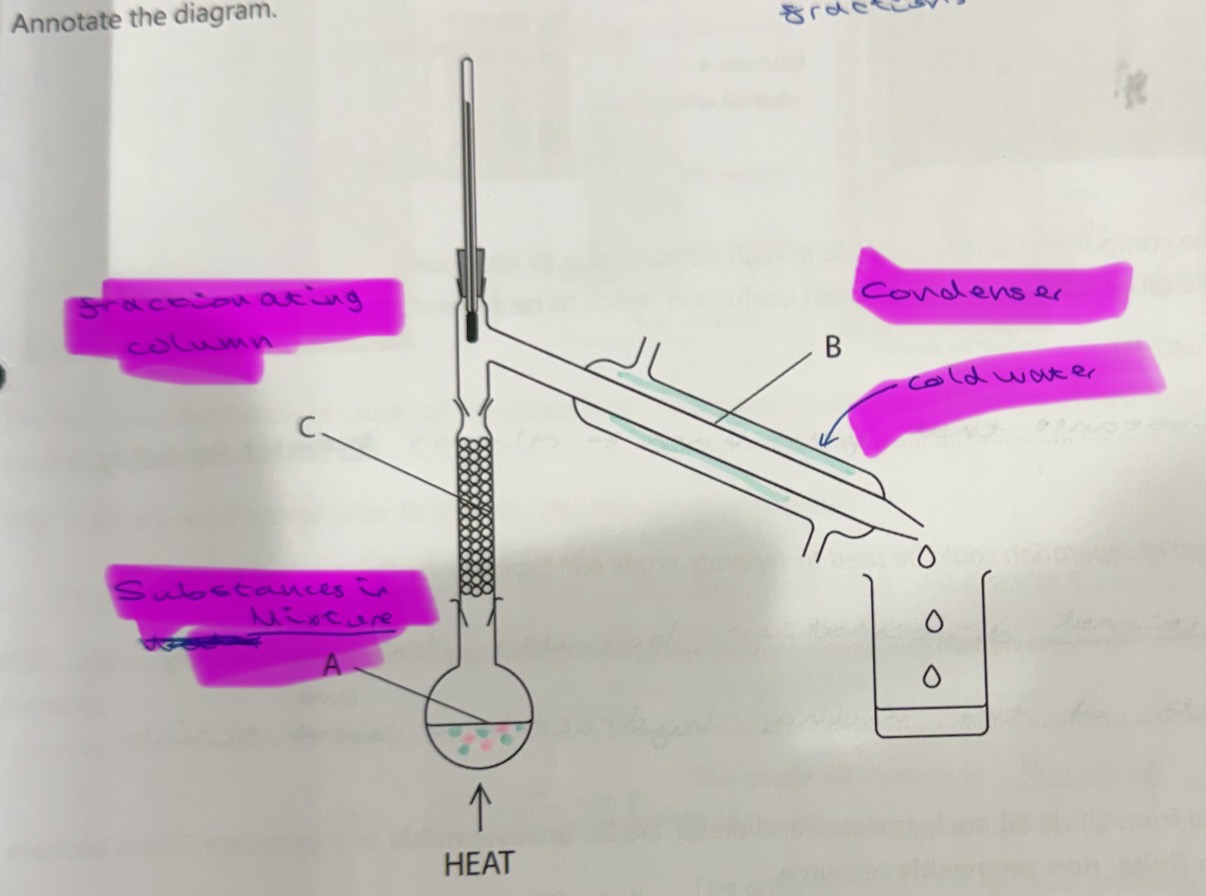
What equipment is required for fractional distillation?
Mixture (containing substances), fractionating column, Condenser
How does fractional distillation work?
Separates mixture into fractions
Mixture is heated and substances vaporise
Vapours of substance rise and the lowest boiling point substance reaches the top of the gractionating column first
Vapours cool and condense into a liquid in the condenser
Separate liquids are collected
What is crude oil?
A mixture of many different hydrocarbons with molecules containing 1-80 Carbon atoms - it has a thick gloopy texture
What is fuel?
A substance that you burn to release heat energy
What are the main problems associated with burning hydrocarbon fuels?
Release of CO2, emission of greenhouse gases, carbon monoxide, nitrous oxides, sulfur dioxide
Petrol and diesel are unsustainable as crude oil is finite