Topic 6 - Radioactivity
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
describe an atom
a positively charged nucleus, consisting of protons and neutrons, surrounded by negatively charged electrons in shells which represent different energy levels, with the nuclear radius much smaller than that of the atom and with almost all of the mass in the nucleus
How does the mass and size of the nucleus compare to that of the entire atom?
Almost all the mass of the atom is contained in the nucleus
The nucleus is much smaller than the atom (nucleus ~10⁻¹⁵ m, atom ~10⁻¹⁰ m)
What is the approximate size of an atom and nucleus?
nucleus ~10⁻¹⁵ m, atom ~10⁻¹⁰ m
What does the atomic number refer to?
Number of protons in the nucleus
How can you tell how many neutrons are present by looking at the mass number and atomic number of an element?
Number of neutrons = mass number − atomic number
Explain why an atom is neutral.
The number of proton and electrons is the same. The electrons have a -1 charge and the protons have a +1 charge. So overall these charges cancel out to neutral.
What is meant by an isotope?
atoms of the same element that have the same number of protons but different numbers of neutrons
What is the relative mass and charge for a proton?
Mass = 1, charge = +1
What is the relative mass and charge for a neutron?
Mass = 1, charge = 0
What is the relative mass and charge for an electron?
Mass = 1/1836, charge = −1
what is a positron
the antiparticle of an electron - it has the same mass as an electron, and the same size of charge, however it has a positive charge
What is the relative mass and charge for a positron?
Mass = 1/1836, charge = +1
Explain how an electron can become excited in an atom.
When an electron is excited it moves up to a higher energy level by absorbing EM radiation of a specific frequency. (aka absorption). This EM radiation correspond to the exact change in energy level. A bigger change in energy will correspond to a higher frequency light absorbed.
Explain how an electron can become de-excited in an atom.
When an electron is de-excited, it moves down to a lower energy level by emitting EM radiation of a specific frequency. (aka emission). This EM radiation corresponds to the exact change in energy level. A bigger change in energy will correspond to a higher frequency light emitted.
Explain how an atom may form a positive ion by losing outer electrons.
If an atom absorbs ionising radiation, the electron will be excited and have enough energy to leave the atom. Electrons can also leave the atom due to friction or by chemical reactions.This will leave more positive protons in the nucleus than negative electrons in the surrounding shells, leaving an overall positive charge. (An Ion)
Describe the plum pudding model of the atom.
the model made by JJ. Thompson, the atom was thought to consist of negatively charged electrons (the ‘plums’) in a positively charged ‘dough’ or ‘pudding’ - the atom was thought to be overall neutral as the negative electrons balanced out the positive ‘dough’
Describe the Rutherford model of the atom. (the nuclear model)
A small dense positively charged nucleus surrounded by orbiting electrons.
Describe the Bohr Model of the atom
electrons orbit the nucleus at different distances, which are discrete energy levels, and the atom has a small, dense, positively charged nucleus at the centre
describe rutherford’s alpha particle experiment
rutherford directed a beam of positively charged alpha particles at a thin gold foil and around the foil, he placed a fluorescent screen to detect where the alpha particles went after hitting the foil
In Rutherford's experiment most alpha particles passed straight through. What does this tell you about the atom?
Most of the atom is empty space.
In Rutherford's experiment some alpha particles are deflected by small angles. What does this tell you about the atom?
The nucleus is small and positive.
In Rutherford's experiment very few alpha particles are deflected back. What does this tell you about the atom?
Most of the atom's mass is concentrated in the nucleus. The nucleus is small and dense.
describe radioactive decay
this is when an unstable nucleus emits radiation in a random process which lowers the overall energy of the nucleus and makes it more stable
Describe the nature of an alpha particle
an alpha particle is the same as a helium nucleus, as it contains 2 protons and 2 neutrons. it has a charge of +2
Describe the nature of a beta minus particle
high-energy, high-speed electrons which are emitted from the nucleus. they are produced in a nucleus when a neutron turns into a proton and an electron. they have a charge of -1
Describe the nature of a beta plus particle
high-energy, high-speed positrons which are emitted from the nucleus. they are produced in a nucleus when a proton turns into a neutron and a positron (positive version of an electron). they have a charge of +1
Describe the nature of gamma radiation
High energy and high frequency electromagnetic (EM) wave. they have a charge of 0.
Describe what happens in the nucleus during alpha decay
The nucleus loses two protons and two neutrons
The atomic (proton) number decreases by 2
The mass (nucleon) number decreases by 4
what would an alpha decay equation look like
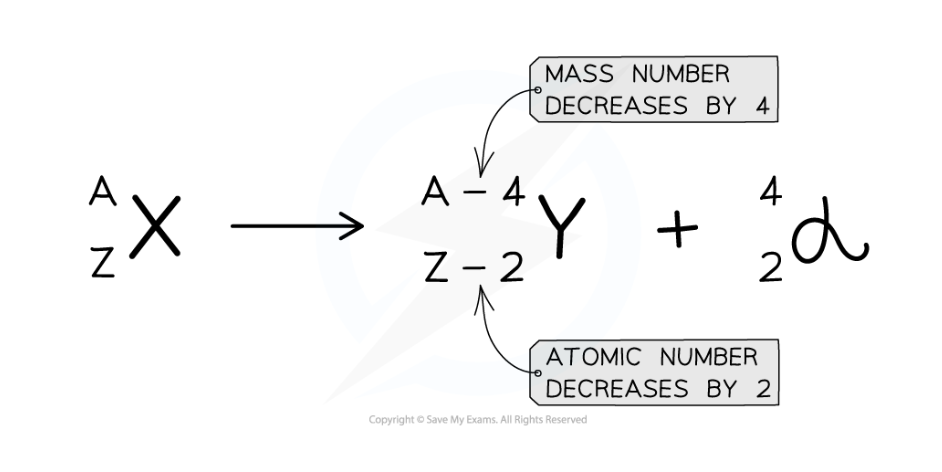
Describe what happens in the nucleus during beta minus decay
A neutron turns into a proton and an electron. The electron is emitted and the proton remains in the nucleus
The atomic (proton) number increases by 1
The mass (nucleon) number does not change
what would the equation for beta minus decay look like
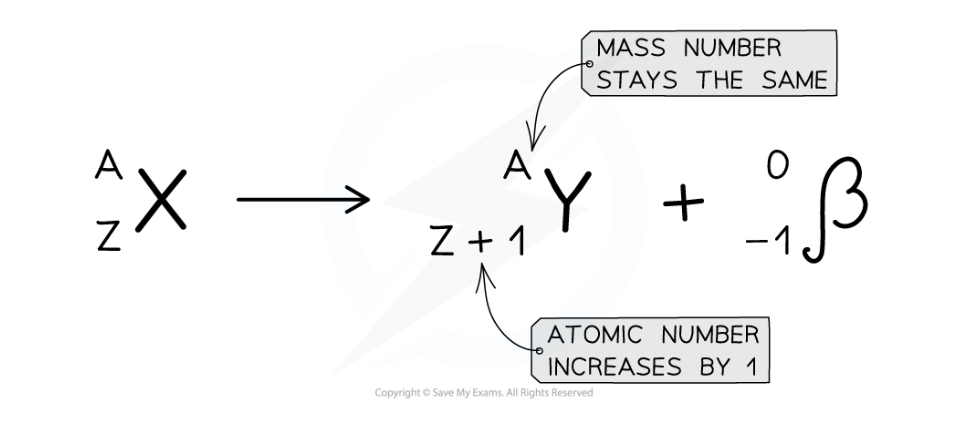
Describe what happens in the nucleus during beta plus decay
A proton turns into a neutron and a positron. The positron is emitted and the neutron remains in the nucleus
The atomic (proton) number decreases by 1
The mass (nucleon) number does not change
what would the equation for beta plus decay look like
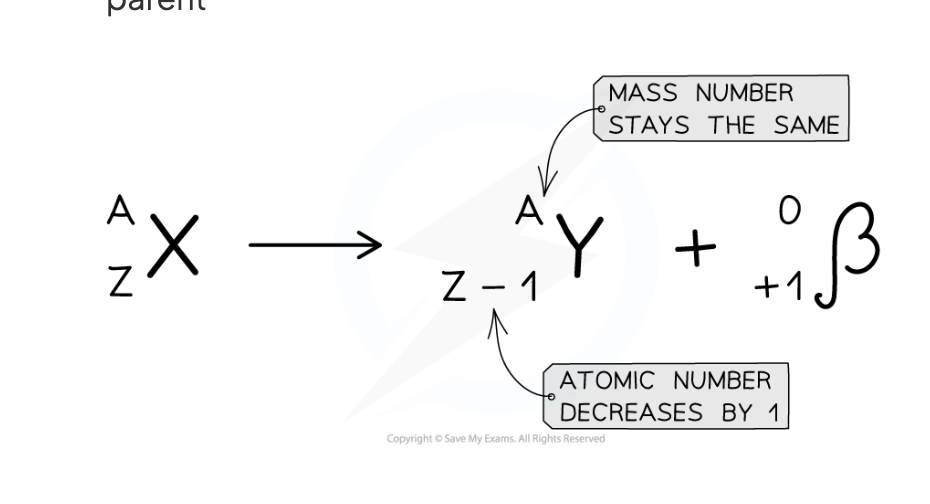
Describe what happens in the nucleus during gamma decay
The nucleus undergoes nuclear rearrangement with a loss of energy as gamma radiation
The atomic (proton) and mass (nucleon) numbers do not change
what would the equation for gamma decay look like
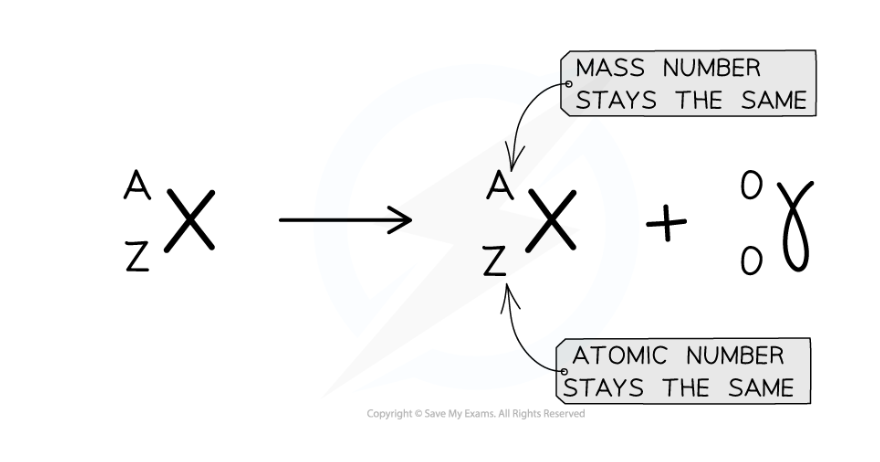
Describe what happens in the nucleus during neutron emission
The nucleus loses one neutron - The element remains unchanged, it is now a different isotope.
The atomic (proton) number does not change
The mass (nucleon) number decreases by 1
What must be the same on both sides of a decay equation?
Mass and charge must be conserved in a decay equation.
List alpha, beta (minus) and gamma in order of least to most ionising
Gamma, beta, alpha
why is alpha good at ionising
due to them being emitted at high speeds and having a high relative mass, they transfer a lot of energy
List alpha, beta (minus) and gamma in order of least to most penetrating
Alpha, beta, gamma
why is gamma good at penetrating
Because gamma rays have no electrical charge, they are not attracted to or repelled by the electrons or nuclei of atoms in the material they are passing through. This lack of interaction means they don't easily get slowed down or stopped.
List alpha, beta (minus) and gamma in order of least to most dangerous inside the body
Gamma, beta, alpha
List alpha, beta (minus) and gamma in order of least to most dangerous outside of the body
Alpha, beta, gamma
What stops alpha radiation?
2-3cm of air or a sheet of paper
What stops beta (minus) radiation?
3-5mm of Aluminium
What stops gamma radiation?
reduced by few mm of lead or several metres of concrete
link between ionising power and range in air
The more ionising a form of radiation is, the sooner it will react with the air it is moving through
Alpha, beta and gamma radiation are ionising. What is meant by this?
They have enough energy to knock electrons off of atoms which produces an ion (an atom with a non-zero charge)
What is meant by background radiation?
Ionising radiation that is present in the environment (can be natural or manmade). It is always present in experiments and must be accounted for.
give examples of natural sources of background radiation
radon gas from rocks and soil, cosmic rays from space, carbon-14 in biological material, radioactive material in food or drink
give examples of man-made sources of background radiation
medical sources, nuclear waste, nuclear fallout from nuclear weapons, nuclear accidents (e.g chernobyl)
Describe how to measure the background radiation count rate.
Use a Geiger-Muller tube connected to a counter and turn it on for 5 minutes. Each time it absorbs radiation, it transmits an electrical pulse to a counting machine. Divide the total count by 5 to give the rate in counts per minute. Repeat this and take an average.
How does photographic film work?
Photographic film changes colour when ionising radiation is absorbed. It measures total radiation dose received. Employees in nuclear power plants wear them and if it darkens to a certain degree they may be redistributed to not expose them to a dangerous dose of radiation.
what does activity mean
The rate at which the unstable nuclei from a source of radiation decays
What is the unit given to the activity of a radioactive source?
becquerels (Bq) for source activity (disintegrations per second) and sieverts (Sv) for biological health effects (effective dose)
what does a result of 1 Becquerel mean?
1 Becquerel is equal to 1 nucleus in the source decaying in 1 second
how to find number of atoms decaying from activity and time?
number of atoms decaying = activity (Bq) x time (s)
What is meant by the phrase 'radioactive decay occurs randomly'?
There is an equal probability of any nucleus decaying
It cannot be known which particular nucleus will decay next
It cannot be known at what time a particular nucleus will decay
The rate of decay is unaffected by the surrounding conditions
It is only possible to estimate the probability of a certain nucleus decaying in a given time period
Describe how to measure the activity of a radioactive source.
Put the radioactive source (Ensuring no others are nearby) in front of a Geiger-Muller tube connected to a counter. Turn it on for 5 minutes (measured with a stopwatch) and then divide total count by 5 to give the rate in counts per minute. Repeat and take an average. You must then subtract the background rate from your average to give the activity of the source.
Sketch a graph to show how the activity of a radioactive source varies with time.
An exponential decay curve drawn. The line must touch the y axis at time = 0, but the line does not touch the x axis
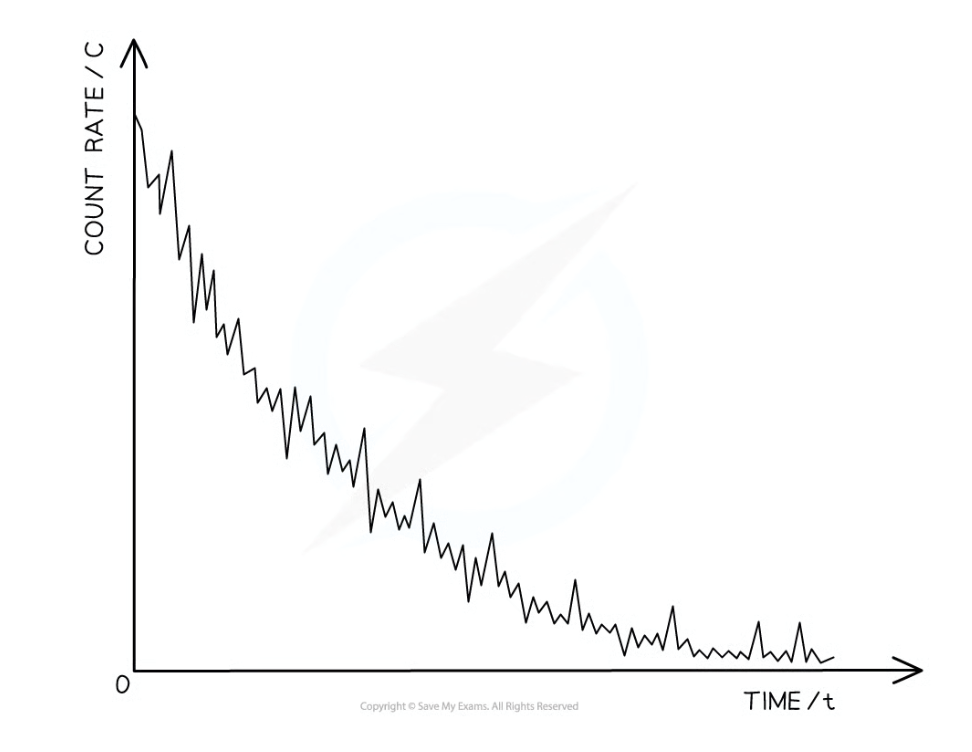
What is the definition of half-life?
The time taken for half the undecayed nuclei to decay or the activity of a source to decay by half
how does half-life work in calculations
if a half life is 10 hours, then it would take 10 hours for the activity to be reduced to 50%, 20 hours for the activity to be reduced to 25%, 30 hours to be reduced to 12.5%, etc.
If radioactive decay is random, how can we quote the half-life of a radioactive isotope?
There are so many atoms that the random nature of the decay becomes very predictable.
ways radioactivity is used
diagnosis and treatment of cancer
irradiating food
Sterilising medical equipment
Checking the thickness of materials
Smoke detectors (alarms)
Describe how radioactivity is used for smoke alarms
Alpha particles are used in smoke detectors. The alpha radiation will normally ionise the air within the detector, creating a current. The alpha emitter is blocked when smoke enters the detector. The alarm is triggered by a microchip when the sensor no longer detects alpha
Explain the choice of source and half life used in smoke alarms.
Alpha is used as it is very ionising so will create a large enough voltage in the circuit and is not very penetrating so won't leave the smoke detector. A long half life is needed so it doesn't need to be replaced often.
Describe how radioactivity is used for irradiating food and sterilising medical equipment
Gamma rays fired at food or medical equipment which kills the bacteria but doesn't damage the food or equipment (note: the food/equipment is not radioactive afterwards) It is irradiated not contaminated.
Explain the choice of source and half life used in food and sterilising medical equipment
Gamma rays need to be used so they can penetrate the packaging without opening so they remain sterile. A long half life is needed so that it does not need to be replaced often.
Describe how radioactivity is used for tracing leaks
Gamma used in pipes to find leaks by adding a source to the pipe. Where there is a leak more gamma will be detected.
Explain the choice of source and half life used in tracing leaks.
Gamma radiation is used so it can penetrate the grounds. A short half-life is used so that the water's activity returns to safe levels quickly as it has been contaminated.
Describe how radioactivity is used for gauging thickness
As a material moves above a beta source, the particles that are able to penetrate it can be monitored using a detector
If the material gets thicker, more particles will be absorbed, meaning that less will get through
If the material gets thinner ,the opposite happens
Explain the choice of source and half life used in gauging thickness.
Beta radiation is used because it will be partially absorbed by the material
If alpha particles were used all of them would be absorbed and none would get through
If gamma were used almost all of it would get through and the detector would not be able to sense any difference if the thickness were to change
A long half-life is used so it doesn't need to be replaced often.
Describe how a PET scan works.
A patient is injected with a beta plus source. When the beta plus radiation (positron) meets an electron in the surrounding tissue, they annihilate producing two gamma rays moving in opposite directions. The gamma rays are detected by a scanner and can be used to locate the tumour.
Explain the choice of source and half life used in a PET scan.
Gamma rays are highly penetrating and can therefore pass through the body and be detected outside the body. The patient is contaminated so the half life needs to be short to minimise the exposure.
Explain why isotopes used in PET scanners have to be produced nearby
Isotopes used in PET scanners have to be produced nearby as they have a short half-life and so need to still be active when they reach the patient so they can be detected. If it were transported long distances, most of it would decay before it could be used.
Explain how radiotherapy works (treatment of cancer)
Beams of gamma rays are directed at the cancerous tumour. Radiation can kill living cells.
Explain the choice of source and half life used in radiotherapy (treatment of cancer)
Gamma rays are used because they are able to penetrate the body, reaching the tumour. A long half life is used so that it doesn’t need to be replaced often.
Explain why exposure to ionising radiation is dangerous to the cells of living tissue
Radiation is effectively used to destroy cancerous tumour cells. However, it can cause damage to healthy tissue if it is not properly targeted. This is mostly from high-energy radiation such as gamma rays and X-rays
Explain how exposure to radiation could cause mutations
If the atoms that make up a DNA strand are ionised then the DNA strand can be damaged. If the DNA is damaged then the cell may die, or the DNA may be mutated when it reforms. If a mutated cell is able to replicate itself then a tumour may form. This is an example of cancer, which is a significant danger of radiation exposure
Explain the precautions that can be taken to limit the exposure of a person to ionising radiation
Radioactive sources should be kept in a shielded container when not in use, for example, a lead-lined box
Radioactive materials should only be handled when wearing gloves, and with tongs to increase the distance from them
It may be appropriate to wear protective clothing to prevent the body becoming contaminated
The time that a radioactive source is being used for should be limited
Describe the differences between contamination and irradiation effects and compare the hazards associated with these two
Contamination is more dangerous than irradiation. With contamination the radioactive isotope is on the skin/clothing and is continually irradiating the tissues inside the body. Irradiation is being exposed to ionising radiation. This only occurs whilst you are near the source - it doesn't occur if you move away from it.
how to prevent irradiation
Shielding is used to absorb radiation
Lead lined suits are used to reduce irradiation for people working with radioactive materials
The lead absorbs most of the radiation that would otherwise hit the person
how to prevent contamination
an airtight suit is used by people working in an area where there may have been a radiation leak
This prevents radioactive atoms from getting inside the person
Explain why a patient who has had a radioactive tracer inserted may need to be quarantined for a short time afterwards.
The patient is contaminated so could irradiate the people around them so needs to be quarantined until the activity is low enough to be safe.
how could a short half life make a radioactive sample dangerous
If an isotope has a short half-life, the nuclei will decay very quickly
This means that the isotope will emit a lot of radiation in a short amount of time
If only a small amount of the isotope is used, having a short half-life can be advantageous, as the material will quickly lose its radioactivity
If a large amount is used, however, the levels of radiation emitted could make handling the isotope extremely dangerous
how could a long half life make a radioactive sample dangerous
If an isotope has a long half-life then a sample of it will decay slowly
Although it may not emit a lot of radiation, it will remain radioactive for a very long time
Sources with long half-life values present a risk of contamination for a much longer time
Radioactive waste with a long half-life is buried underground to prevent it from being released into the environment
Compare and contrast the treatment of tumours using radiation applied internally or externally
External - Gamma rays can be used to treat cancer by aiming them at the cancer from different angles so it gets a higher dose than the surrounding tissue. This irradiates the patient so the half-life should be long so it doesn't have to be replaced often. Internal - Radioactive seeds are inserted next to the tumour, emitting beta radiation. The patient is contaminated so half life should be short.
Describe the process of nuclear fission
A slow moving neutron is absorbed by a large unstable nucleus which splits to produce two lighter daughter nuclei and two to three neutrons and energy is released. This process occurs in the reactor of nuclear power stations.
explain the process of the nuclear fission of uranium 235
During induced fission, a neutron is absorbed by the uranium-235 nucleus to make uranium-236
This is very unstable and splits by nuclear fission almost immediately, breaking it into two smaller daughter nuclei and 2 or 3 neutrons
These products of the fission reaction move away very quickly
Energy is transferred from the nuclear store to the kinetic store of the products
Eventually this energy can be used to heat water to produce steam to generate electricity within the nuclear power station
Explain the principle of a controlled nuclear chain reaction for nuclear fission.
the neutrons produced by nuclear fission can start another fission reaction, which again creates further excess neutrons
This process is called a chain reaction
Explain the role of control rods.
Control rods are made of boron and absorb neutrons. When lowered, they absorb more neutrons, which decreases the rate of fission, so they aren't absorbed by the uranium nuclei. This slows down the chain reaction, keeping the reaction in control.
Explain the role of the moderator
The moderator, usually made from graphite, slows down neutrons. This allows them to be more easily absorbed by uranium nuclei and speeds up the rate of reaction.
Describe how thermal energy from the chain reaction is used in the generation of electricity in a nuclear power station.
The heat energy is used to heat the boiler. This heats water into steam. This high-pressure steam turns the turbines which turns the generator. When the generator is turned, electrical energy is transferred to the national grid.
Give two advantages of nuclear power
No carbon dioxide is produced; Reliable source of power as it does not rely on weather or location; A small mass of fuel produces a high power; The supply of nuclear fuel is abundant.
Give two disadvantages of nuclear power
Nuclear power stations are expensive to build and decommission; Nuclear waste is radioactive and needs to be stored carefully; Risk of nuclear accident such as Chernobyl.
Explain why nuclear waste is dangerous
Nuclear waste is contaminated and therefore radioactive so produces ionising radiation which damages DNA in human cells. This waste has a long half-life so stays radioactive for a very long time.
Describe the process of nuclear fusion.
The joining of two small nuclei to make a bigger nucleus releasing energy. This process occurs in stars.
give differences between nuclear fusion and fission (process, size of nuclei, where it occurs, what it produces, what it requires)
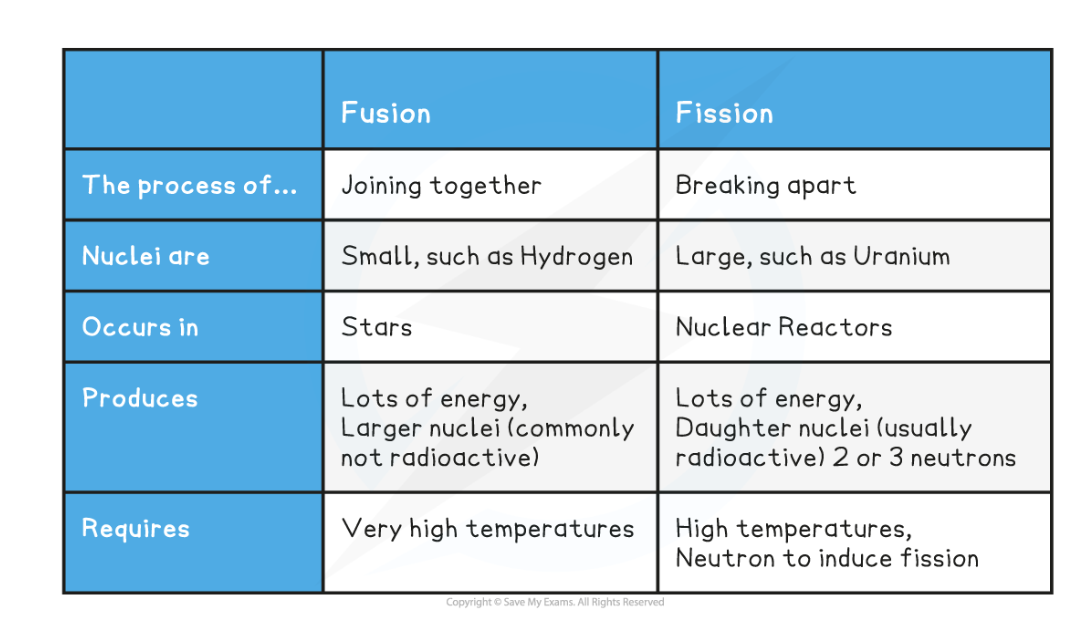
Explain why nuclear fusion does not happen at low temperatures and pressures
The nuclei you are trying to fuse are both positively charged and so repel each other. You need high temperatures so that they are travelling fast enough to overcome this repulsive force. High pressures means the particles are closer together and so increases the chance of fusion.
Give two advantages of nuclear fusion as an energy resource
No carbon dioxide is produced; The fuel is cheap and abundant; No long-lived nuclear waste is produced.