AS level chemistry organic
1/153
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
154 Terms
Only type of amine group we need to learn for AS level
primary amine
Describe primary amine synthesis

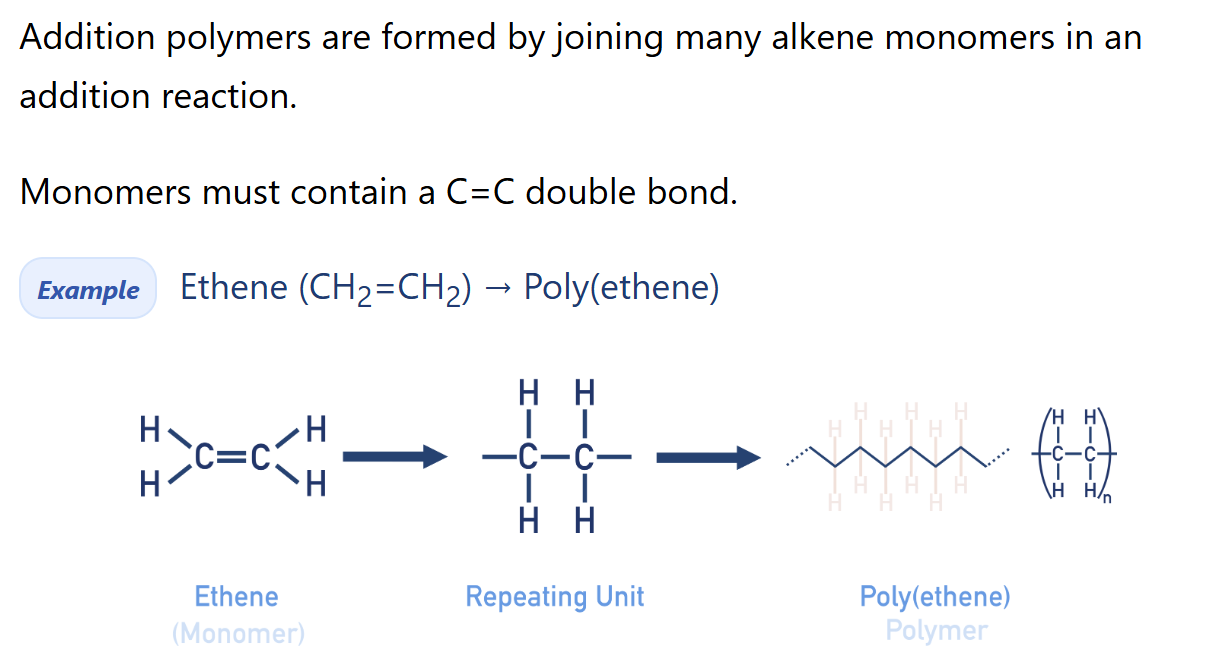
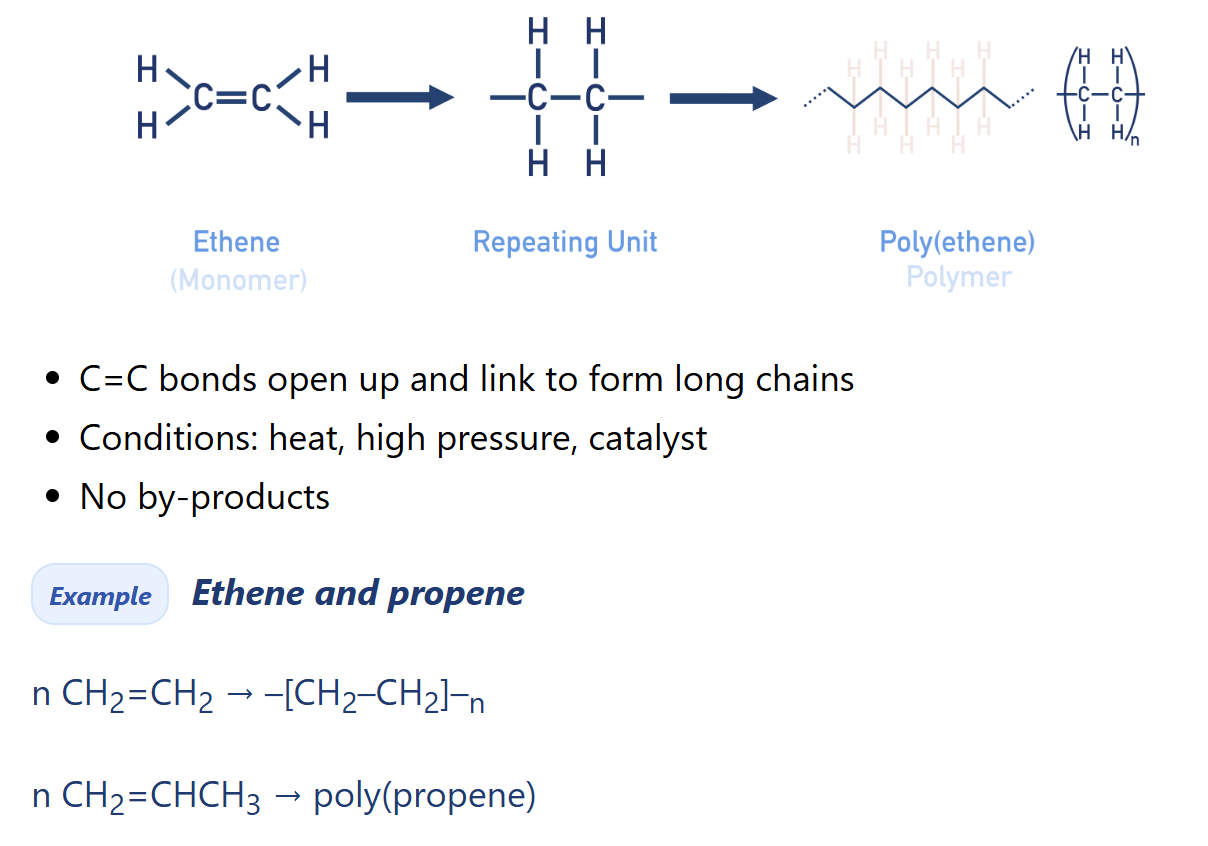
Describe addition polymerisation
Addition polymers are (very) large molecules made up of repeating units, bonded together over and over again. Repeating units are formed from small molecules called monomers.
Properties of addition polymer
Addition polymers are chemically inert because strong C–C and C–H bonds make addition polymers very unreactive.
They are also Non-biodegradable: These materials are not broken down naturally, causing long-term environmental issues. The strength and stability of the carbon chain makes these materials resistant to biological and chemical degradation.
Environmental issues of addition polymer
Disposal: Polyalkenes accumulate in landfill due to their resistance to decay (non-biodegradable).
Combustion products:
Burning poly(chloroethene) (PVC) releases toxic hydrogen chloride (HCl) gas.
Incineration can contribute to air pollution and acid rain if not properly managed.
As a result, care must be taken when disposing of polymers, particularly halogenated ones like PVC, due to the risk of toxic emissions.
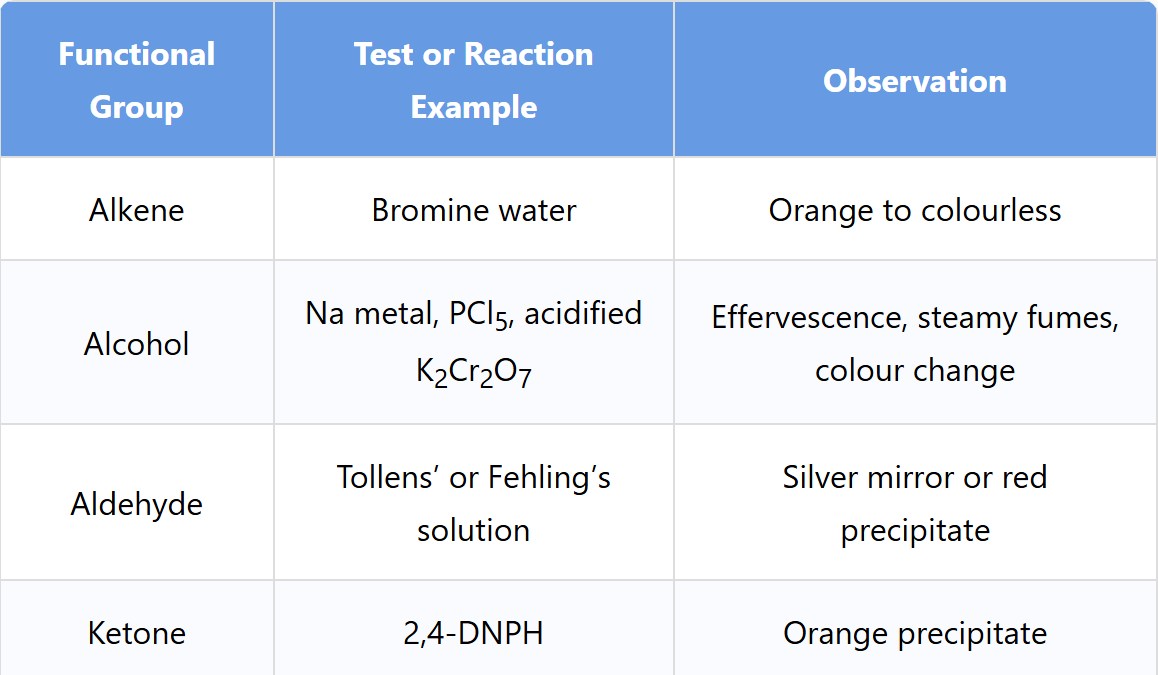
List back and describe all the tests for Alkene, alcohol, aldehyde, and ketone
List all mechanism in AS level chemistry
Free-radical substitution
Electrophilic addition
Nucleophilic addition
Nucleophilic substitution
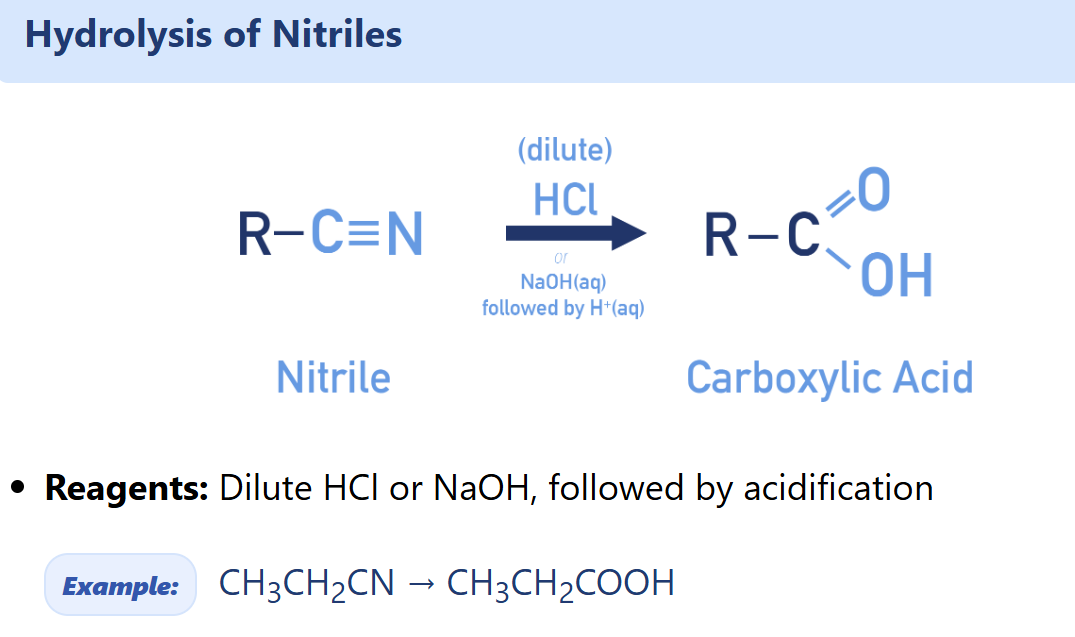
Describe synthesis of carboxylic acid (using nitriles) and there’re 2 alternatives
Dilute acid —→ acidification
Dilute alkali —→ acidification
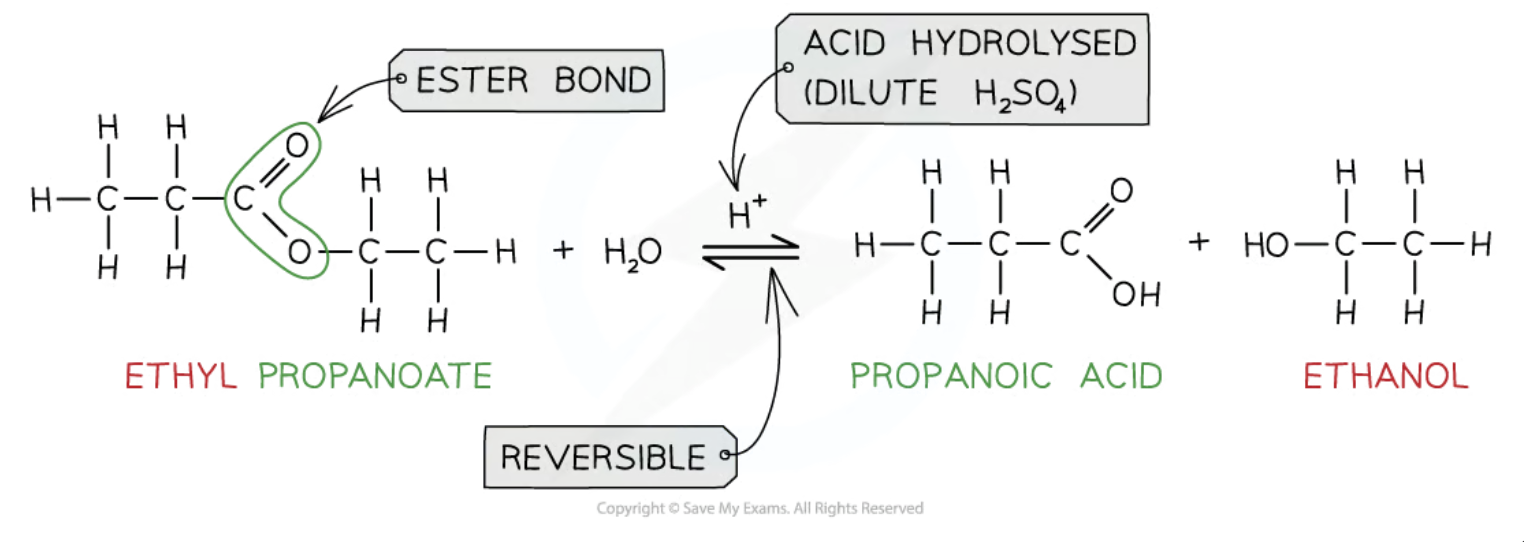
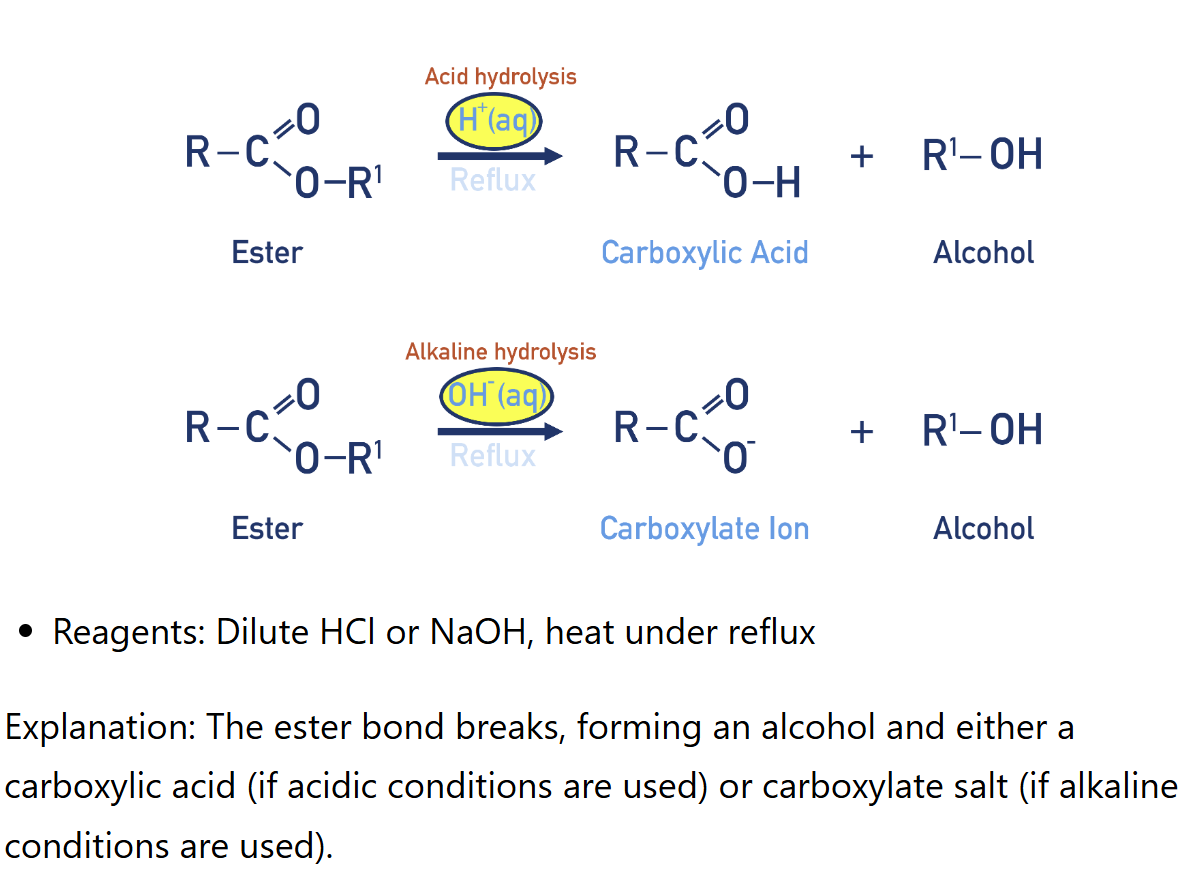
Acid Hydrolysis of ester (carboxylic acid synthesis)
Reagents: dilute H2SO4 or HCl
Conditions: heat under reflux
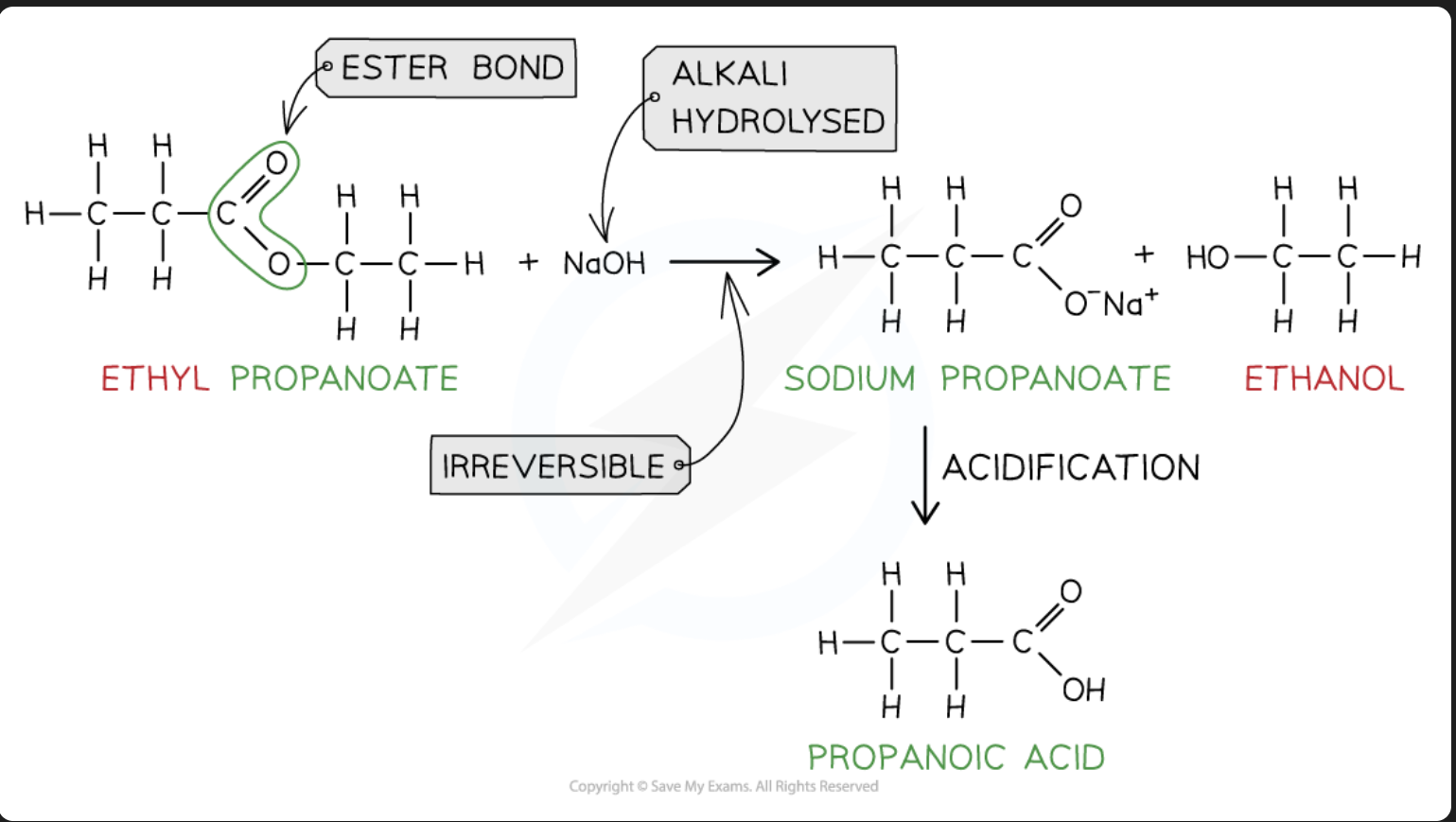
Alkali Hydrolysis of ester (carboxylic acid synthesis)
Reagents: Dilute NaOH
Conditions: heat under reflux, follow by acidification
Describe carboxylic acid Redox Reaction with Reactive Metals (carboxylate salt synthesis)
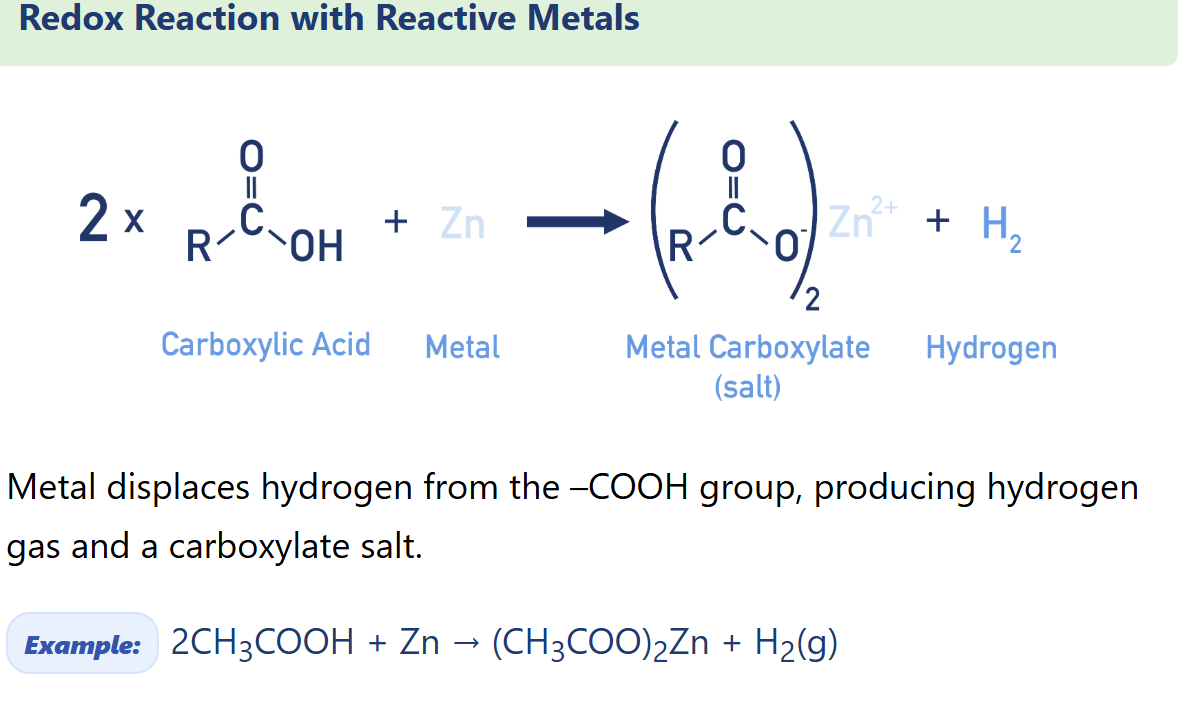
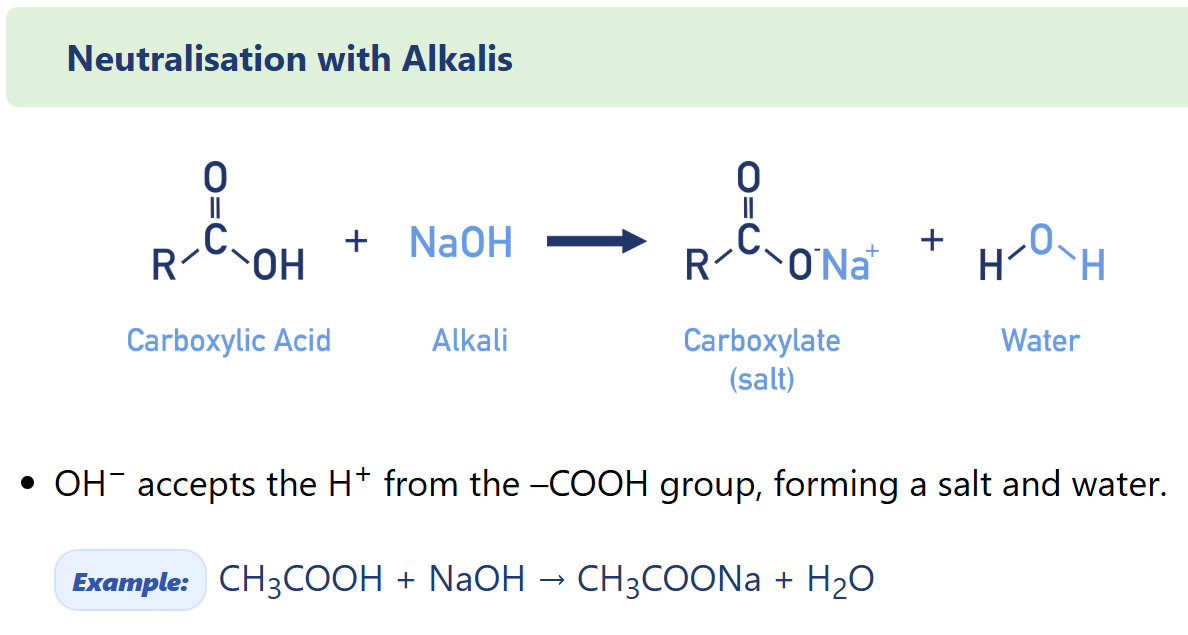
Describe carboxylic acid Neutralisation with Alkalis
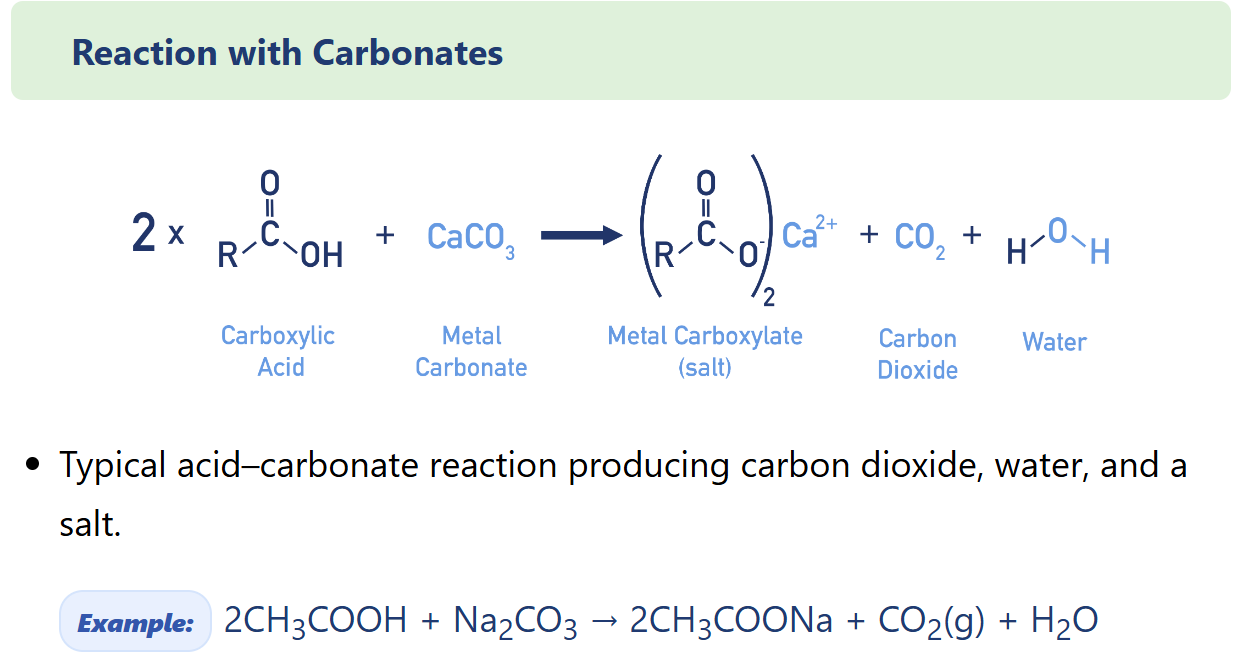
Describe carboxylic acid Reaction with Carbonates
Esterification of carboxylic acid (Ester synthesis)

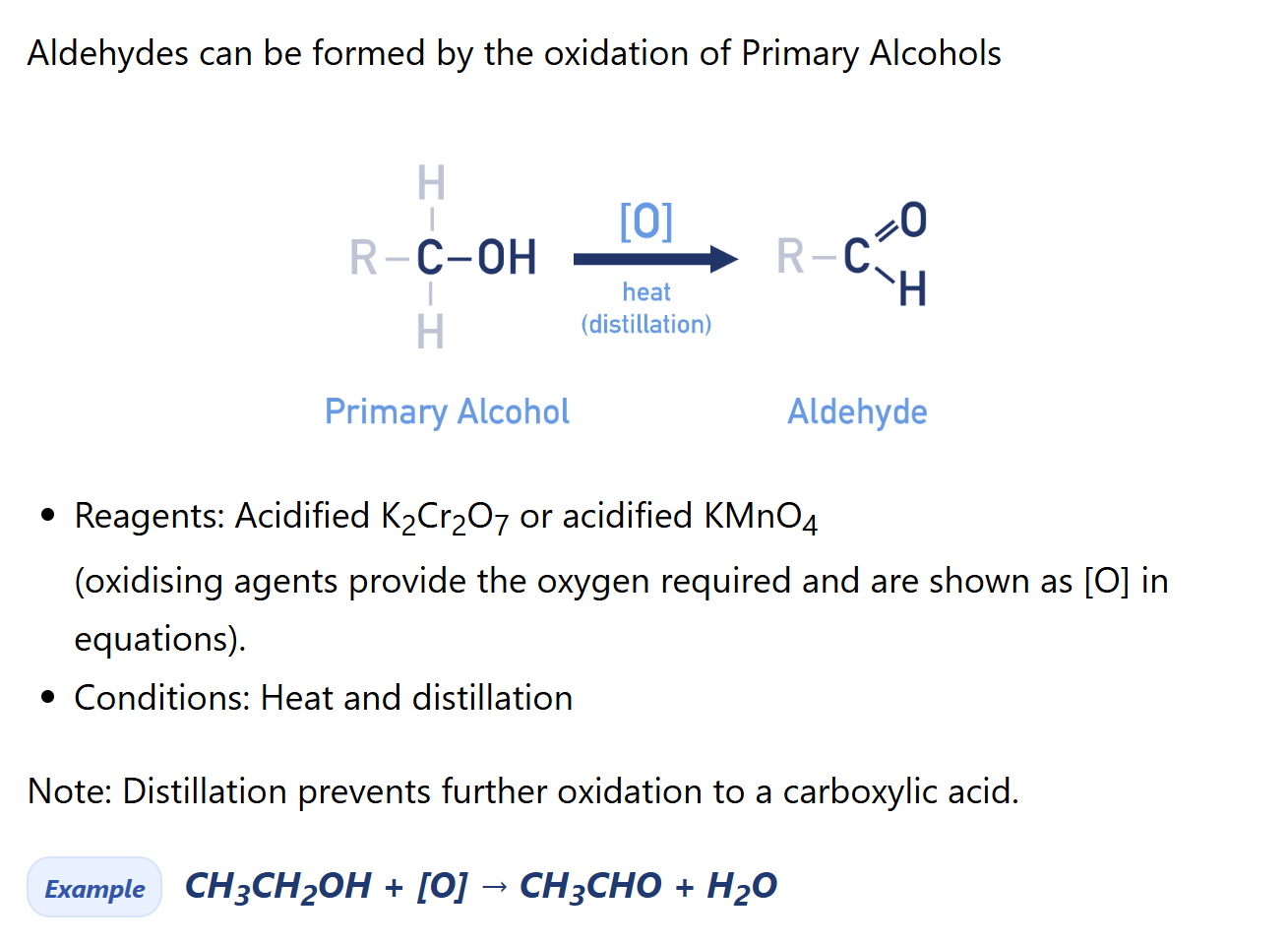
Describe synthesis reaction of aldehyde using primary alcohol
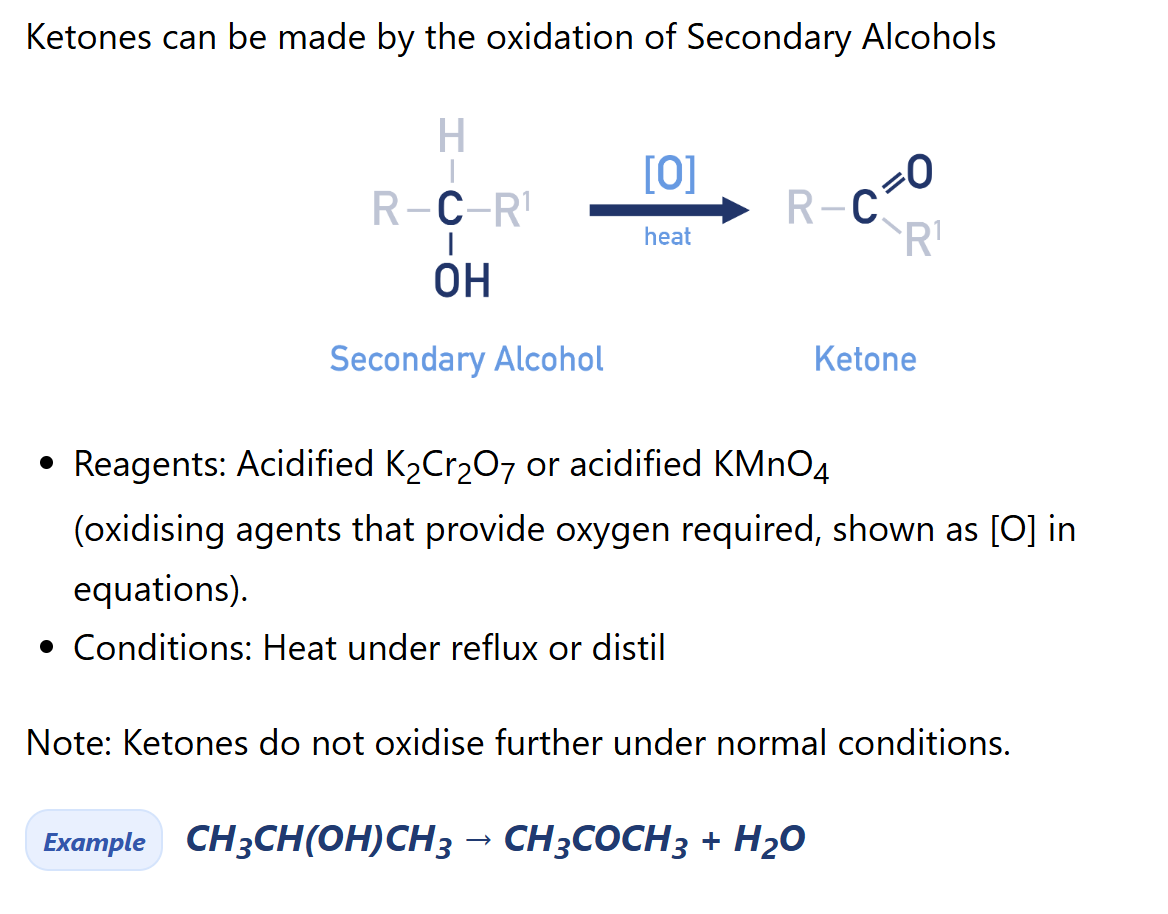
Describe synthesis reaction of ketone using secondary alcohol
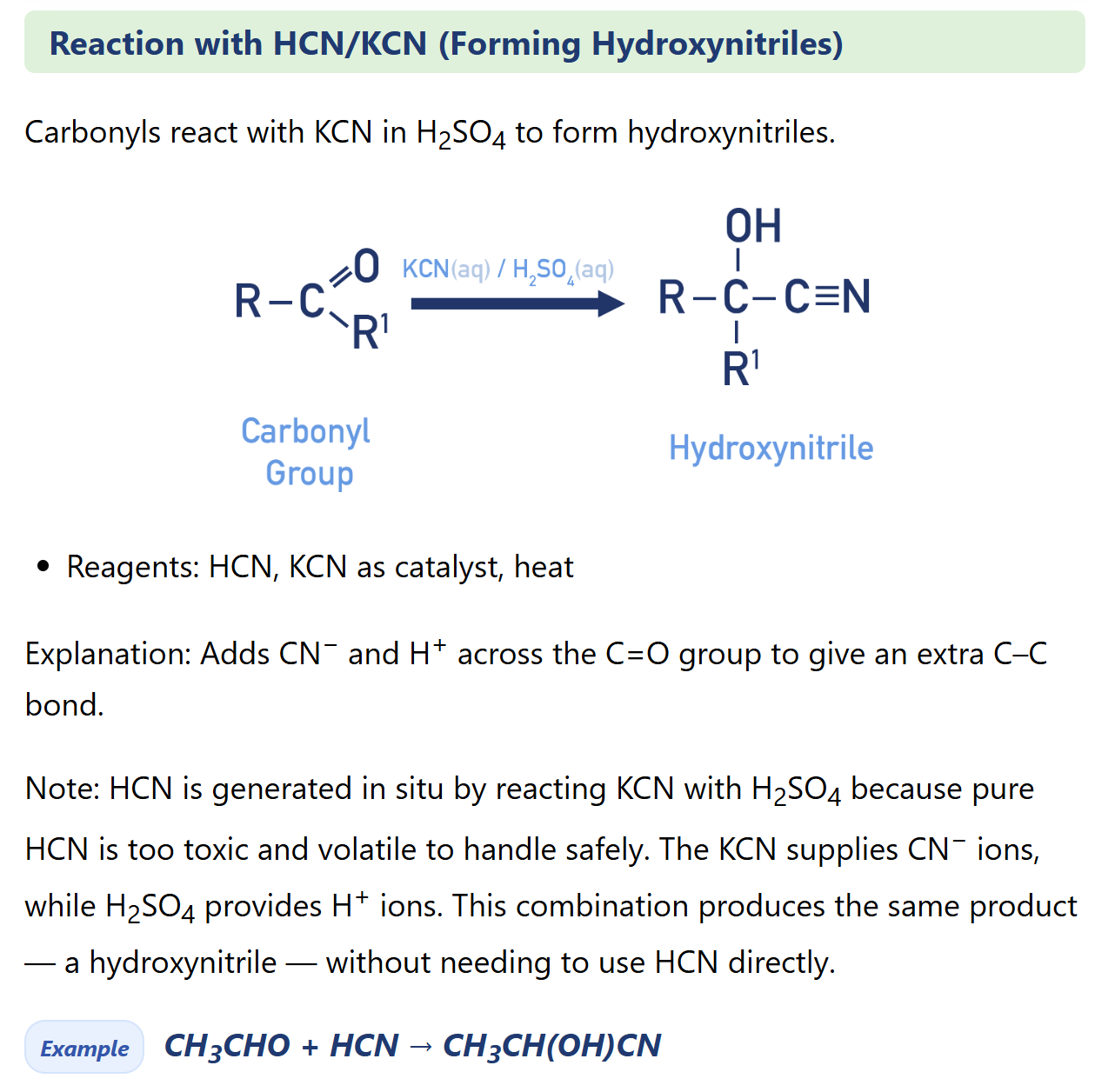
Describe reaction of aldehyde/ketone to form hydroxynitrile
describe nucleophilic addition mechanism (of HCN in forming hydroxynitriles from carbonyl group)
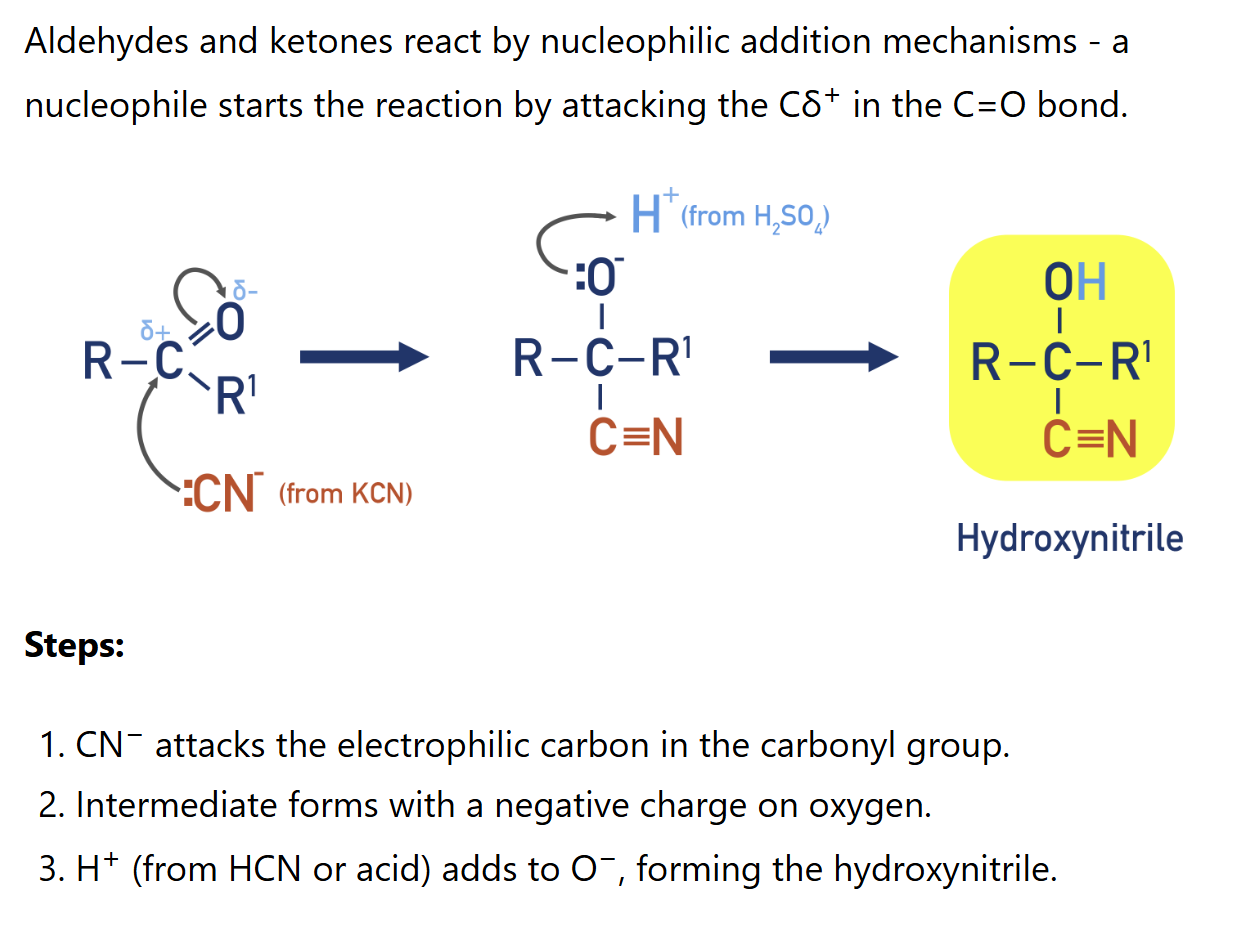
Describe test with 2,4-DNPH
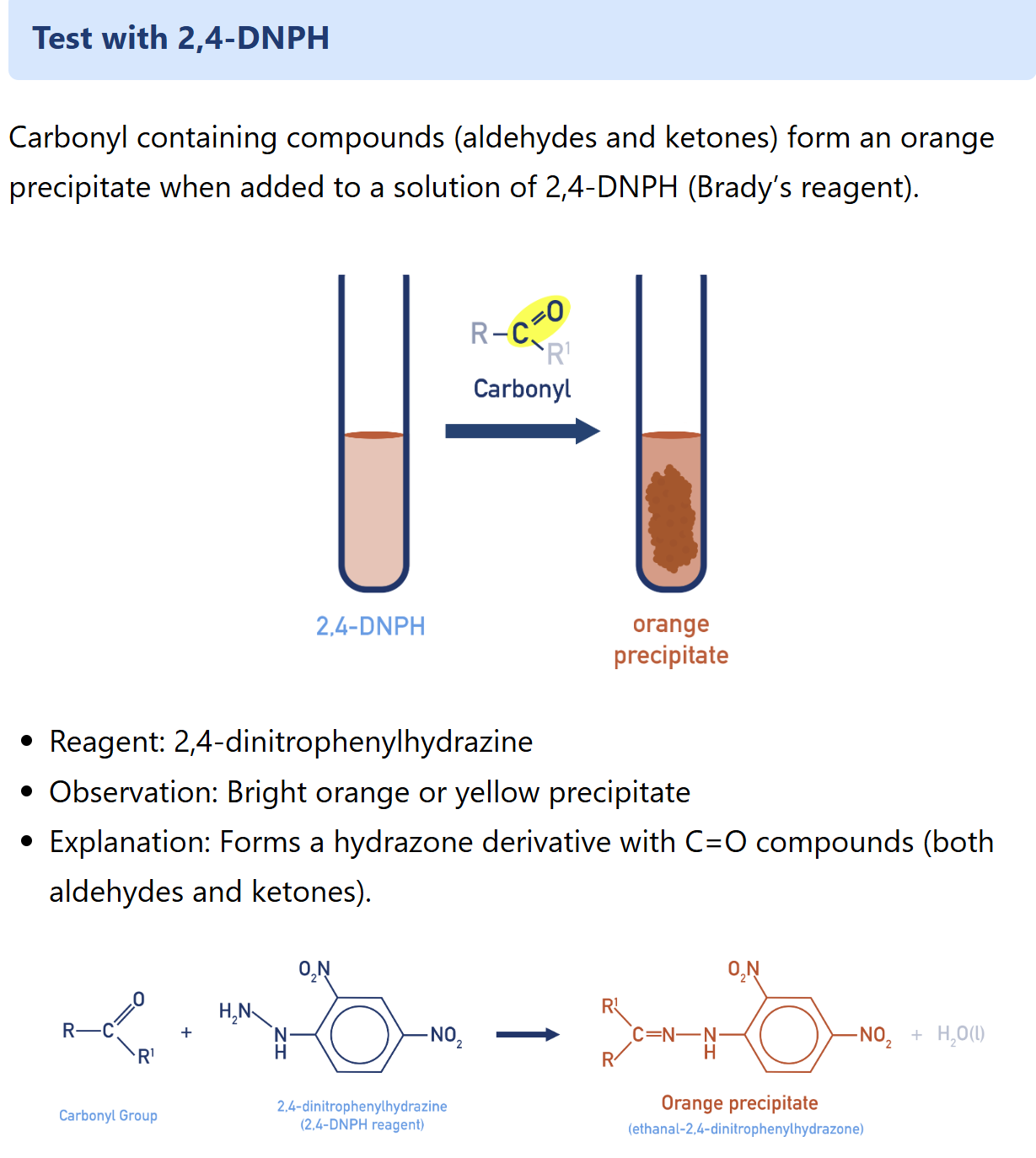
Describe Fehling’s test
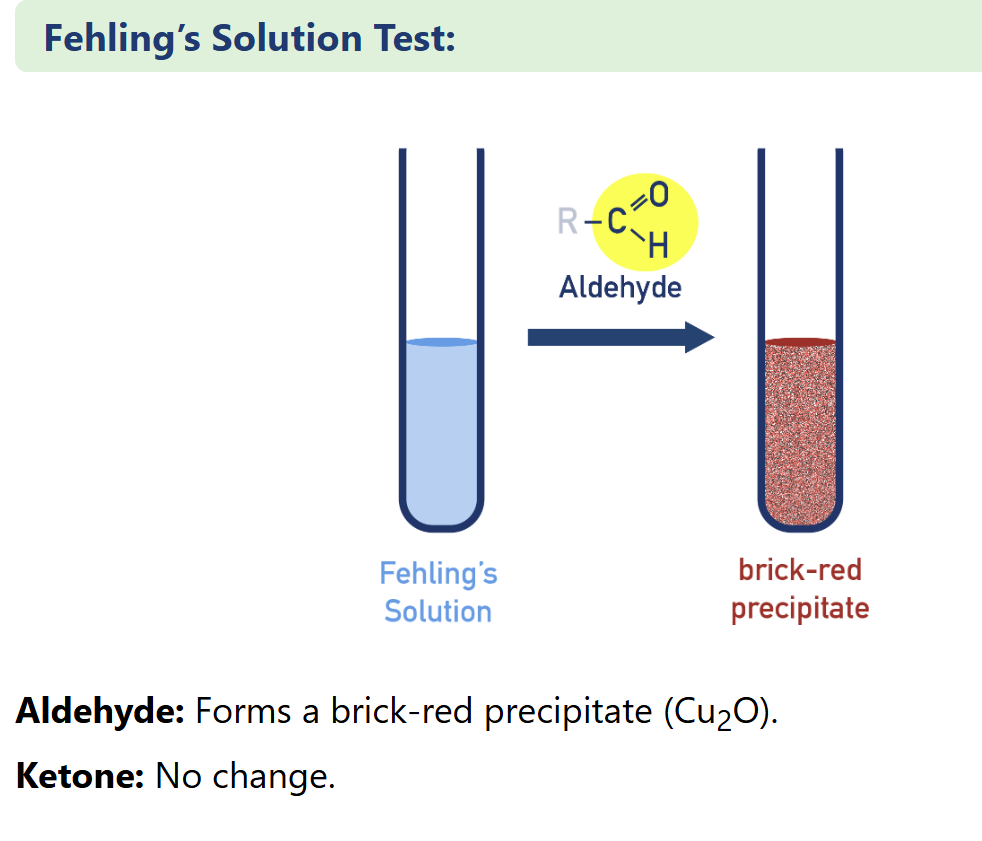
describe Tollens’ Silver Mirror Test
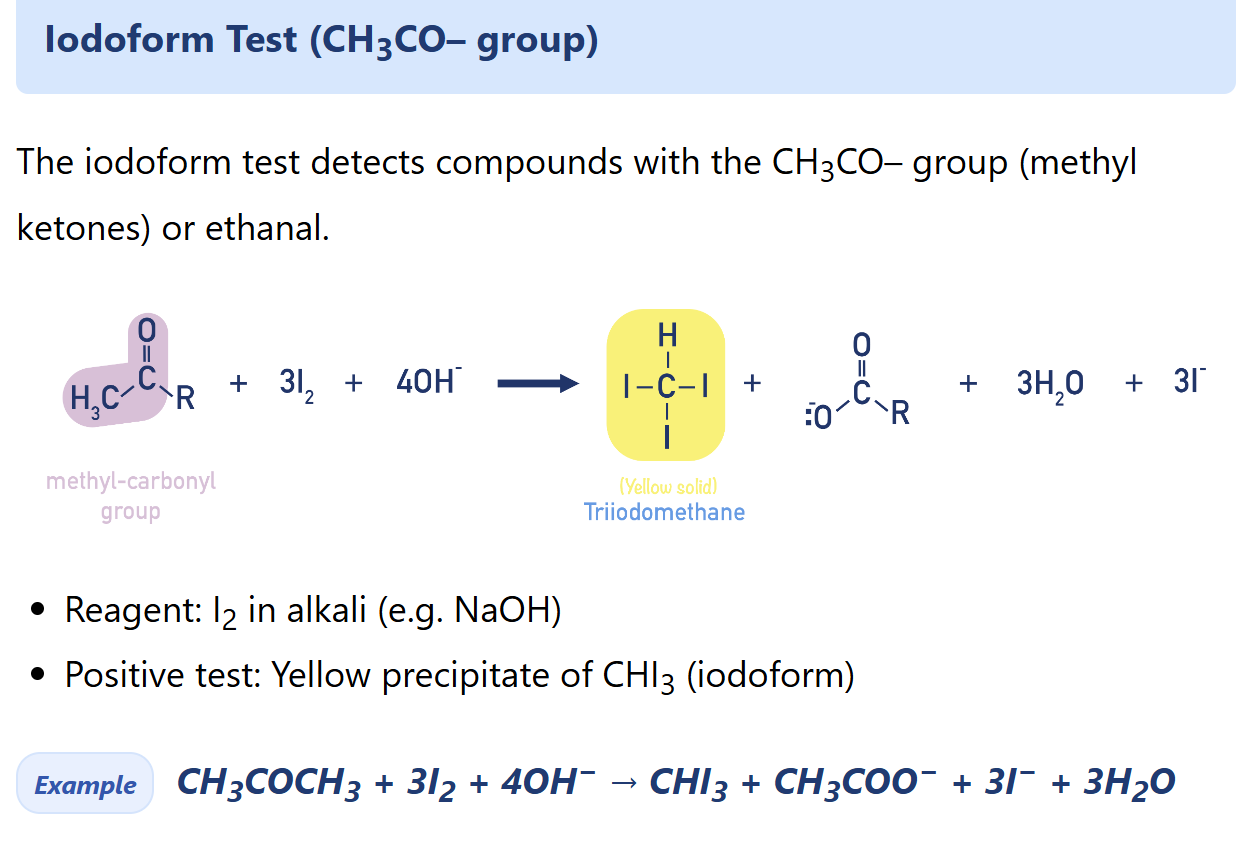
Describe iodoform test
Alcohol
organic compounds that contain a carbon atom bonded to a hydroxy, OH group
Reduction of aldehydes/ ketones
(alcohol synthesis)
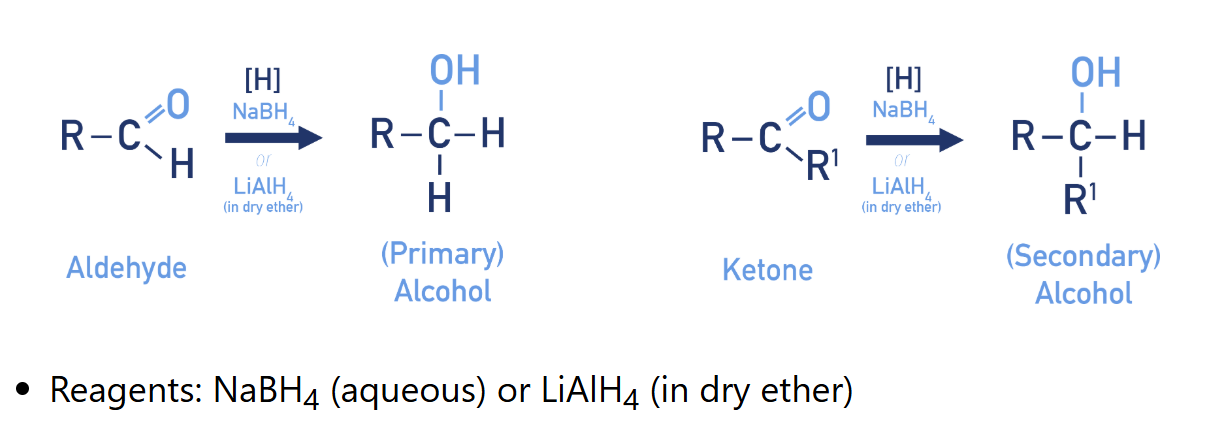
Why reduction using LiAlH4 need to be in dry ether?
LiAlH4 is a more powerful reducing agent than NaBH4, because of this if LiAlH4 is used no water can be present and the reaction must be carried out in dry ether.
What NaBH4 and LiAlH4 stand for?
NaBH4 : sodium borohydride
LiAlH4 : Lithium aluminum hydride
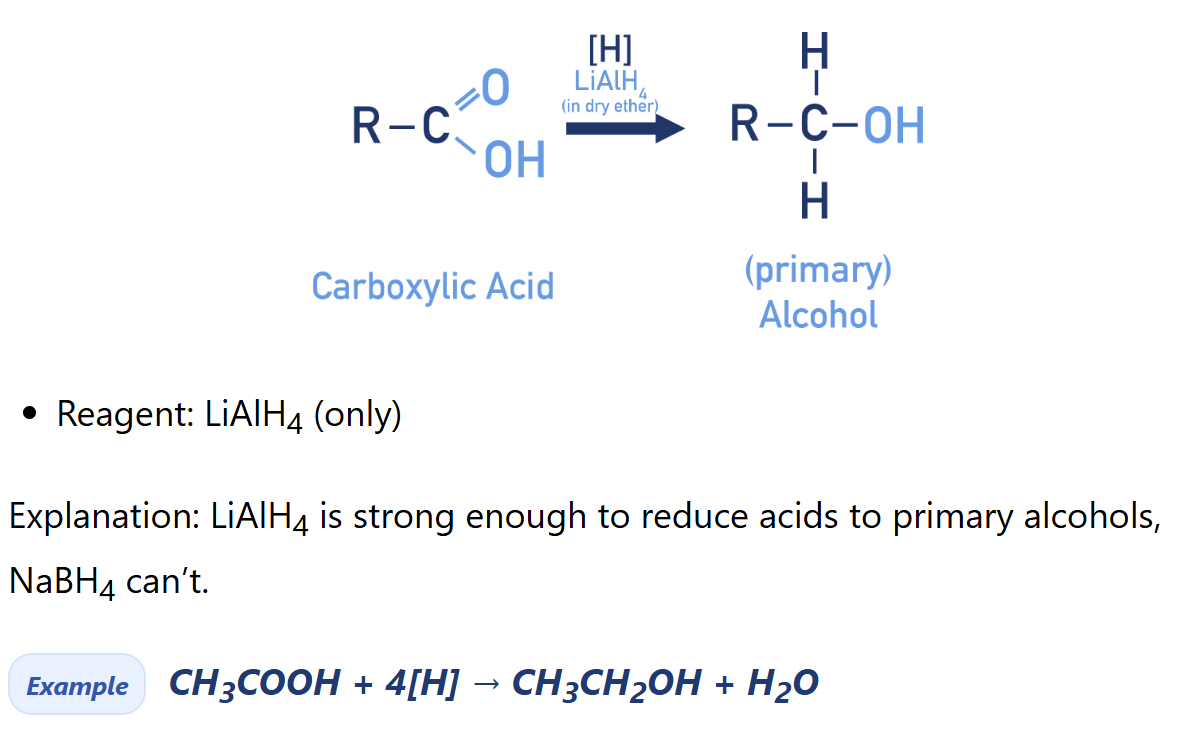
Reduction of Carboxylic Acids
(alcohol synthesis)
Hydrolysis of Esters (there are two alternative by-products)
(Alcohol synthesis)
Alcohol combustion
Alcohols (especially shorter chain alcohols) burn cleanly to form CO2 and water making them useful as fuels.
Example CH3CH2OH + 3O2 → 2CO2 + 3H2O
Alcohol reacts with sodium (Alkoxide synthesis)
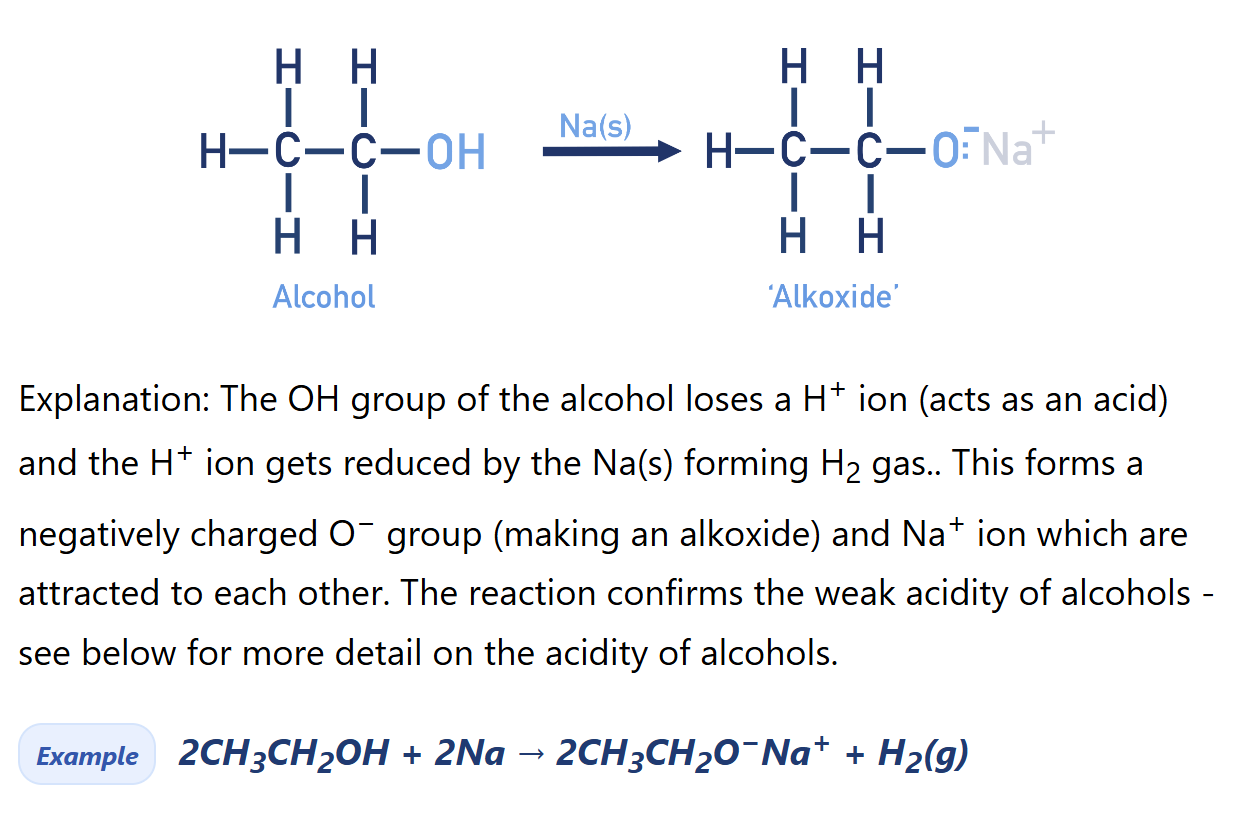
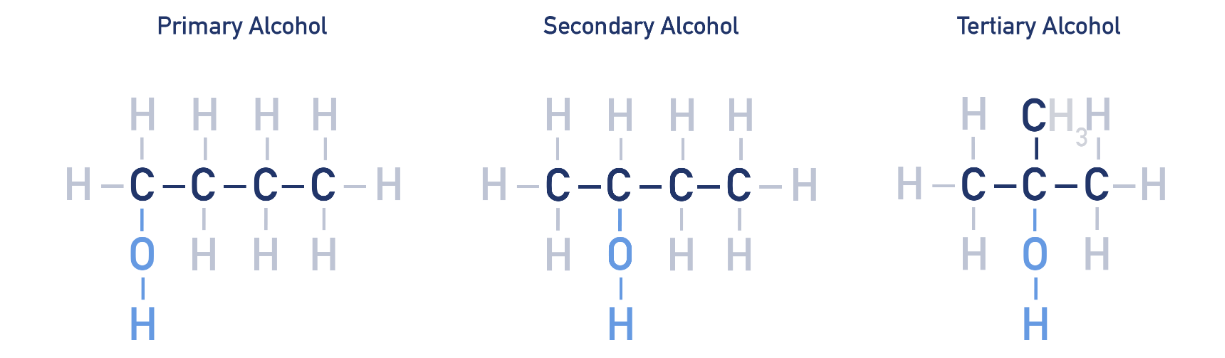
Different types of alcohol
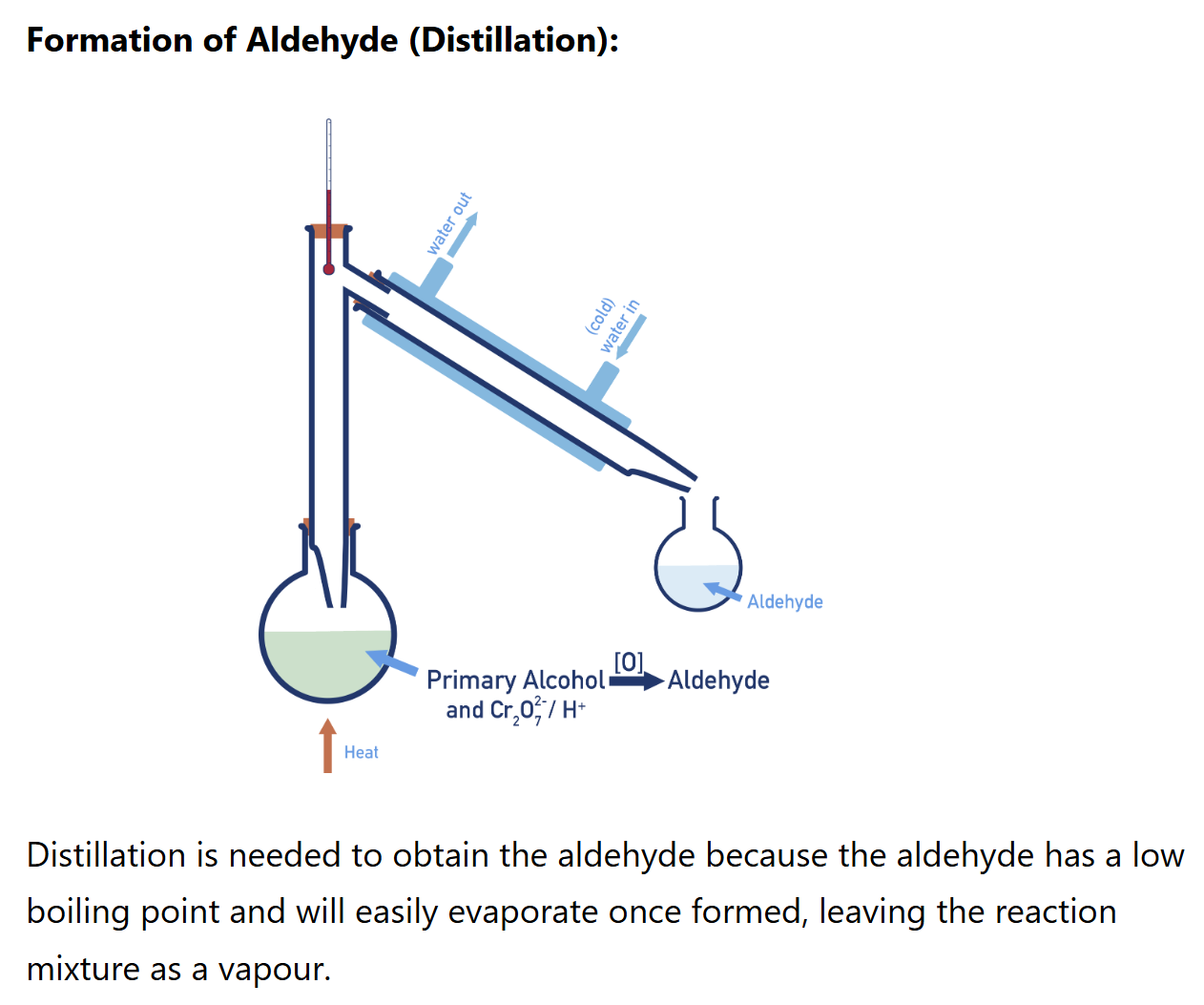
Primary alcohol oxidation (aldehyde synthesis)
Primary alcohols oxidise first to aldehydes, then to carboxylic acids
reagent: K2Cr2O7/ H+
condition: distillation + heat
Observation when alcohol oxidized to form aldehyde / carboxylic acid
Observation: Orange Cr2O72− (dichromate) turns green (Cr3+ formed)
Route for oxidation of primary alcohol
Alcohol ——[O] —→ Aldehyde ——-[O]——> Carboxylic acid
![<p>Alcohol ——[O] —→ Aldehyde ——-[O]——> Carboxylic acid</p>](https://assets.knowt.com/user-attachments/e222e3da-dbe7-4dac-a8f9-146e3ac743f3.png)
Why need distillation during the oxidation of alcohol to form aldehyde
to remove aldehyde before it can further oxidised into carboxylic acid (this will create a mixture)
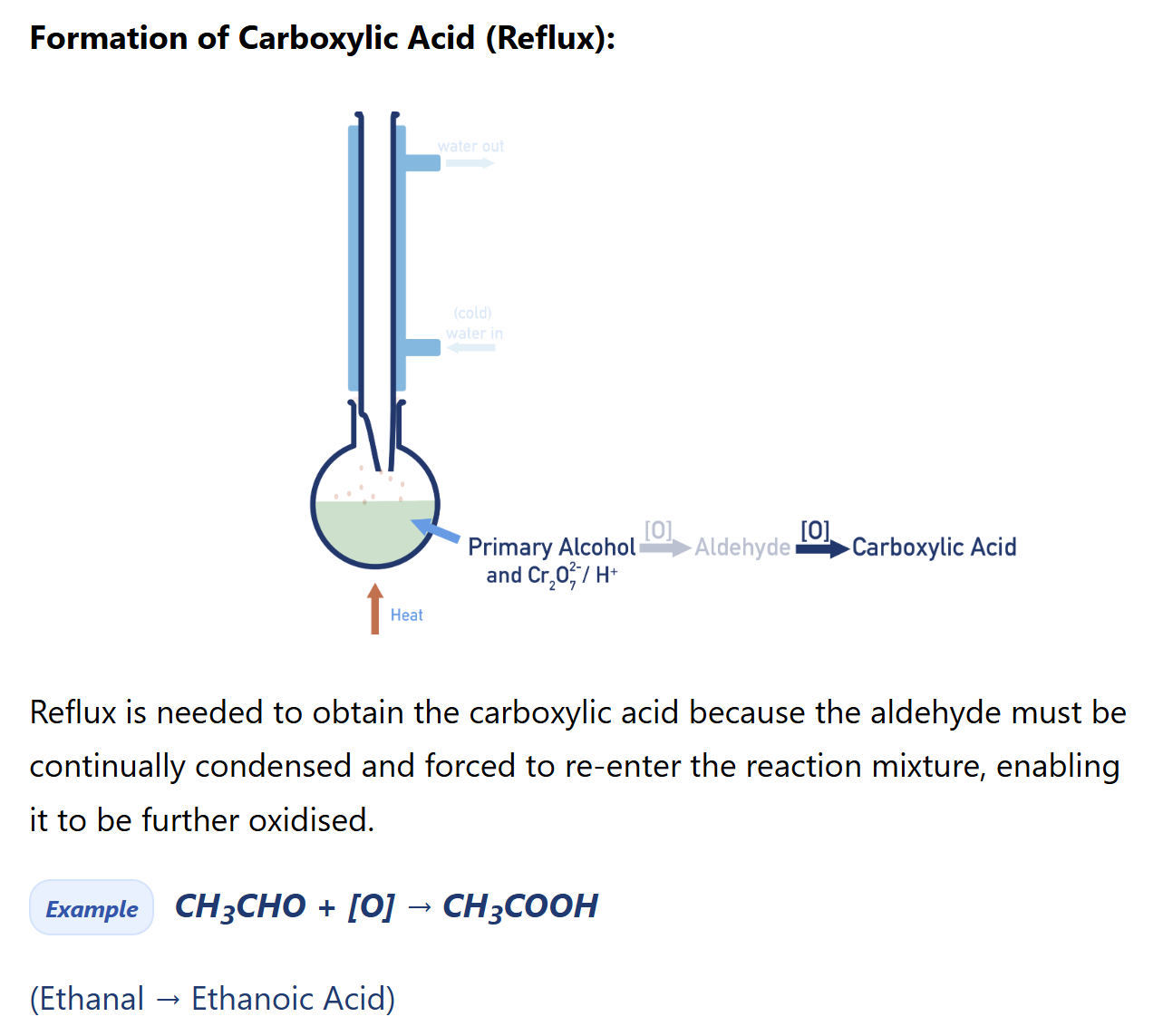
Primary alcohol oxidation (carboxylic acid synthesis)
reagent: K2Cr2O7/ H+ OR (KMnO4/H+ , quite strong=> result to carboxylic acid directly)
condition: heat in reflux
Oxidation for secondary alcohol (ketone synthesis)
reagent: K2Cr2O7/ H+
condition: heat under reflux
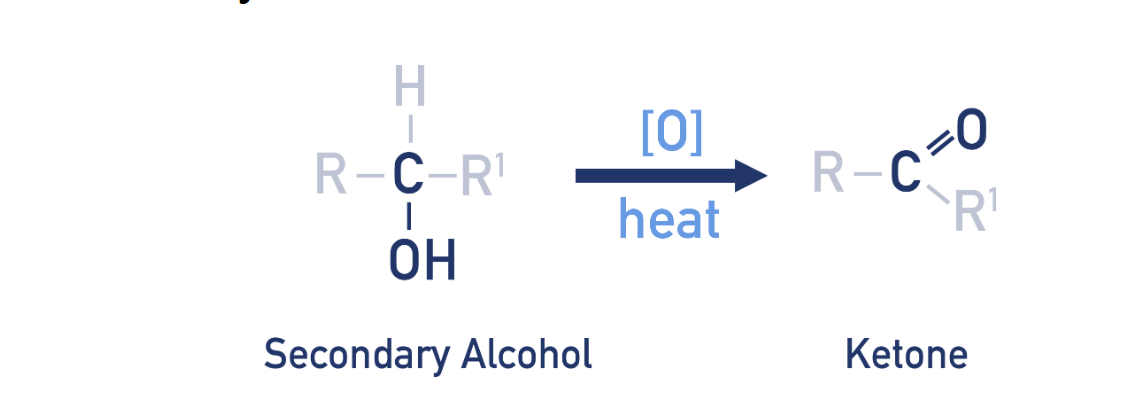
Observation for secondary alcohol oxidation (ketone synthesis)
Orange Cr2O72− (dichromate) turns green (Cr3+ formed).
Why ketone/ tertiary alcohol can’t oxidise further
Because there are no more hydrogen bonded to the carbon that bonded with the oxygen
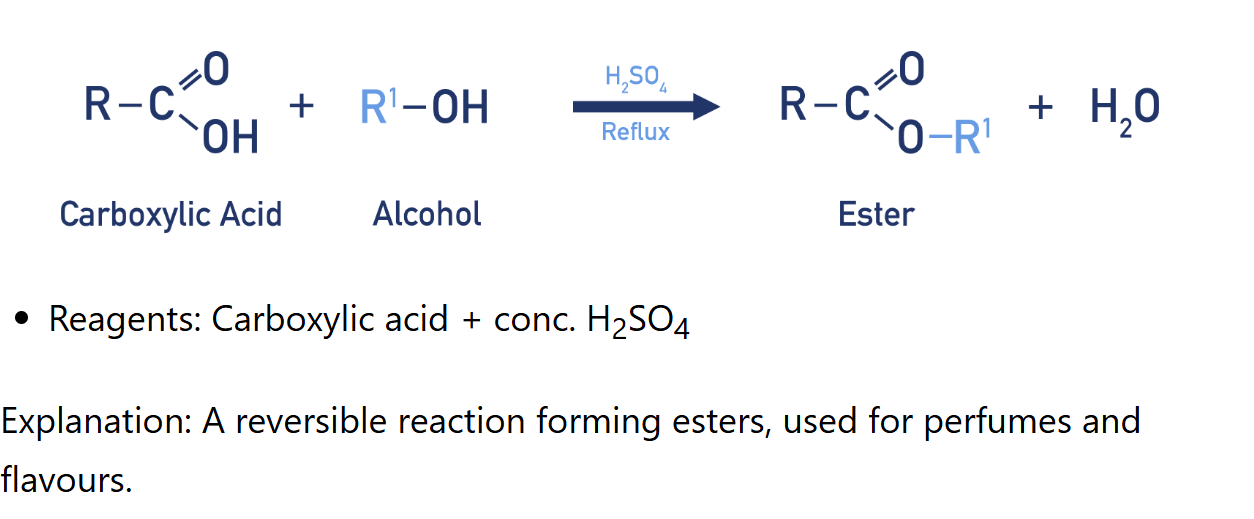
Esterification of alcohol
Reagents: conc.H2SO4 , carboxylic acid
Condition: heat under reflux
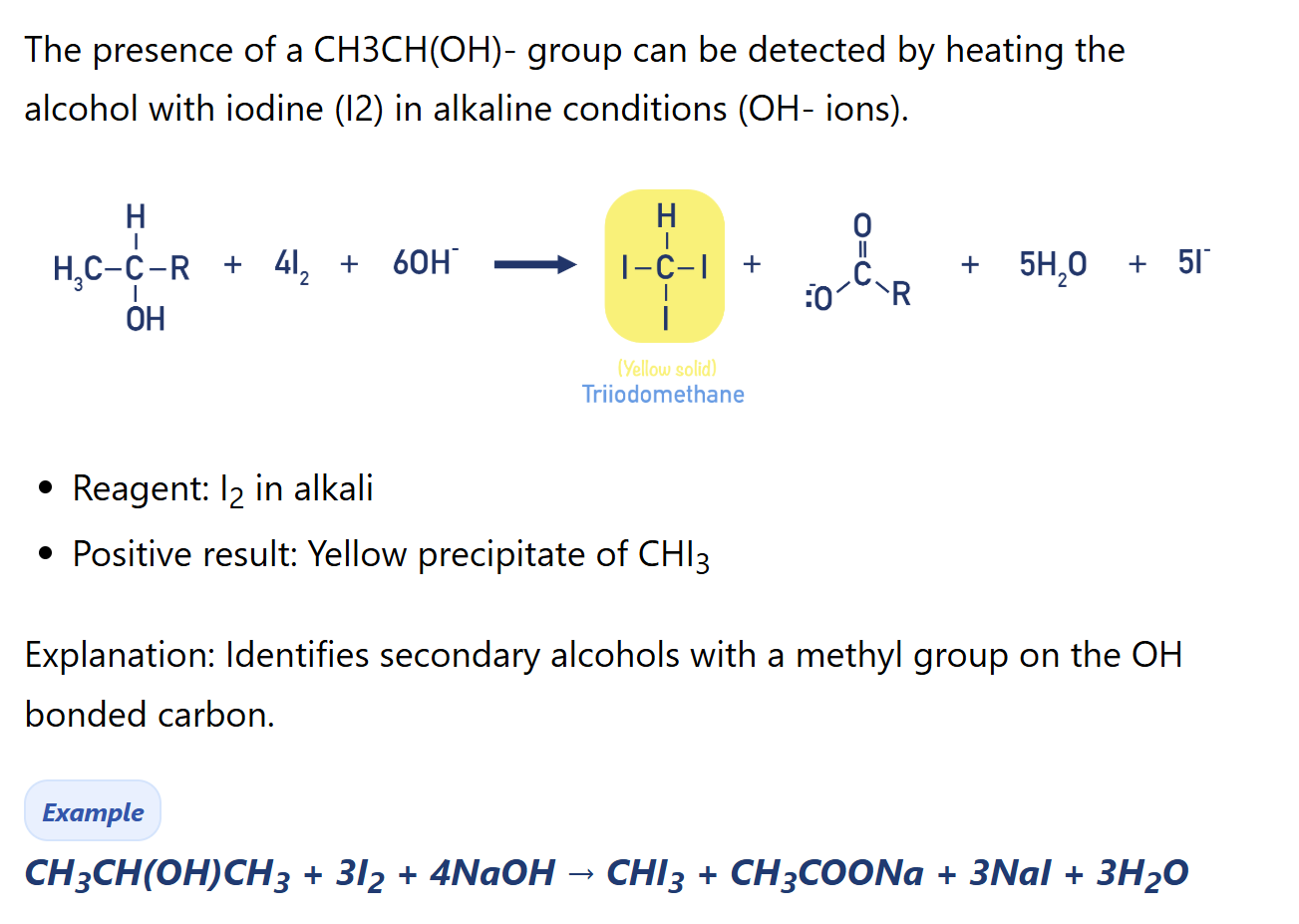
What iodoform test used for?
To test the presence of CH3CH(OH)–R Group
How iodoform test find the presence of CH3CH(OH)- group
Products: tri-iodomethane, RCO2- (by-products: iodide, water)
Compare acidity of alcohol with water
Alcohols are much weaker acids than water. They are less able to lose H+ ions.
This is because the alkoxide ion (RO−) formed after losing a H+ ion is less stable than the hydroxide ion (OH−) formed from water. This is due to the electron-donating nature of the alkyl group, which increases the electron density on the negatively charged oxygen, making it less stable and less likely to form. In contrast, the hydroxide ion is more stable, so water more readily donates a proton, making it a stronger acid than alcohols.
Halogenoalkane
molecules contain a halogen (F, Cl, Br, I) bonded to a carbon
Alcohols nucleophilic substitution (halogenoalkane synthesis)
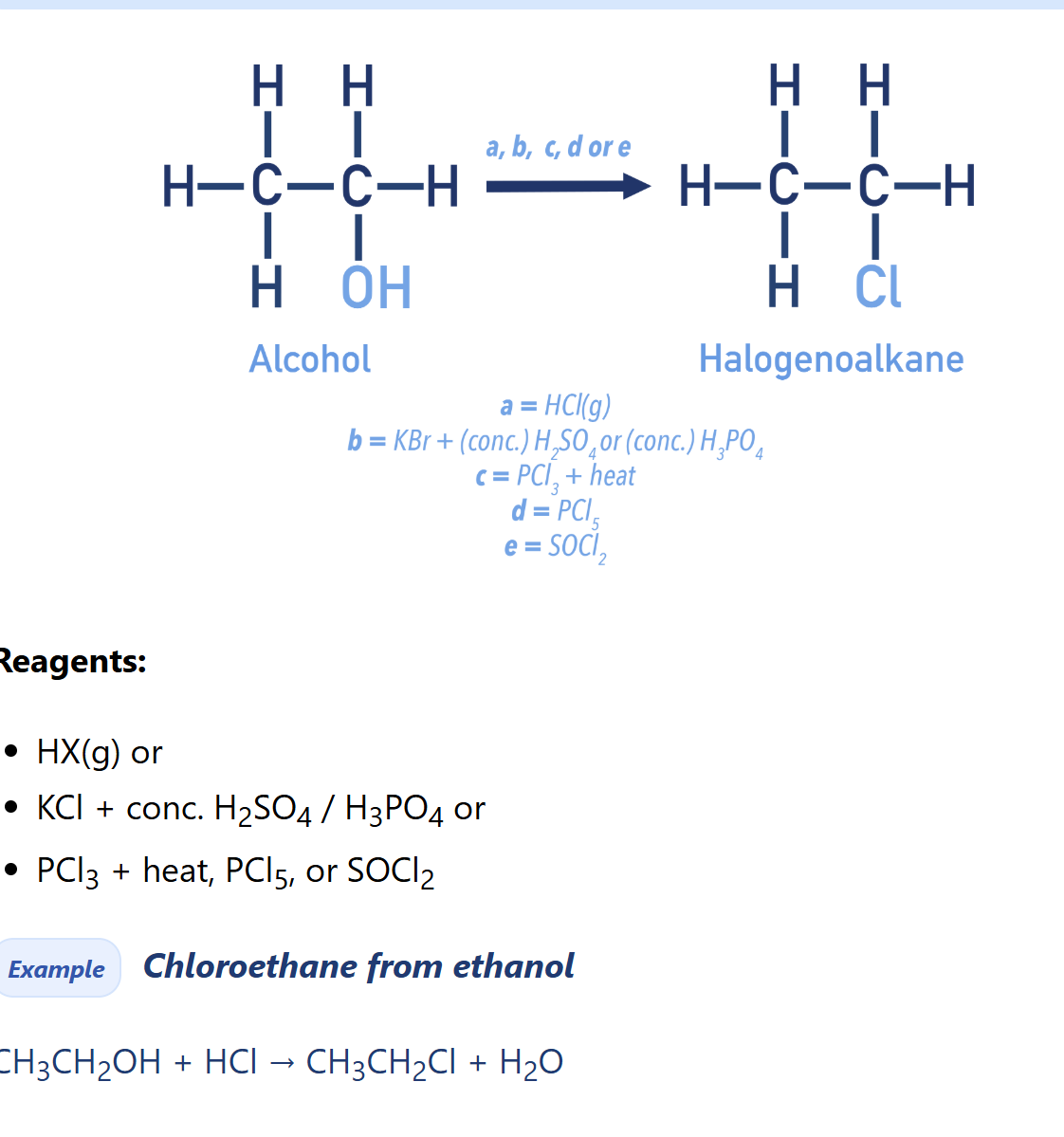
Types of halogenoalkane
Primary | halogen bonded to C with 1 alkyl group |
Secondary | bonded to C with 2 alkyl groups |
Tertiary | bonded to C with 3 alkyl groups |
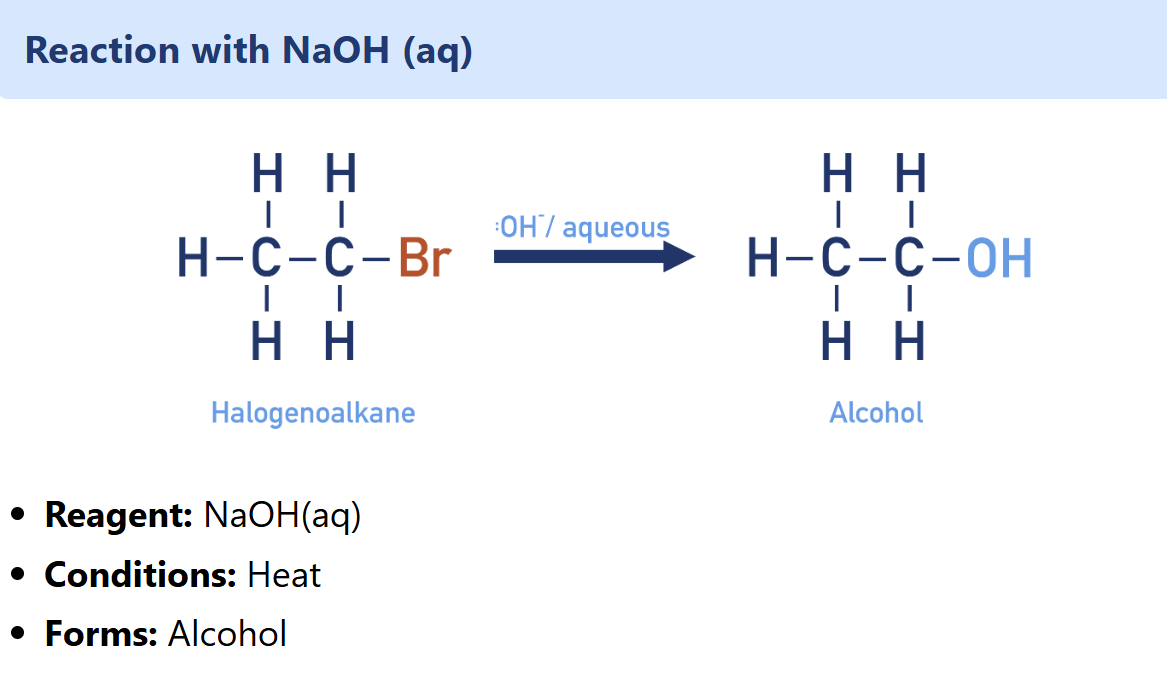
Halogenoalkane nucleophilic substitution with NaOH
Alcohol synthesis
NaOH must be aqueous
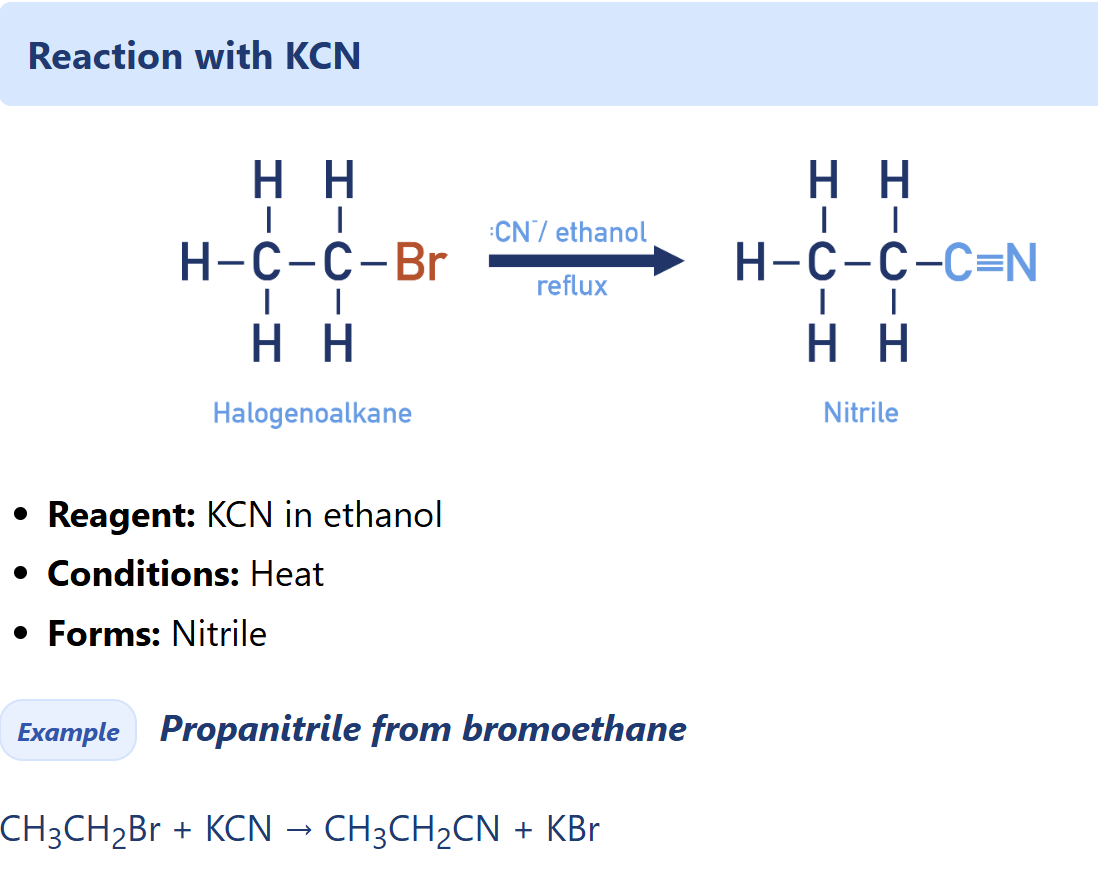
Halogenoalkane nucleophilic substitution with KCN
nitrile synthesis
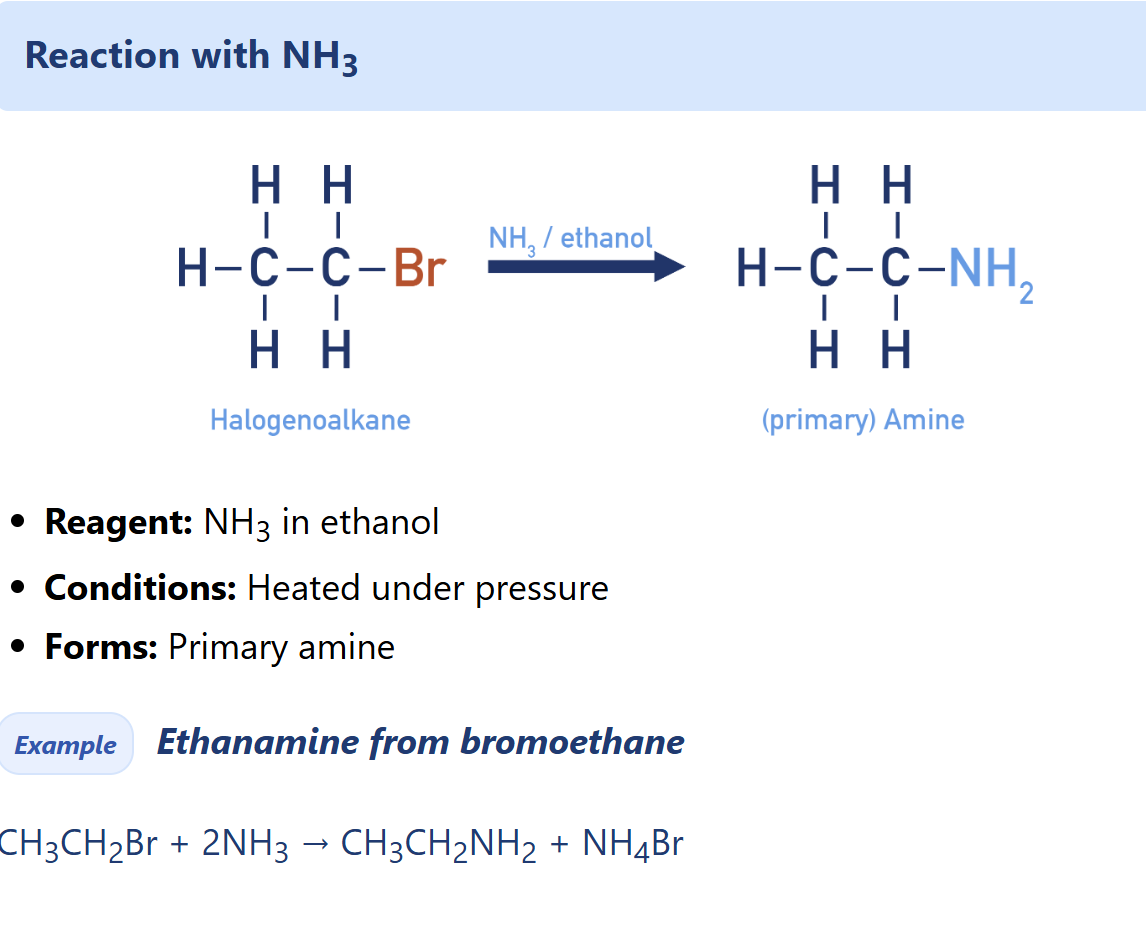
Halogenoalkane nucleophilic substitution with NH3
Test for identify halide in halogenoalkane (reagents, conditions, positive results for each halides Cl, Br, I)

Nucleophilic substitution reactions
when a nucleophile (electron pair donor) replaces a leaving group (a halide ion) in a halogenoalkane.
two possible mechanisms of nucleophilic substitution
SN1 and SN2
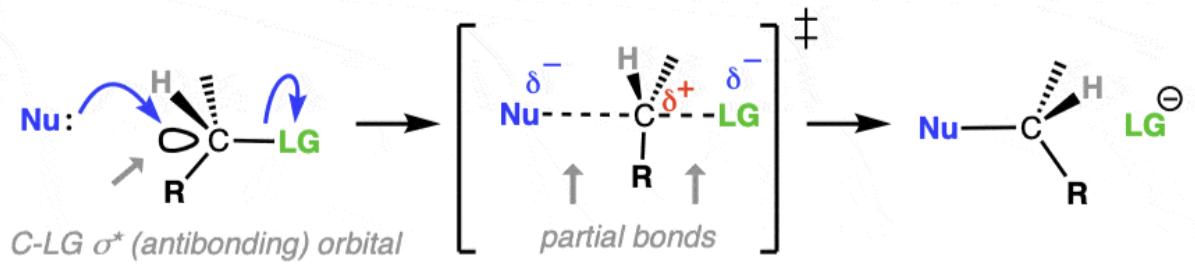
Describe SN2 mechanism
Occurs in one step (both reactants are involved in the same step).
The nucleophile attacks the carbon at the same time as the leaving group (halide) departs.
A transition state is formed with partial bonds — both the nucleophile and the leaving group are briefly attached.
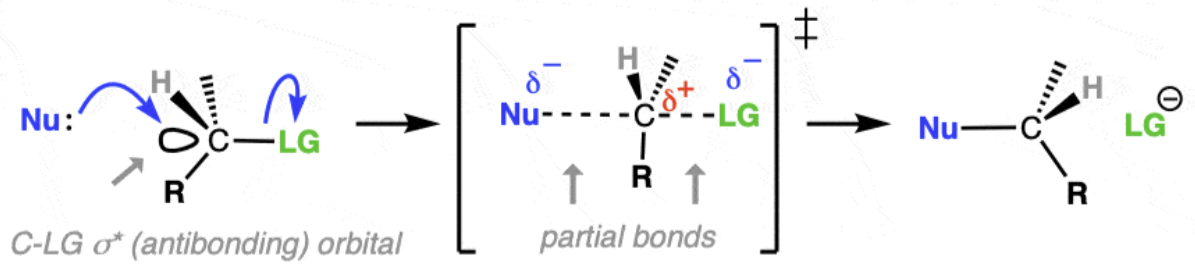
Which types of halogenoalkane favour SN2 mechanism
Favoured by primary halogenoalkanes (sometime secondary), because of less steric hindrance (blocked by carbon alkyl groups) allowing the nucleophile to easily from the back
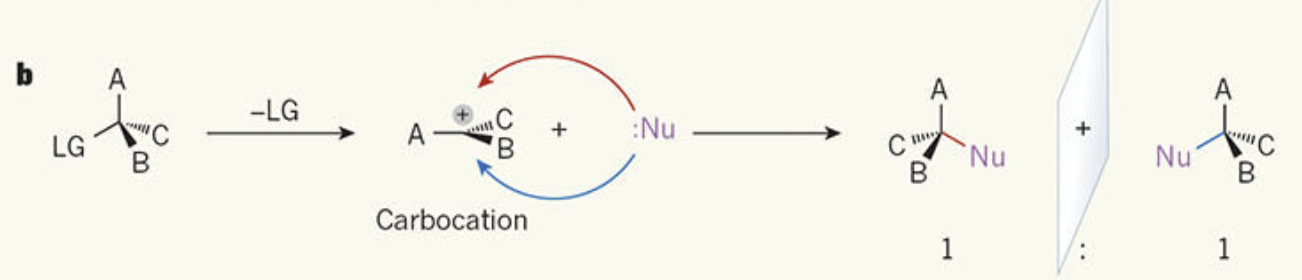
Describe SN1 mechanism
Occurs in two steps.
1. The halide leaves first, forming a carbocation (slow step – rate-determining).
2. The nucleophile then attacks the positively charged carbon (carbocation intermediate)
3. New bond formed between the C and OH.
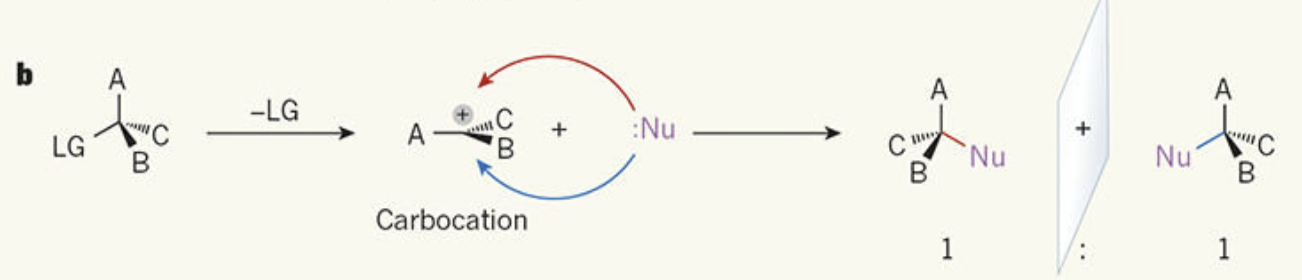
Which types of halogenoalkane favour SN1 mechanism
Favoured by tertiary halogenoalkanes, where the carbocation is stabilised by alkyl groups via the positive inductive effect (electron-donating effect of surrounding alkyl groups).
Order of reactivity (and rate of reaction) of halogenoalkane and explain why
Order of reactivity (fastest→slowest): Iodoalkanes > Bromoalkanes > Chloroalkanes > Fluoroalkanes
stronger bond strength → need more energy to break→ low reactivity
order of bond strength (strongest→weakest): C-F > C-Cl > C-Br > C-I
Alkane
Alkanes are saturated hydrocarbons, meaning they contain only single C–C and C–H bonds.
General formula: CnH2n+2
They are non-polar and insoluble in water.
Why alkanes are generally unreactive
C–C and C–H bonds are strong and non-polar.
Molecules have low polarity, so they don't attract polar reagents.
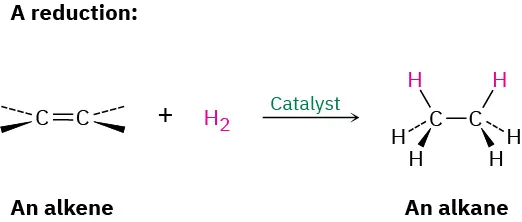
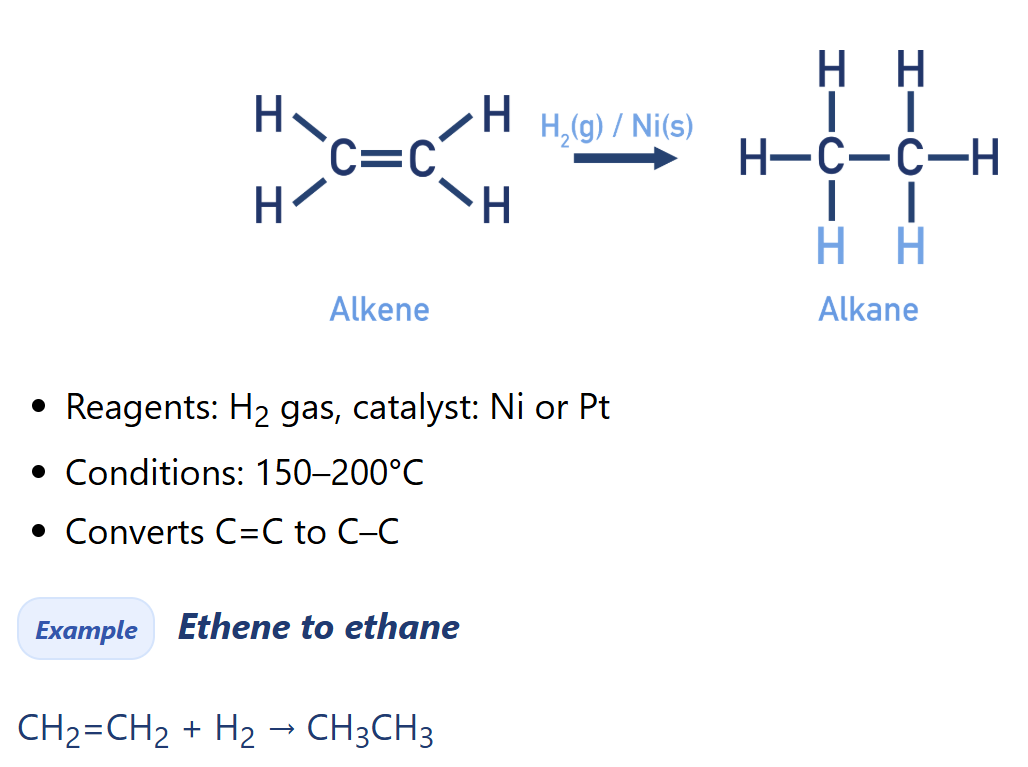
[Conditions] hydrogenation/ reduction of alkene [alkane synthesis]
Catalyst: Ni or Pt
Temp: 150 - 200 Celcius
Why need cracking
Cracking breaks large hydrocarbon molecules into smaller, more useful ones.
Shorter chain hydrocarbons are in greater demand for use of fuels (they ignite more easily and are less likely to undergo incomplete combustion).
[conditions] catalytic cracking (alkane and alkene synthesis)
alkane —> alkene + (shorter) alkane
Catalyst: Al2O3 or zeolite
Heat: 450–750°C
Complete combustion [reactions of alkane]
occurs when there is enough oxygen present and carbon can be fully oxidised
Alkane + O2 ——→ CO2 + H2O
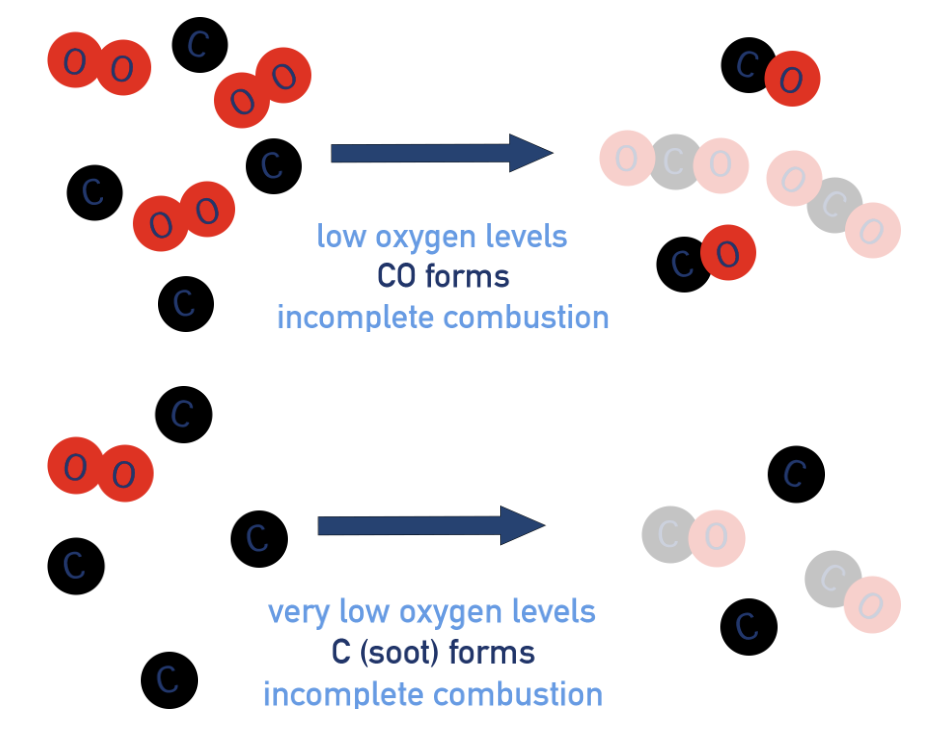
Incomplete combustion [reactions of alkane]
occurs when there is limited oxygen present and carbon can’t be fully oxidised, meaning carbon monoxide (CO) or carbon (soot) gets formed as a product (and water).
CH4 + 1.5O2 → CO + 2H2O
CH4 + O2 → C + 2H2O
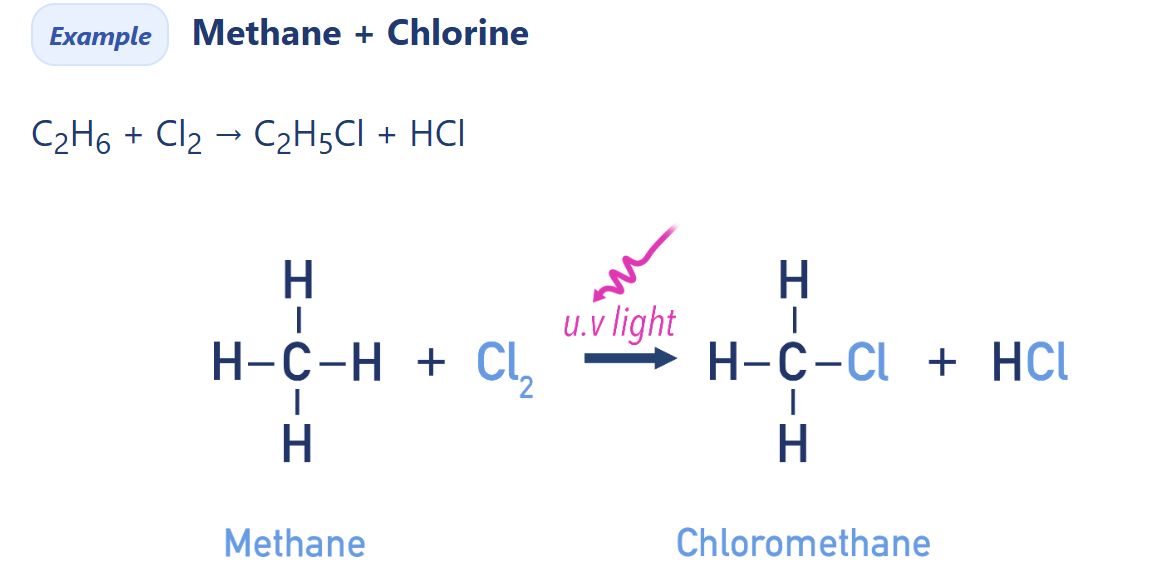
Free radical substitution
Alkanes react with halogens (Cl2, Br2) under UV light to form halogenoalkanes by free radical substitution.
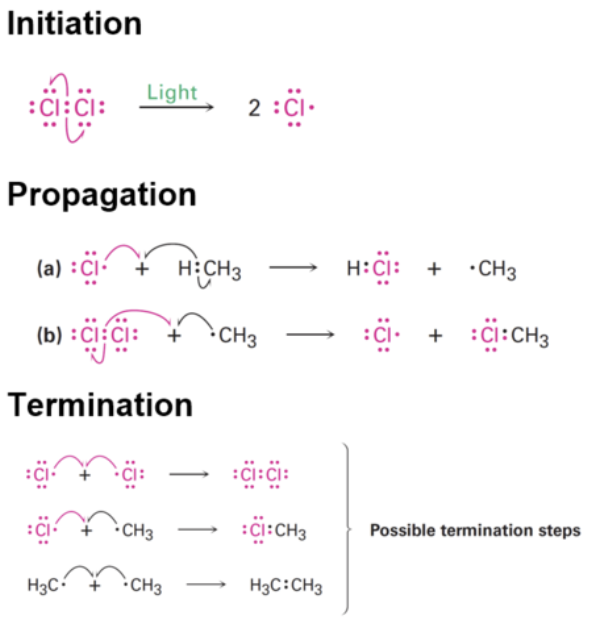
mechanism of free radical substitution
Step 1: Initiation
UV light provides energy to break the Cl–Cl bond by homolytic fission. Each chlorine atom ends up with an unpaired electron (•), making it a radical.Step 2: Propagation
Radicals react to form new radicals in a chain reaction. Chlorine radical reacts with methane, forming a methyl radical. Methyl radical reacts with Cl2, forming chloromethane and a new Cl• radical.Step 3: Termination
Radicals combine to form stable (non-radical) molecules, stopping the reaction.
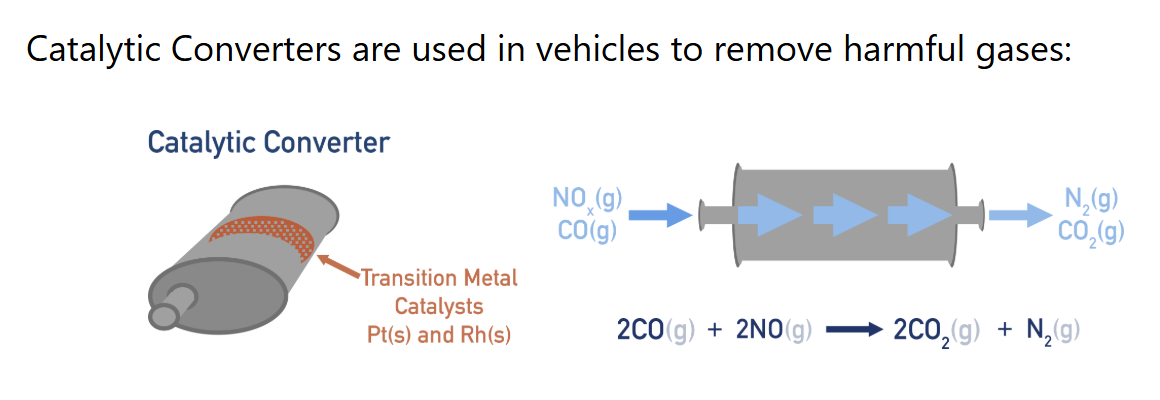
Which harmful molecules formed from Internal Combustion Engines
CO, NO and NO2, C, unburnt hydrocarbon
Harmful effects of CO, oxides of nitrogen, C, and unburnt hydrocarbon
Carbon Monoxide, CO
binds to haemoglobin and is toxicNitrogen Oxides, NO and NO2
formed when N2 and O2 react at high temps; causes acid rain and smogCarbon, C (soot)
causes respiratory problemsUnburnt hydrocarbons
contribute to photochemical smog
How harmful products of internal combustion engine removed
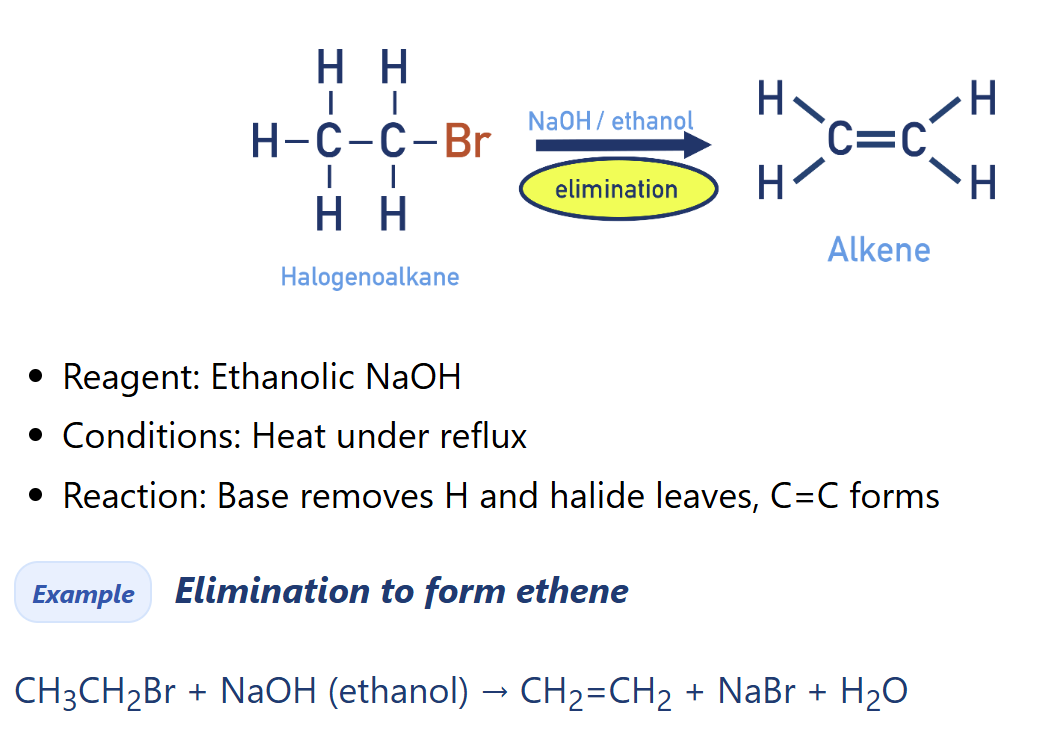
Describe elemination of HX from Halogenoalkanes [alkene synthesis]
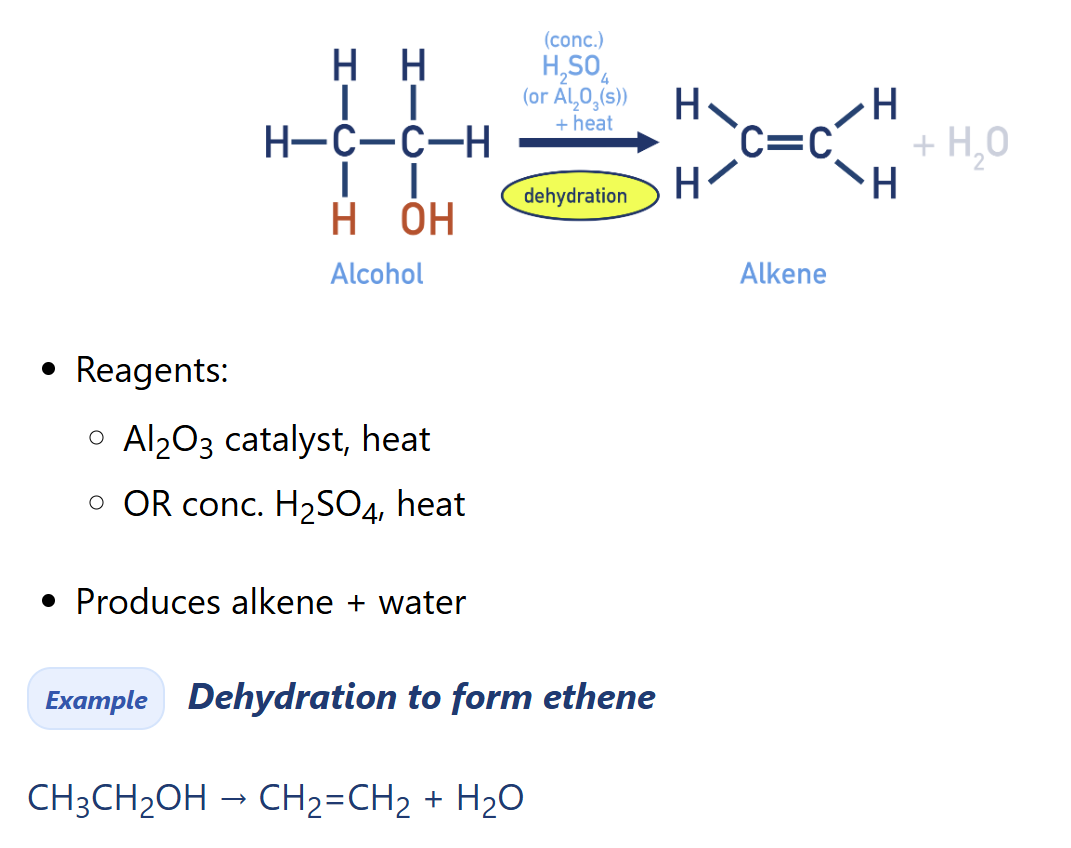
Describe dehydration of alcohol [alkene synthesis]
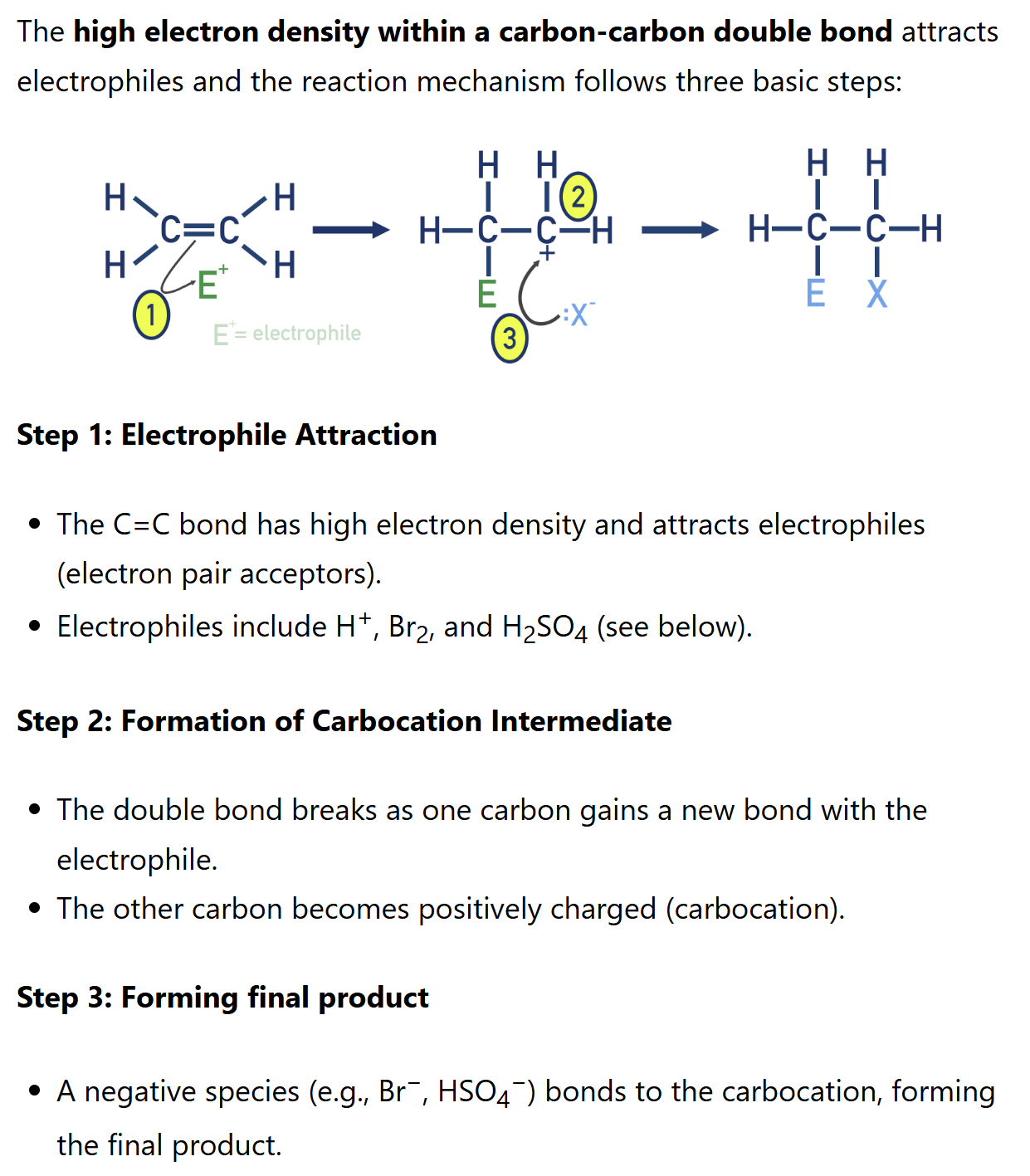
Alkene electrophilic addition
Alkenes react with electrophiles in electrophilic addition reactions. The high electron density within a carbon-carbon double bond attracts electrophiles.
Alkene electrophilic addition of hydrogen
Alkane synthesis
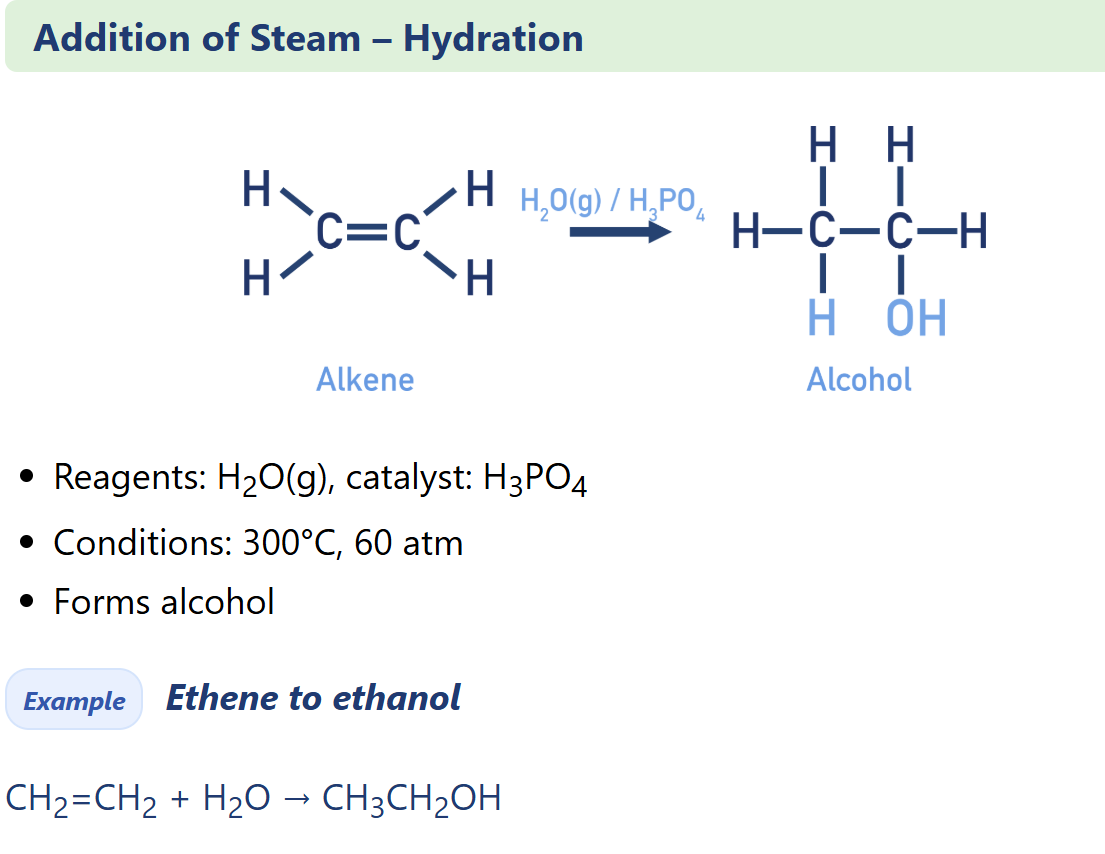
Alkene electrophilic addition of steam
Alcohol synthesis
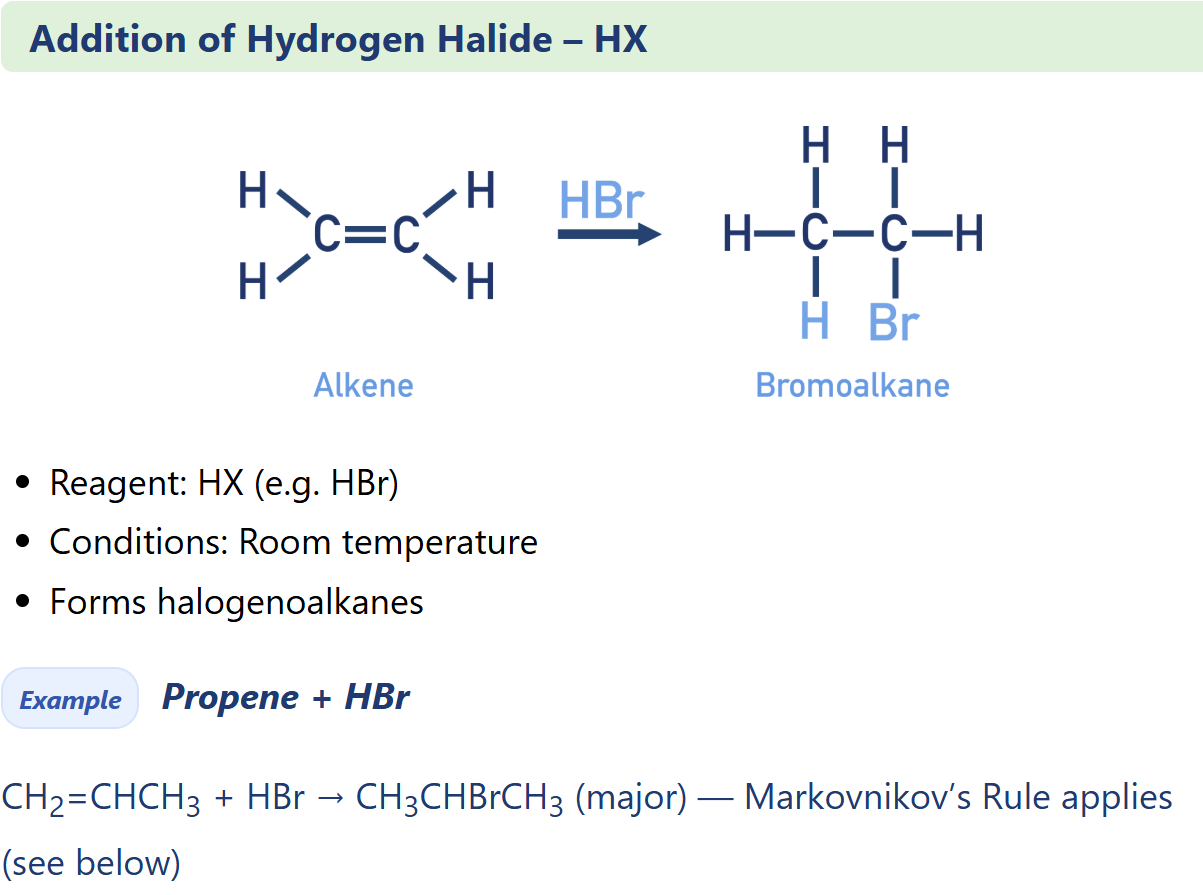
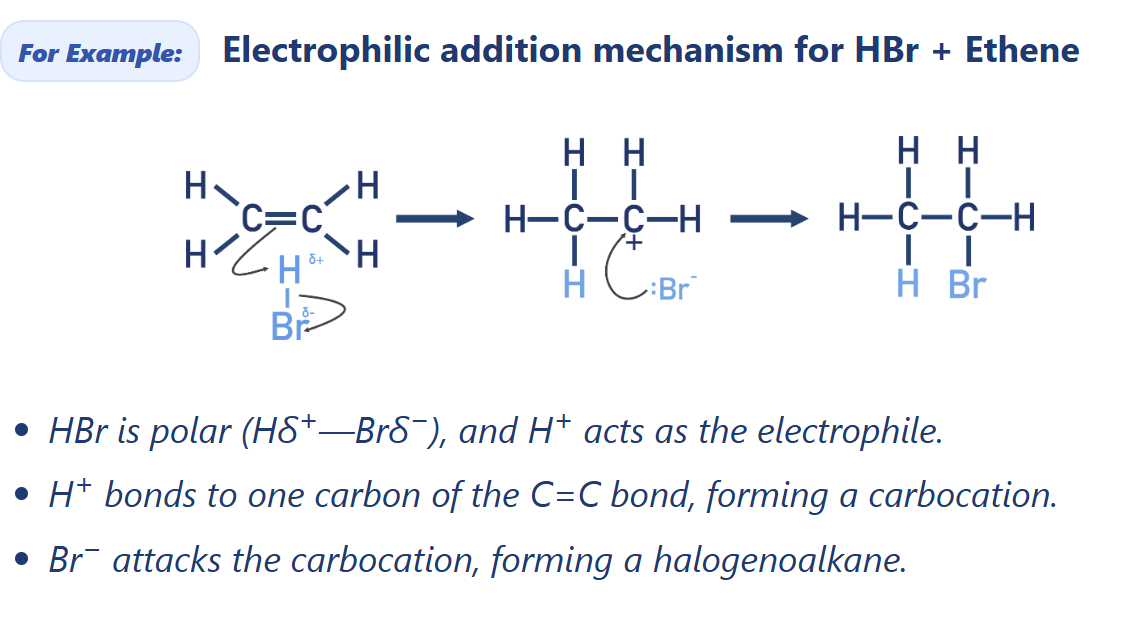
Alkene electrophilic addition of hydrogen halide (HX(g))
Halogenoalkane synthesis
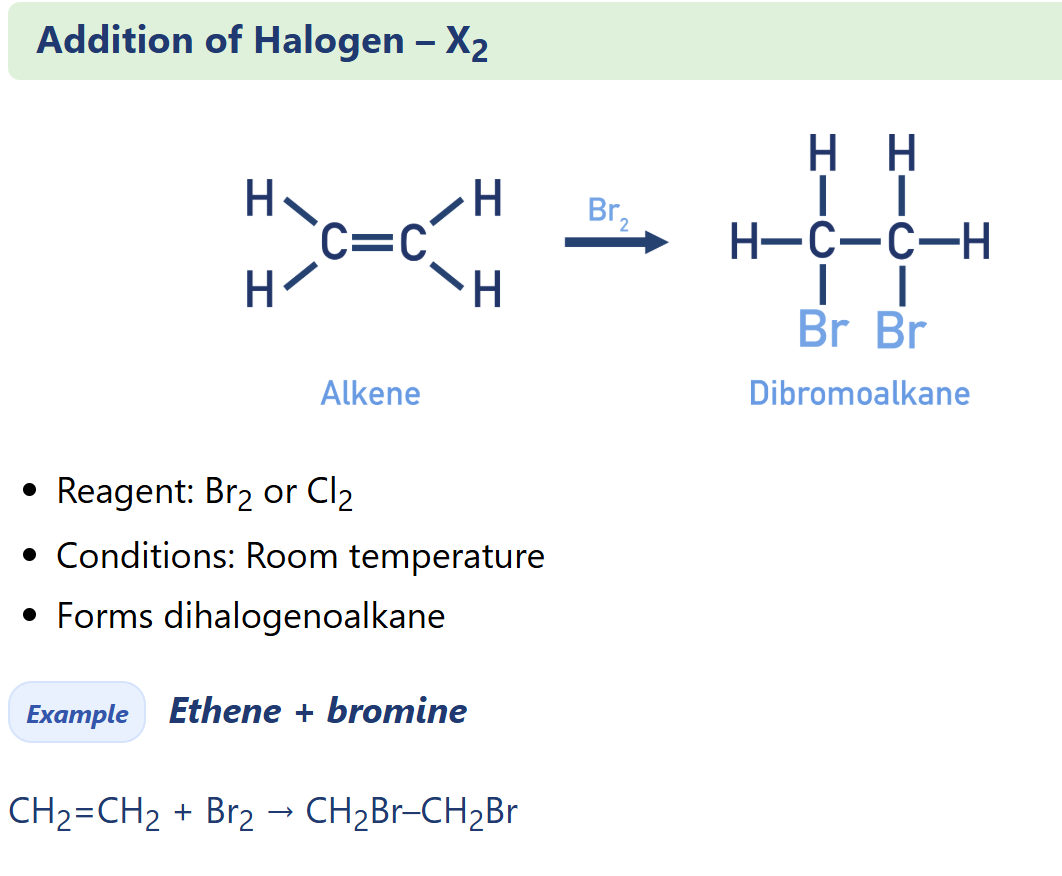
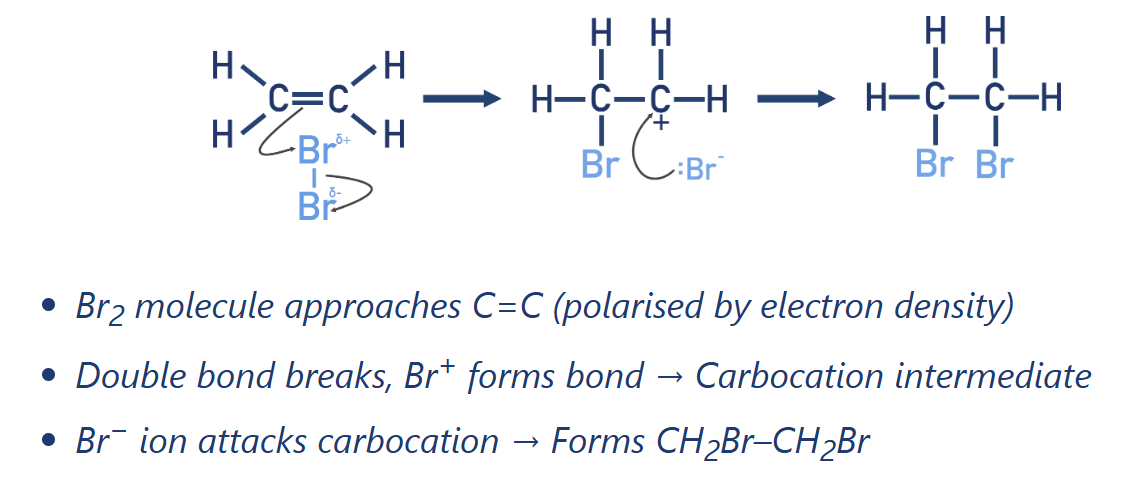
Alkene electrophilic addition of halogen (X2)
Halogenoalkane synthesis
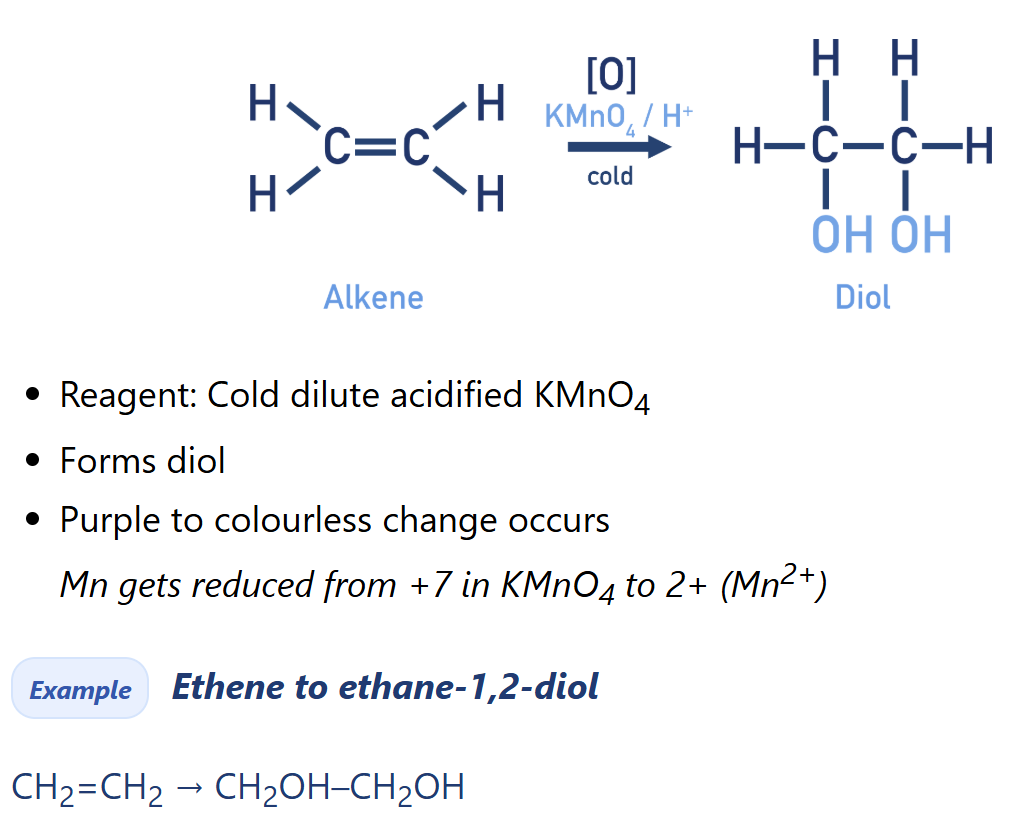
Alkene oxidation with Cold Dilute KMnO4 (Potassium permanganate)
Diol synthesis
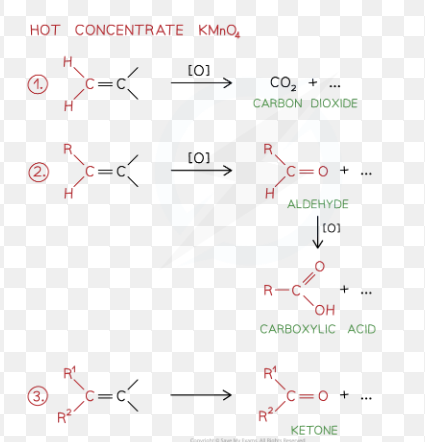
Alkene oxidation with Hot Conc. KMnO4 (Potassium permanganate)
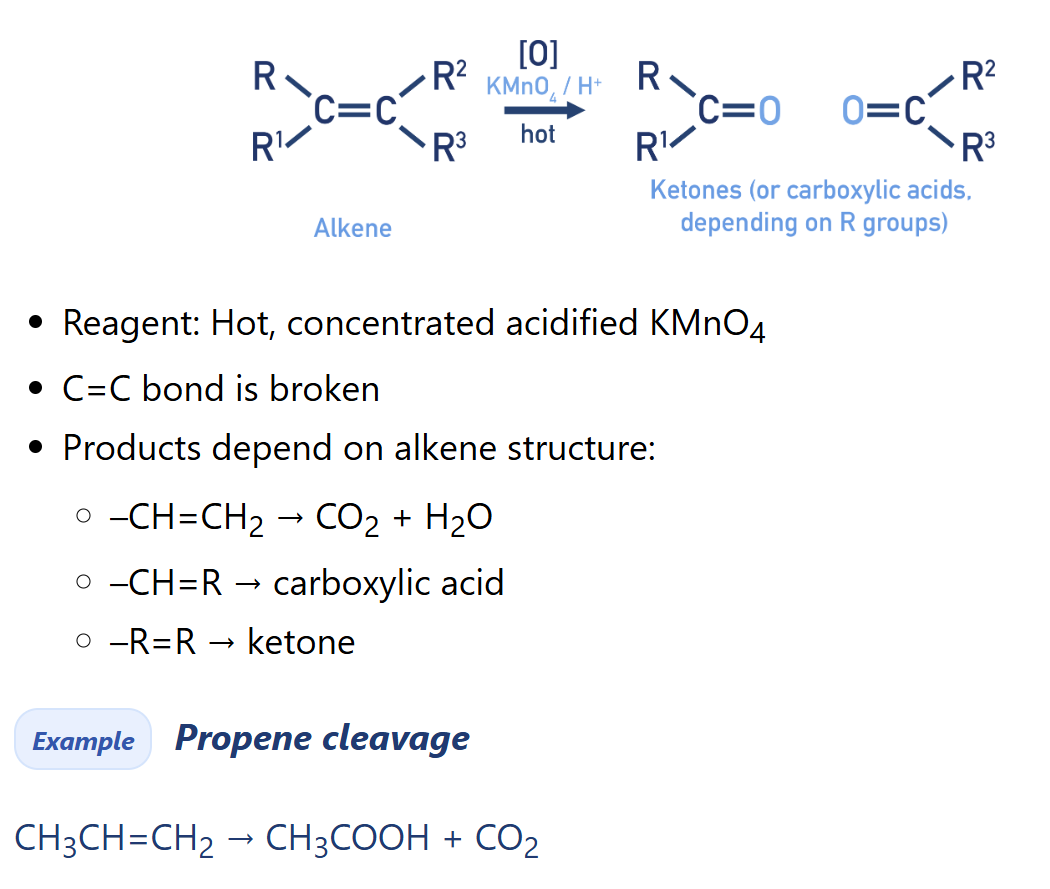
Cleavage (in chemistry)
splitting or breaking of bonds between ions, atoms, molecules
How many alternative products are there in alkene oxidation with Hot Conc. KMnO4
3 product pathways (depend on the cleavage of the bond in alkene)
Describe addition polymerisation
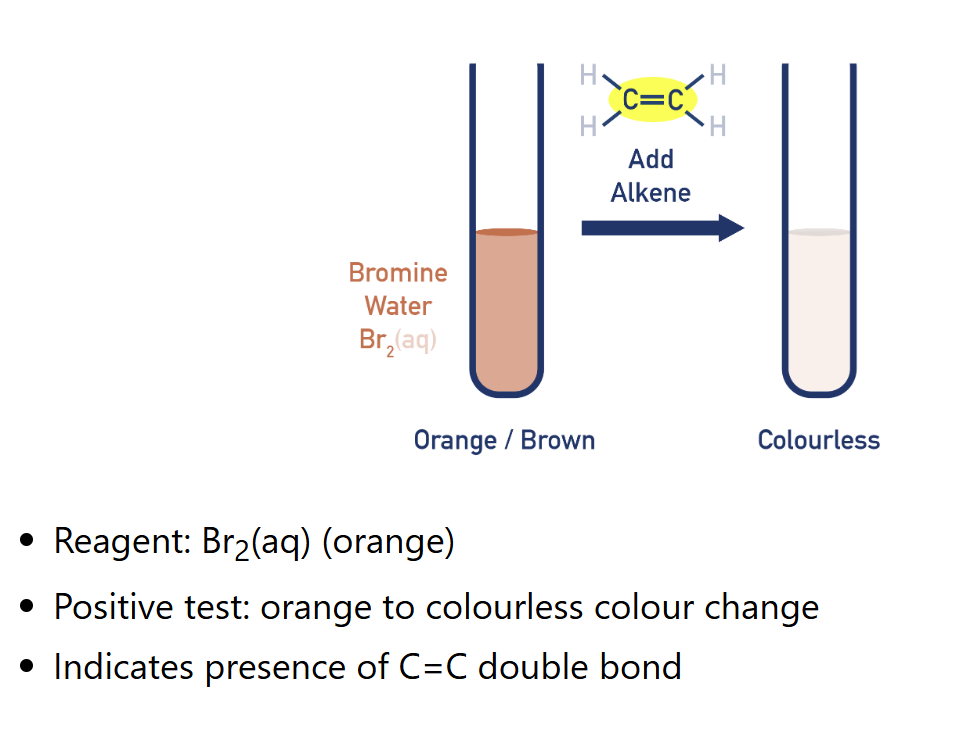
Describe test for alkene (Bromine water)
Mechanism of electrophilic addition
Draw electrophilic addition mechanism for Bromine + Ethene
Draw electrophilic addition mechanism for HBr + Ethene
Major and minor product
Major : products that formed the most
Minor : products that formed less
Alkyl groups
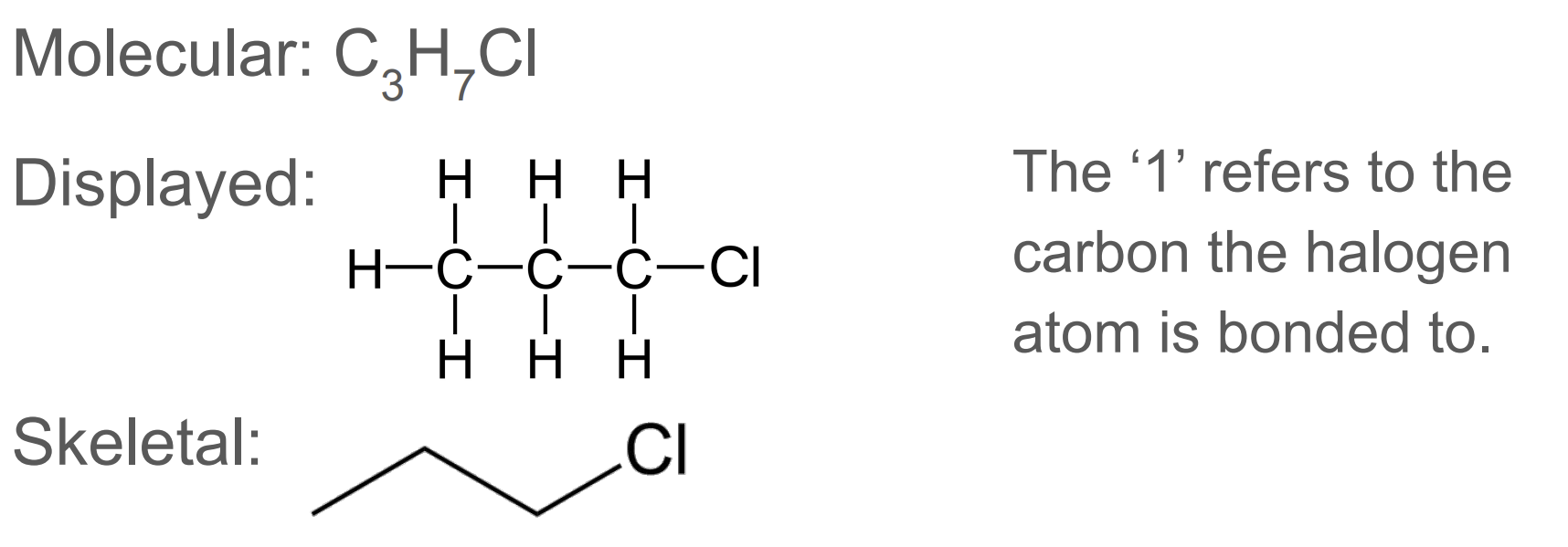
A chain of carbon backbone (CH3, C2H5, C3H7…)
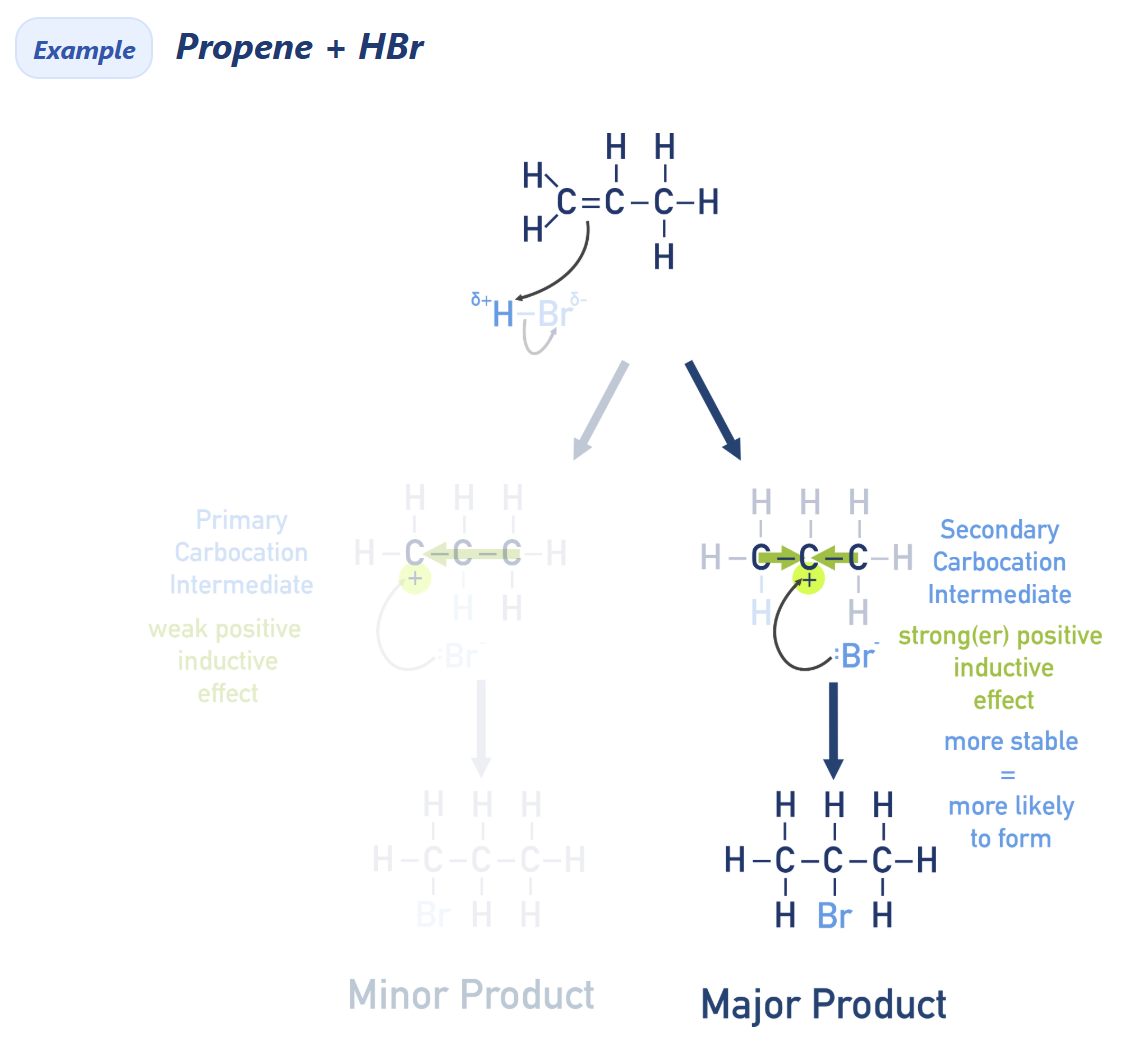
Markovnikov’s rule
Major product will be the one where H from HX bonds to carbon in C=C that is bonded to the most hydrogens
Carbocation
a carbon positively charged ion
Types of carbocation and their order of stability
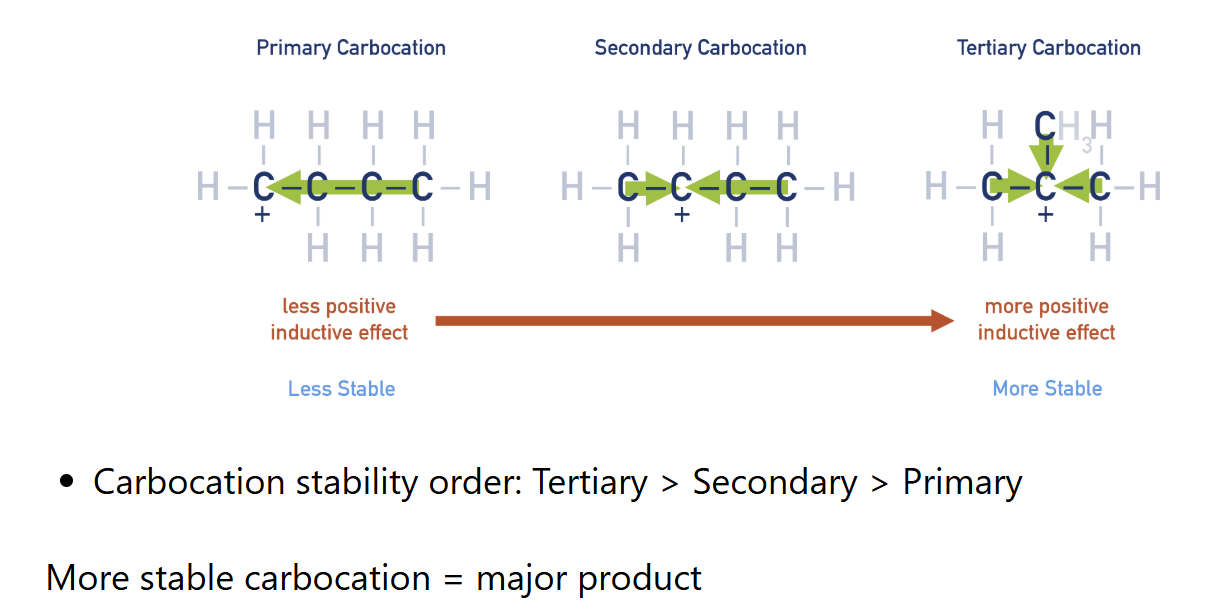
Inductive effect
When alkyl groups gives electron density to the carbocation in order to stabilize it. More alkyl groups bonded, more stable carbocation.
Use inductive effect to explain Markovnikov’s rule
HX get polarised
Hydrogen (+) bonded with less stable carbon (C=C bond)
X (or any other species) bonded with more stable carbocation

a group of atoms that determine a molecule’s physical and chemical properties

A family of compounds with the same functional group and general formula. Also, each successive members differ by a unit of -CH2-

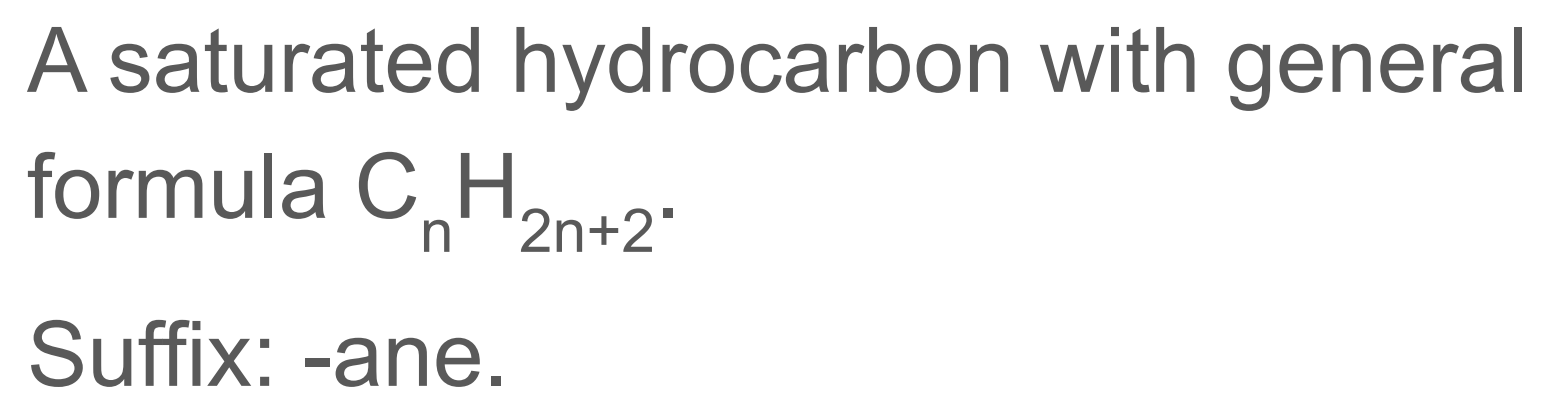
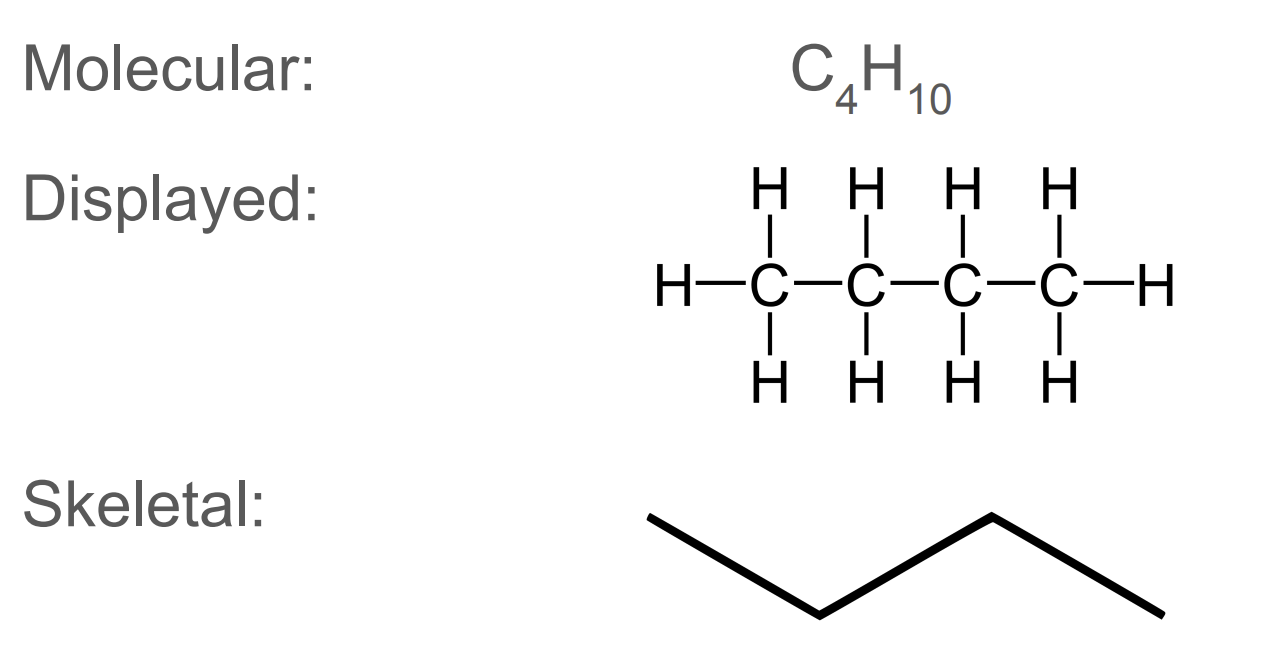






![<p>[and its general formula]</p>](https://assets.knowt.com/user-attachments/bacd1dd0-bd45-4b17-9ce6-824dc0633847.png)
[and its general formula]
CnH2n+1X where X is a halide