E.1 Structure of the atom
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
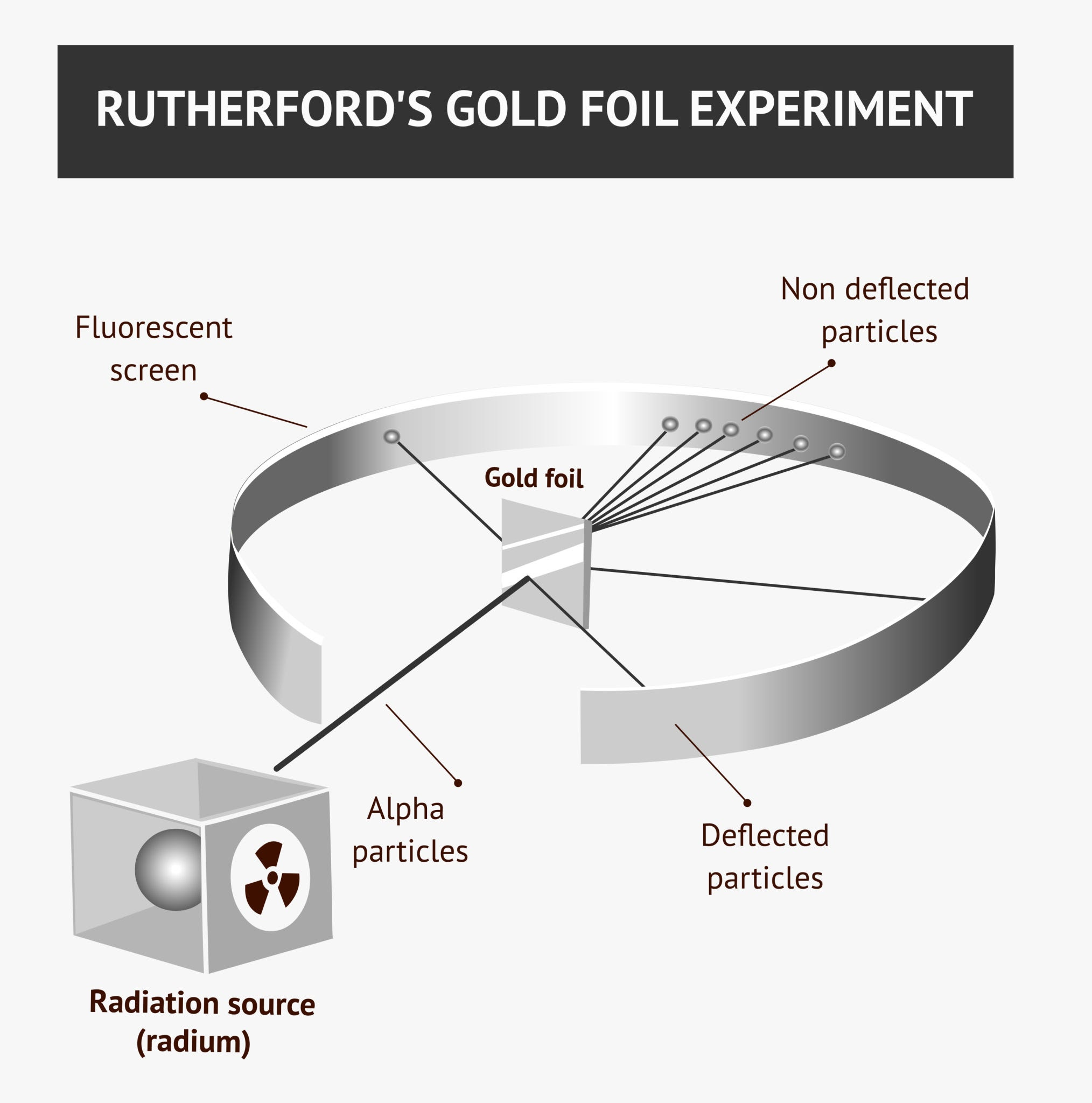
What did the Geiger–Marsden–Rutherford experiment reveal about the atom?
It showed that the atom is mostly empty space with a small, dense, and positively charged nucleus at its center.
In the nuclear notation Z-X-A, what do A and Z represent?
A is the nucleon number (total protons and neutrons) and Z is the proton number (total protons).
How do emission and absorption spectra prove the existence of discrete energy levels?
They consist of distinct lines rather than a continuous spectrum, showing that electrons only exist at specific, fixed energy values.
What happens to a photon during an atomic transition to a lower energy state?
A photon is emitted with an energy exactly equal to the difference between the two energy levels.
What is the formula for the energy of a photon in words?
Energy equals the Planck constant times the frequency of the light.
What is the relationship between a nucleus radius and its nucleon number?
The radius is proportional to the cube root of the nucleon number.
What is an implication of the radius formula regarding nuclear density?
It implies that the density of a nucleus is approximately constant and independent of the size of the nucleus.
Why do deviations from Rutherford scattering occur at very high energies?
At high energies, the incoming particle gets close enough to the nucleus for the strong nuclear force to overcome the electric repulsion.
How is the distance of closest approach calculated in head-on scattering?
By setting the initial kinetic energy of the particle equal to the electric potential energy at the point where it momentarily stops.
What is the formula for energy levels in the Bohr model for hydrogen?
Energy in electronvolts equals negative thirteen-point-six divided by the principal quantum number squared.
What is the condition for the quantization of angular momentum in the Bohr model?
Angular momentum (mass times velocity times radius) must equal an integer times the Planck constant divided by the quantity two times pi.
How can emission spectra be used to identify chemical composition?
Every element has a unique set of energy levels, meaning every element produces a unique "barcode" of spectral lines.
In the context of atomic transitions, what does h represent?
It represents the Planck constant, which links the frequency of a photon to its energy.