2.1 Metals and Reactivity Series 🫧
1/37
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
Reactivity Series
Potassium
Sodium
Calcium
Magnesium
Aluminium
Zinc
Iron
Copper

More reactive metals
react more vigorously and lose electrons easier (positive ion)
Metal + Oxygen →
Metal Oxide
Potassium and oxygen (Observations)
lilac flame
white solid formed
Sodium and oxygen (Observations)
yellow/orange flame
white solid formed
Calcium and oxygen (Observations)
brick red flame
white solid
Magnesium and oxygen (Observations)
white light observed
white solid formed
Aluminium and oxygen (Observations)
as a powder
bright white light
white solid formed
Zinc and oxygen (Observations)
glows orange
yellow solid, turning white when cooled
Iron and oxygen (Observations)
as filings
orange sparks
black solid
Copper and oxygen (Observations)
glows orange
green/blue flame
black solid
Cutting potassium and sodium
soft metals expose shiny surface which tarnishes dull
Tarnishing of reactive metals
less reactive metals take longer to tarnish
Metal + water →
metal hydroxide + hydrogen
Potassium and water (Observations)
Floats
Moves very rapidly across the surface
Bubbles of gas released
Heat is released
Ignites with a lilac flame
Crackles at the end/explosion
Colourless solution formed
Sodium and water (Observations)
Floats
Moves rapidly across the surface
Bubbles of gas released
Melts to form a sphere of molten metal
Colourless solution forms
Calcium and water (Observations)
Granules sink and rise continuously
Bubbles of gas released
Heat is released
Grey powdered solid forms in the water
Magnesium and water (Observations)
Very slow (days)
Few bubbles of gas produced
Metals that don’t react with water
aluminium, zinc, iron and copper
Why does aluminium not react
surface forms protective layer of aluminium oxide
Metal + steam →
metal oxide + hydrogen
Magnesium and steam (Observations)
bright white light
white powder
Aluminium and steam (Observations)
glows orange
white powder
Zinc and steam (Observations)
glows orange
yellow solid, turning white when cooled
Iron and steam (Observations)
glows red
black solid
Why do some metals react with steam
increased temperature of reaction
Displacement
more reactive metal takes the place of a less reactive metal in a compound
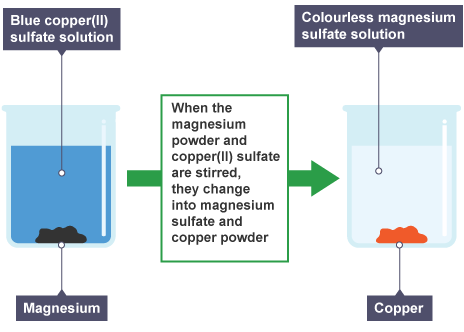
Finding less reactive elements
usually found in earths cruse pure
Finding more reactive metals
usually combined with other elements in a compound
Metals extracted using electrolysis
potassium, sodium, calcium, magnesium and aluminium
Why is electrolysis only used for most reactive metals
expensive, cheaper to heat with carbon/carbon monoxide
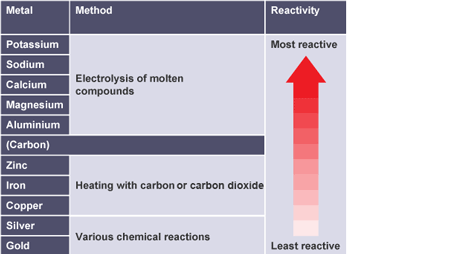
Metals extracted using reduction with carbon/carbon monoxide
zinc, iron and copper
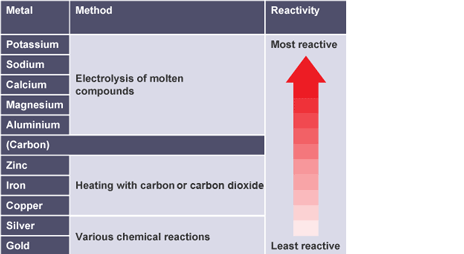
Metals extracted using various chemical reactions
silver and gold
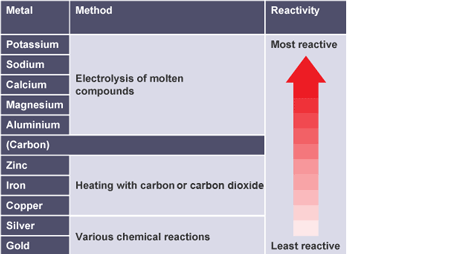
Phytomining
plants used to absorb metal ions from ground using roots
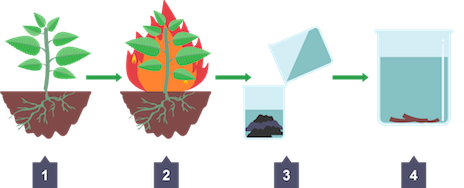
Process of phytomining
Plants used to absorb metal compounds e.g copper(II) compounds
Harvested, burned to produce ash containing metal compounds
Sulfuric acid added to produce solution containing dissolved metal compounds called leachate
Copper obtained by displacement using scrap iron
Disadvantages of phytomining
slow
more expensive than mining
growing conditions e.g weather, altitude and soil quality
Advantages of phytomining
reduces need to obtain new ore by mining
metal ores are limited, alternative
energy produced, can be used
plants remove co2
no disposal of waste rock
Why is mining bad
negative environmental impact through noise and dust pollution