Chemistry Topic 9 - Separate Chemistry II
1/131
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
132 Terms
flame test
A method of identifying a metal cation by the colour it produces in a non-luminous Bunsen flame.
How to carry out a flame test
- Dip clean nichrome wire in concentrated hydrochloric acid to avoid contamination
- Then dipped in solid sample and placed in clean blue flame
Why is Nichrome wire used in flame tests?
Because it's unreactive
Lithium ion colour (flame test)
red
Sodium ion colour (flame test)
yellow
potassium ion colour (flame test)
lilac
Calcium ion colour (flame test)
orange-red
copper ion colour (flame test)
blue-green
copper ion precipitate colour (upon adding NaOH), soluble in excess?
light blue , no
calcium ion precipitate colour (upon adding NaOH), soluble in excess?
white, no
aluminium ion precipitate colour (upon adding NaOH), soluble in excess?
white, yes
Iron(II) ion precipitate colour (upon adding NaOH), soluble in excess?
green, no
Iron(III) ion precipitate colour (upon adding NaOH), soluble in excess?
red brown, no
How to distinguish between calcium and aluminium ions
Start adding a little NaOH to solution then start increasing the NaOH added, if the precipitate dissolves then its aluminium otherwise it's calcium
Test for ammonium ions
add sodium hydroxide, warm gently, damp red litmus paper turns blue and a pungent smell is produced
Ammonium ion symbol
NH4+
NH4+ (aq) + OH- (aq)→
NH3 (g) + H2O (l)
Test for carbonate ions
1.Add dilute acid and test the gas released.
Effervescence should be seen and the gas produced is 2.CO2 which forms a white precipitate of calcium carbonate when bubbled through limewater:
CO3²⁻ (aq) + 2H+ (aq) →
CO2 (g) + H2O (l)
Test for sulfate ion (SO₄²⁻)
Add some dilute hydrochloric acid (to remove any unwanted anions such as carbonate ions) and then add some barium chloride solution. A white precipitate of barium sulfate should form.
Test for halide ions (generic)
Acidify with dilute nitric acid (HNO3) followed by the addition of silver nitrate solution (AgNO3).
Colour will vary depending on the halide
Silver chloride colour
white
Silver bromide colour
cream
Silver iodide colour
yellow
Why do we add nitric acid for halides and hydrochloric acid for sulfates?
To remove carbonate ions
Ba2+ (aq) + SO42- (aq) →
BaSO4 (s)
Advantages of instrumental methods over traditional methods
Greater accuracy
Faster and easier to use
Automated and can perform multiple simultaneous sampling and testing
Modern instruments are very sensitive and can work with multiple sample sizes
functional group
the portion of a molecule that is active in a chemical reaction and that determines the properties of many organic compounds
Displayed formula
A formula showing the relative positioning of all the atoms in a molecule and the bonds between them.
Homologous series
A series of organic compounds with the same functional group but with each successive member differing by CH2. Therefore, they have similar chemical properties and there is a trend in physical properties
molecular formula
a chemical formula of a molecular compound that shows the kinds and numbers of atoms present in a molecule of a compound
Hydrocarbons
Compounds composed of only carbon and hydrogen
Alkanes
a homologous series of saturated hydrocarbons that only contain carbon and hydrogen connected by simple bonds (no double bonds) and end in -ane, general formula: CnH2n+2
Why are alkanes considered to be saturated?
They contain no C=C double bond
methane, ethane, propane, butane (molecular formulae)
CH4, C2H6, C3H8, C4H10
Alkene
A homologous series of unsaturated carbons with a C=C double bond and end in -ene, general formula: CnH2n
What does having a C=C bond mean for alkenes?
It means they can form more bonds with other atoms by releasing their C=C double bond and each carbon atom will form 4 single bonds instead of 2 single and 1 double bond.
Ethene, propene, butene, pentene (molecular formulae)
C2H4, C3H6, C4H8, C5H10
isomers
Compounds with the same formula but different structures.
Example of isomer
but-1-ene and but-2-ene
What does but-1-ene mean?
The C=C bond is between the first 2 carbon atoms
What does but-2-ene mean?
The C=C bond is at the 2nd point between 2 carbons from the left, in other words it is between the second and third carbon.
combustion equation
Hydrocarbon + O2 --> CO2 + H2O
How to distinguish between alkanes and alkenes
Bromine water test: Shake the test tube with bromine water added to both Orange bromine water stays orange with alkanes (C-C single bonds) but becomes clear with alkenes (C=C double bonds). This is because unlike the alkane the alkene reacted with the bromine water to form a colourless product
Addition reactions of alkenes
atoms are added across the C=C double bond so that the double bond becomes a single carbon-carbon bond.
meth/eth/prop/butane + bromine goes to
1,2-dibromometh/eth/prop/butane
polymer
a substance of high average relative molecular mass made up of small repeating units called monomers
What is each monomer in a polymer called?
A repeat unit
How are monomer units connected together?
Through covalent bonds
Examples of polymers
carbohydrates, proteins, and nucleic acids(naturally occurring polymers)
plastics (synthetic polymer)
What is the process called by which monomers join to make a polymer?
Polymerisation
How does addition polymerisation of alkenes work
Double bonds break and join together with the adjacent monomer
Requirements for polymerisation reactions
high pressures
use of a catalyst
How to write out a polymer structure and a repeat unit and a monomer
For polymer:
1.Draw two C atoms that were in a double bond with a single covalent bond
2.Draw brackets and label n
3. Add the links outside the brackets
4.Add the atoms that were attached to each atom of the C bonds
For repeat unit it is the same as a polymer but without brackets or n
Monomer: put the normal molecule with a double bond in brackets preceded by n
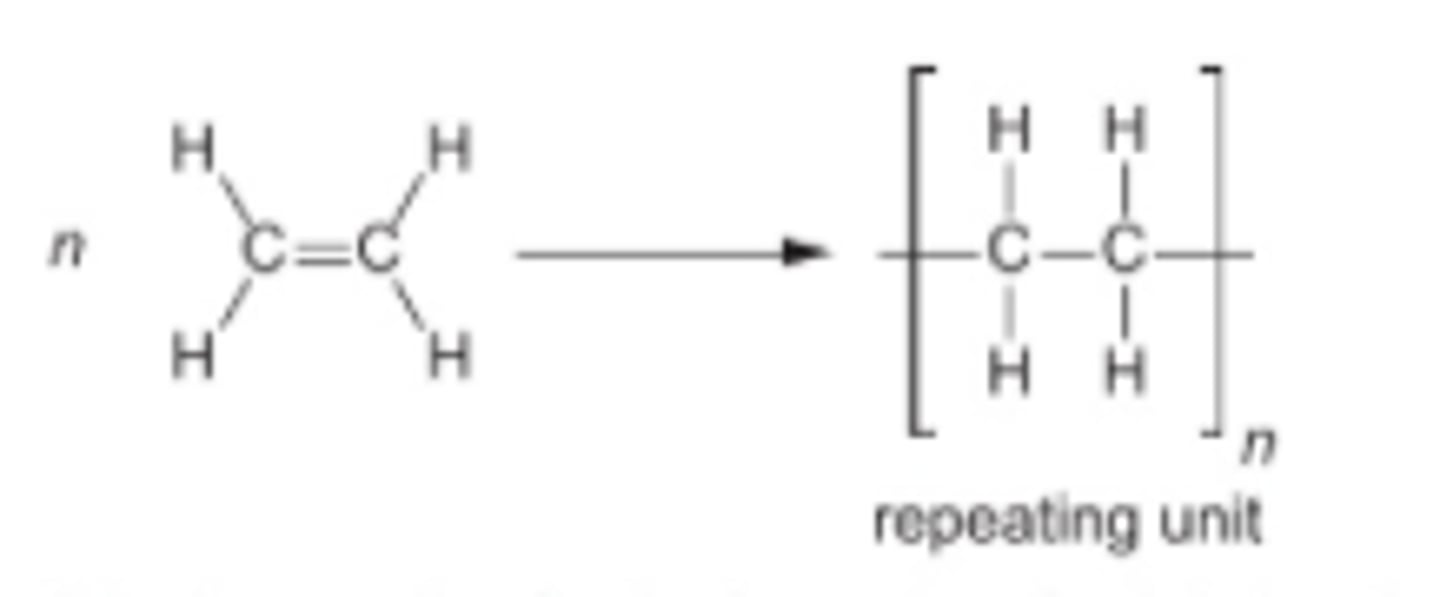
properties of polyethene
Flexible, cheap, good electrical insulator
uses of polyethene
plastic bags, plastic bottles, wire insulation, cling film, polytunnels
properties of polypropene
Flexible, does not shatter/strong
uses of polypropene
buckets, bowls, crates, ropes, carpets
properties of polychloroethene (PVC)
tough, cheap, long lasting, good electrical insulator
Uses of polychloroethene (PVC)
window frames, gutters, water pipes insulation for electrical wires
properties of polytetrafluoroethene(PTFE)
tough and slippery
Uses of polytetrafluoroethene (PTFE)
non-stick coating for frying pans, stain-proofing clothing, carpets
condensation polymers
Polymers formed by the elimination of small molecules(mainly water) when monomers bond together.
How does condensation polymerisation work?
- each molecule has a functional group at each end
- each functional group can react with the functional group of another monomer creating long chains of alternating monomers
- for each new bond that forms, a small molecule (e.g. water) is lost
alcohol + carboxylic acid→
ester + water
how does alcohol and a carboxylic acid release water
The H from the OH and the OH from the O=COH join to make water
ester link
The O=C-O group which is found between monomers in a polymer.
How many water molecules are lost per ester link?
1
Problems with polymers - supply
Made from alkenes which are obtained from crude oil which is a finite resource so this means the price is increasing
Problems with polymers - recycling
Polymers can be recycled but different polymers must be separated from each other
This process is difficult and expensive
Problems with polymers - landfills
Waste polymers are disposed of in landfill sites but this takes up valuable land, as polymers are non-biodegradable so micro-organisms such as decomposers cannot break them down
This causes sites to quickly fill up
Problems with polymers - incineration
Polymers release a lot of heat energy when they burn and produces carbon dioxide which is a greenhouse gas that contributes to climate change
Polymers that contain chlorine such as PVC release toxic hydrogen chloride gas when burned
If incinerated by incomplete combustion, carbon monoxide will be produced which is a toxic gas
Advantage of non biodegradability
Long-lasting
Advantages of recycling polymers
1) Reduces amount of non-biodegradable items in landfills.
2) Reduces emissions of greenhouse gases.
3) Uses up less water/energy.
4) Reduces use of crude oil.
5) Saves money as polymers don't have to be made from scratch
6) Creates jobs
Disadvantages of recycling polymers
1. Sorting plastics by type of polymer is a tedious and labour intensive process which is costly
2. Recycling counts on what is collected in as the raw material, therefore production of certain types of polymers may not be possible due to a lack of starting ingredients
3. Melting polymers produces toxic gases that are harmful to plants and animals
4. Polymers can only be recycled a number of times before they lose their properties and become useless
5. Recycling runs the risk of mixing different polymers together, which again will affect their properties. This is particularly risky for polymers designed for specialist use such as aircraft or automobile parts, where safety is of utmost importance
Alcohol
a homologous series with a functional group of -OH and end in -ol. Alcohols are colourless liquids that dissolve in water to form neutral solutions.
General Formula: CnH2n+1OH
Uses of alcohols
Fuels
Solvents
Alcoholic drinks
methanol, ethanol,propanol, butanol
CH3OH
C2H5OH
C3H7OH
C4H9OH
alcohols experiment
Aim: Investigating which fuel releases the most energy
equipment : set up 100 cm3 of water in a conical flask using a support under a spirit burner for each alcohol. A thermometer should be in the water and everything should be inside a draught screen for insulation
Method: - Record initial temp and mass of water
- Heat each time until temp reaches 40 degrees, measuring time taken for each alcohol
- Then calculate the energy change by multiplying mass in grams x 4.2 x change in temperature
the highest energy change is the most effective fuel
carboxylic acid
a hoomologous series whose fucntional group is carboxyl, COOH.
Their name is alkan-oic acid
methanoic ethanoic propanoic butanoic acids formulae
HCOOH, CH3COOH,C2H5COOH,C3H7COOH
What do -oic acids go to in ion form?
-oate ion
General formula for carboxylic acids
CnH2n+1COOH
Keep in mind that methanoic acid starts at n=0(HCOOH) and n=1 for ethanoic acid and so on
chemical properties of carboxylic acids
- react with bases to form salt and water
- react with metals to form salt and hydrogen
- soluble in water
- react with carbonates to form water co2 and salt
alcohol + oxidising agent (eg acidified potassium dichromate) =
carboxylic acid + water
All members of a homologous series have:
The same general formula
The difference in the molecular formula between one member and the next is CH2
Gradation in their physical properties
Same functional group
Similar chemical properties
fermentation reaction
Glucose (+ yeast enzymes)--> ethanol + carbon dioxide
Conditions for fermentation reaction and why
15 - 35 degrees (not too low for slow RoR and not too high for enzyme denaturation)
in the absence of oxygen (so that yeast can anaerobically respire - if it wasn't anaerobic then only CO2 and H2O would be produced)
What is the concentration of ethanol produced as a result of the fermentation reaction
15%
How to get more concentrated solution of ethanol
Fractional distillation
BP of ethanol(brutal)
78
3 types of particles based off of diameter
coarse particles(aka particulate-matter/dust)
fine particles
nanoparticles
size of nanoparticles
1-100 nm
usually contain only a few hundred atoms
diameter of fine particles
diameters of 100-2500 nm
nanoscience
the research into the production and application of nanoparticles
Is the SA:V ratio of a nanoparticle low or high?
Very high
As particle size decreases what happens to their SA:V ratio?
It increases
fullerenes
nanoparticles made of carbon
What is the main industrial application of nanoparticles and why
Inn catalysis
This is due to their high SA:V ratios and the higher the ratio then the more SA is available for reaction hence the better the catalyst
What is the nanoparticle form of titanium dioxide used in and why?
Sunscreens
This is because it blocks UV light but leaves no white marks on the skin