Med Chem Last Quiz (L36-38)
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
Metaxalone
-skeletal muscle relaxant
-used as adjunct to relieve discomfort associated with acute and painful musculoskeletal conditions
Baclofen (Liorseal)
-skeletal muscle relaxant
-GABA-B agonist (pre-synaptic) --> Decrease release of excitatory neurotransmitters
-structure: GABA with phenyl ring attached (Cl on phenyl blocks hydroxylation)
-rapidly absorbed, little metabolism (most eliminated unchanged)
-less sedation than diazepam
-used to treat spasticity associated with MS and trauma to spinal cord
Cyclobenzaprine
-skeletal muscle relaxant for muscle spasms
-Amrix: ER formulation for QD dosing
-extensive metabolism: tertiary amine demethylation (Active metabolites), epoxide formation in middle ring double bond
-MOA unknown
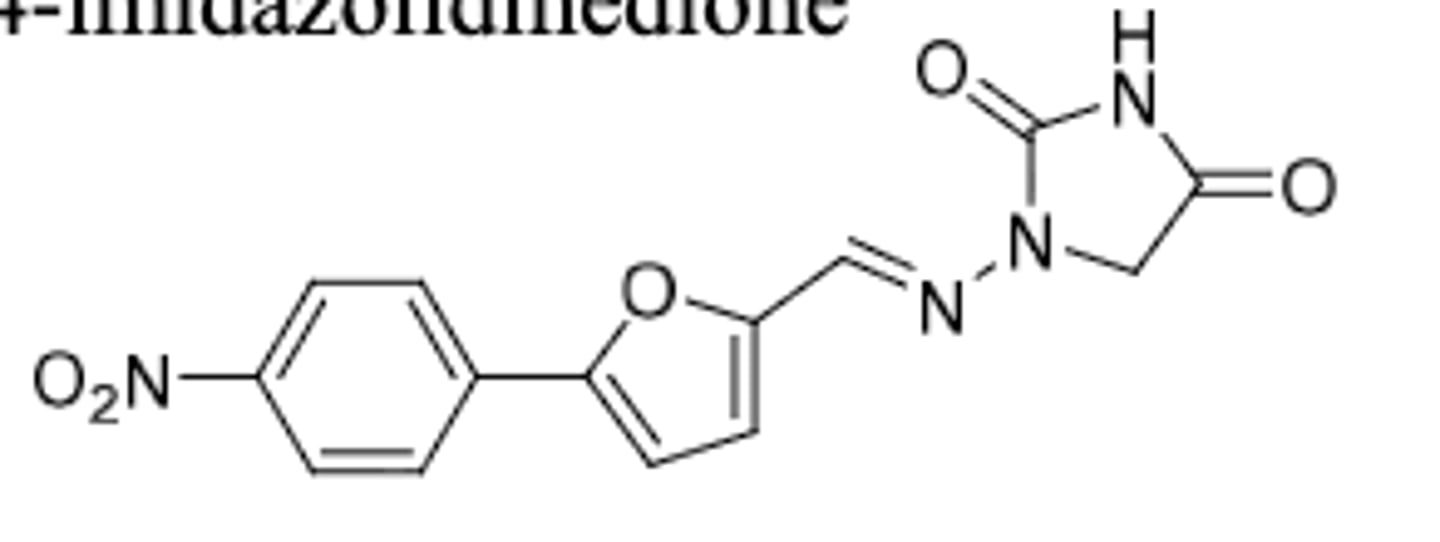
Chlorzoxazone
-acts on spinal cord by expressing reflexes
-skeletal muscle relaxant used for treatment of muscle spasm, strains, sprains, or back pain
Dantrolene
-ryanodine receptor (RyR) antagonist --> slowed neurotransmission by controlling calcium release
-used for Malignant Hypothermia (MH) --> deadly condition where skeletal muscle increases oxidative metabolism, too much CO2 produces, and body fails to regulate temperature
-IV first for initial dose, followed by oral
-side effect: can cause muscle weakness and hepatic toxicity
-PO can also be used to treat spasticity from stroke, MS, spinal cord injury, and cerebral palsy
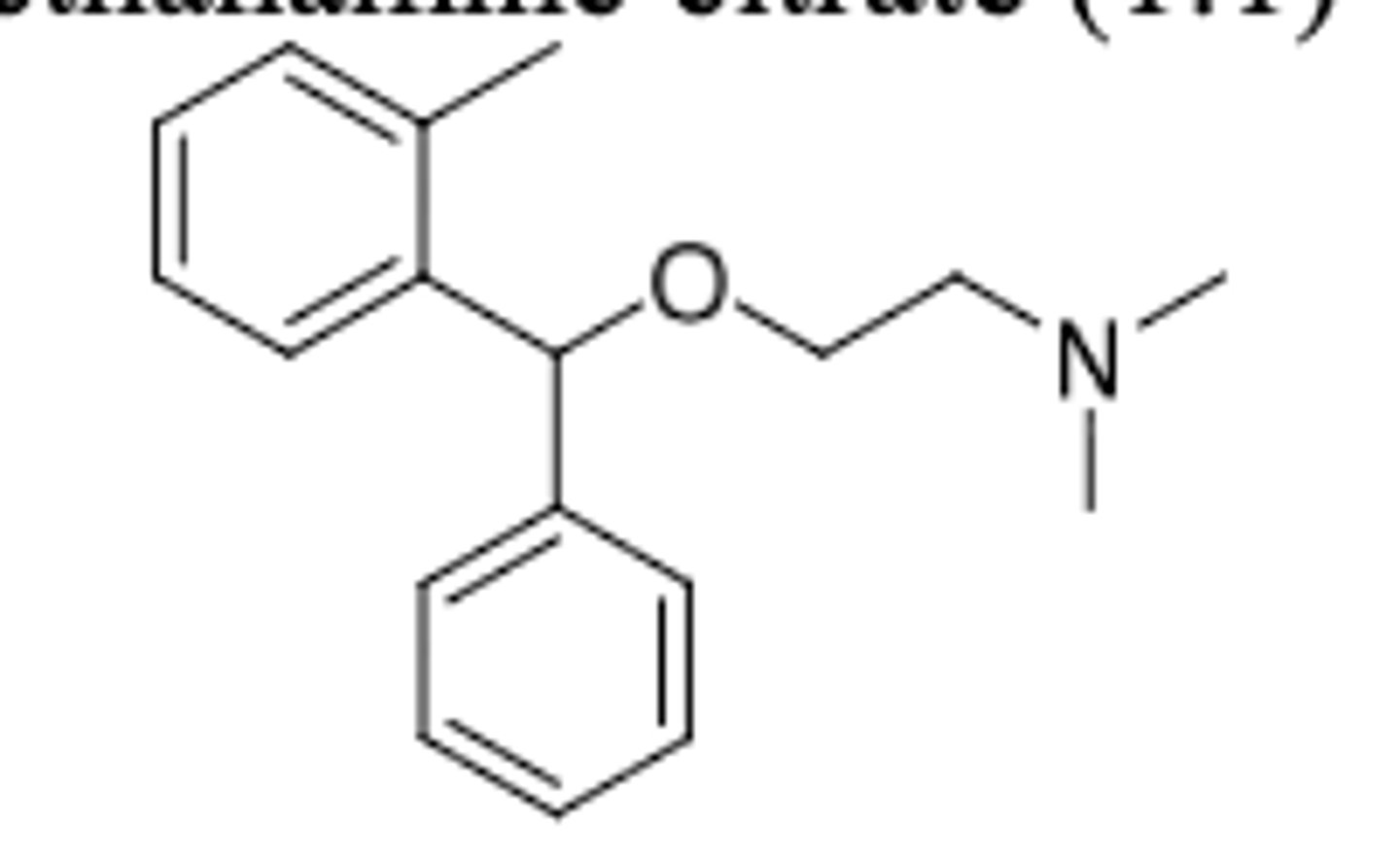
Orphenadrine
-very similar structure to diphenhydramine (with additional methyl on top benzene)
-skeletal muscle relaxant as adjunct to relieve discomfort with acute and painful musculoskeletal conditions
-may also be used to treat leg cramps
-has anticholinergic effects
Tizanidine
-Alpha 2 agonist
-skeletal muscle relaxant for acute and intermittent management of increased muscle tone associated with spasticity
-1/10 - 1/50 the activity of clonidine BP lowering effects
Schizophrenia: a severe mental disorder characterized by distortions in thinking, perception, emotions, language, sense of self and behavior
These agents produce a calming effect in agitated individuals and they induce a lessening of reactivity to emotional stimuli without affecting consciousness
Many of the conditions are believed to be due to imbalances of monoamine neurotransmitters such as dopamine, norepinephrine, and serotonin in the brain. These imbalances lead to mood and behavioral alterations.
Much evidence suggests that the antipsychotic agents function by blocking dopamine at the post-synaptic D-2 receptors. This is how the phenothiazines and
butyrophenones work.
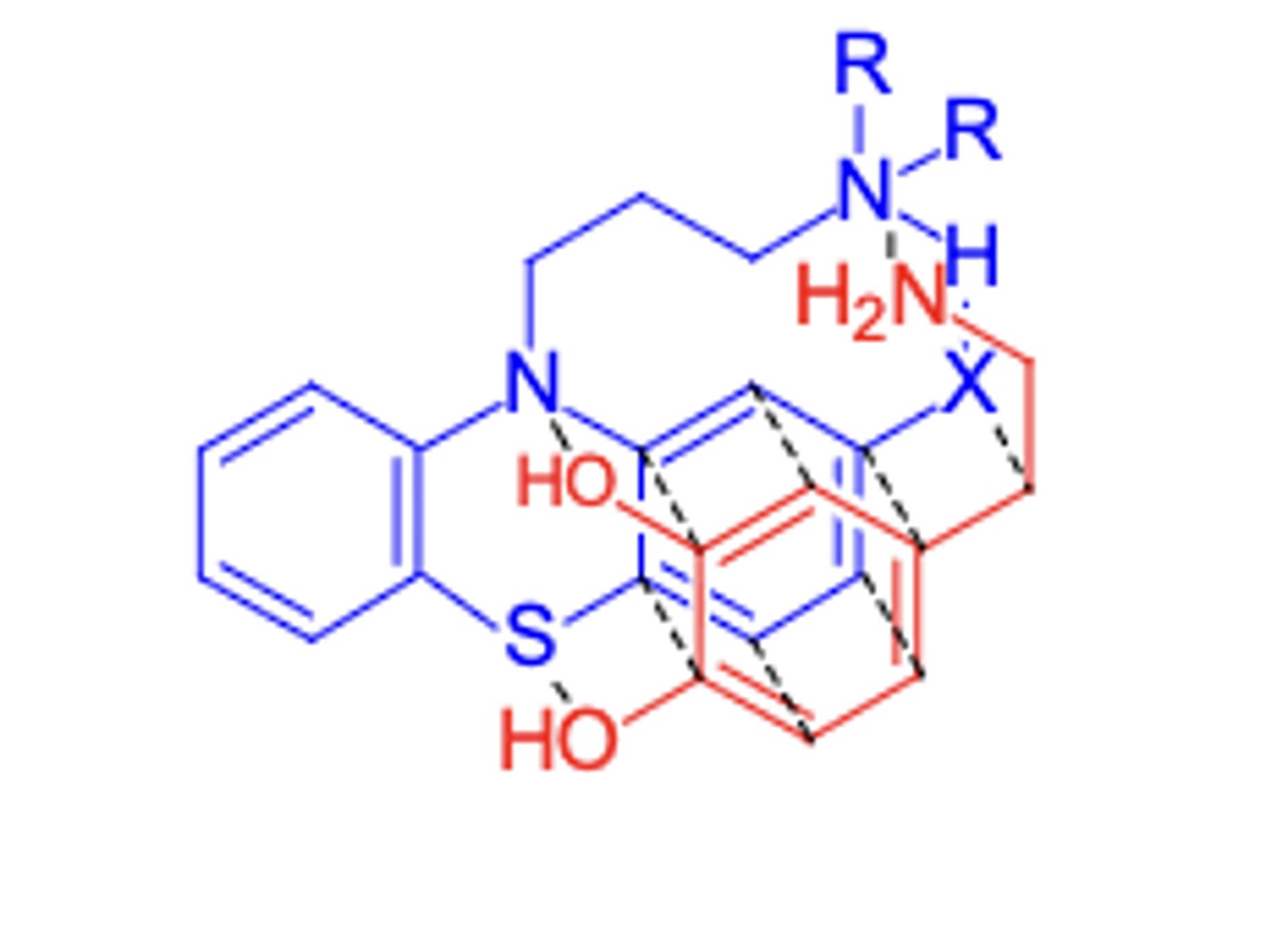
Antipsychotic agents
-X: EWG at 2 position to increase activity (more EW = hihger neuroleptic potency)
-3-Carbon chain between two N's is optimal for maximum neuroleptic activity
-R's should be dimethyl or alkyl groups of <2 linear C's
-A piperazine or piperazineethanol group as side chain gives enhanced activity (piperazineethanol > piperazine > dimethylamino)
Phenothiazines have antiemetic and anticholinergic effects
Packed in dark container to prevent oxidation --> increases shelf life for all phenothiazines (prevent oxidation from air)
Phenothiazine SAR
Chlorpromazine
-aliphatic phentothiazine
-D2 antagonist --> Antipsychotic and control of N/V
Metabolism:
-sulfur oxidized to sulfoxide (one double bond to O) then further oxidized to sulfone (2 double bonds to O) leading to inactivation
-7-hydroxy metabolite
-demethylation on tertiary amine
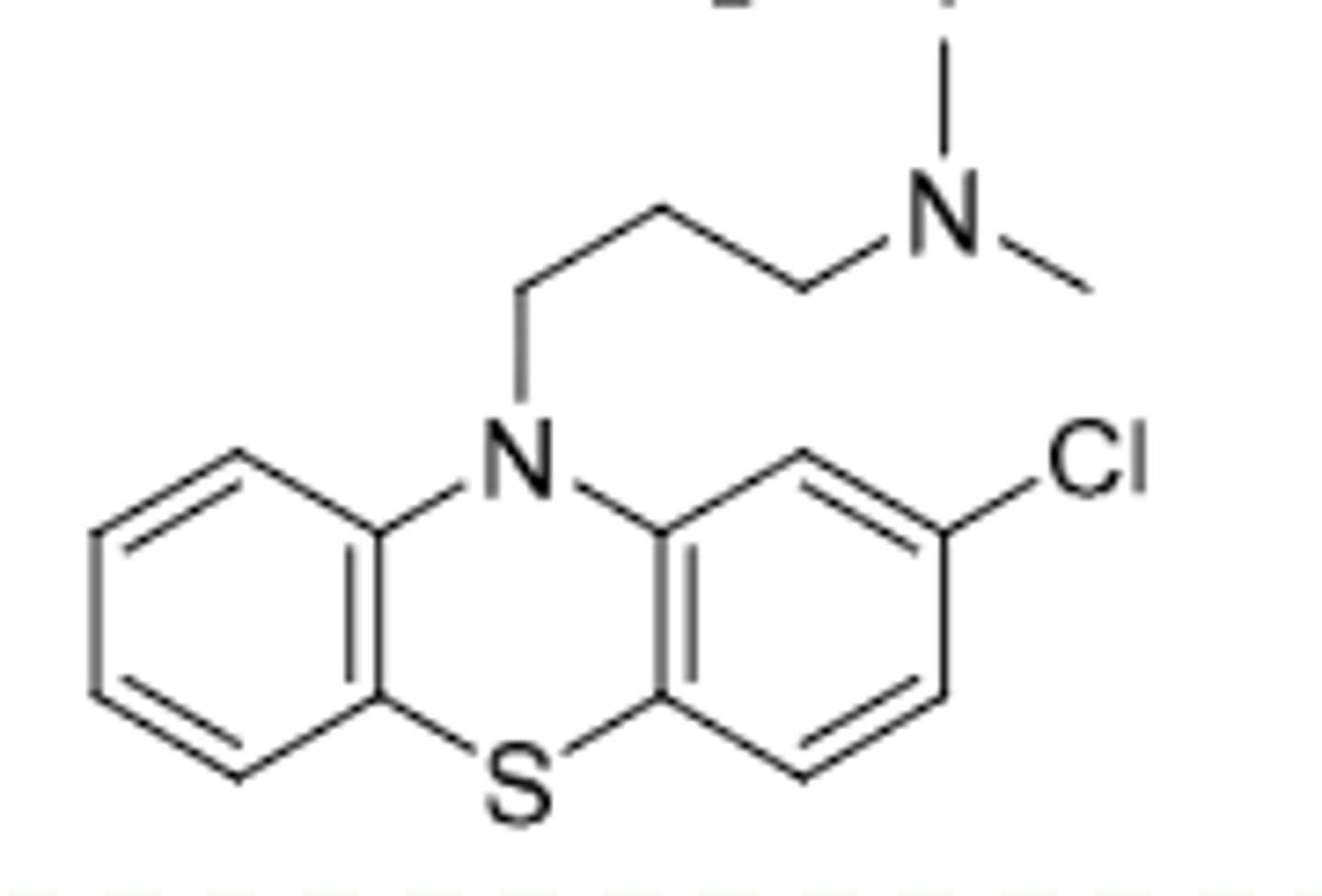
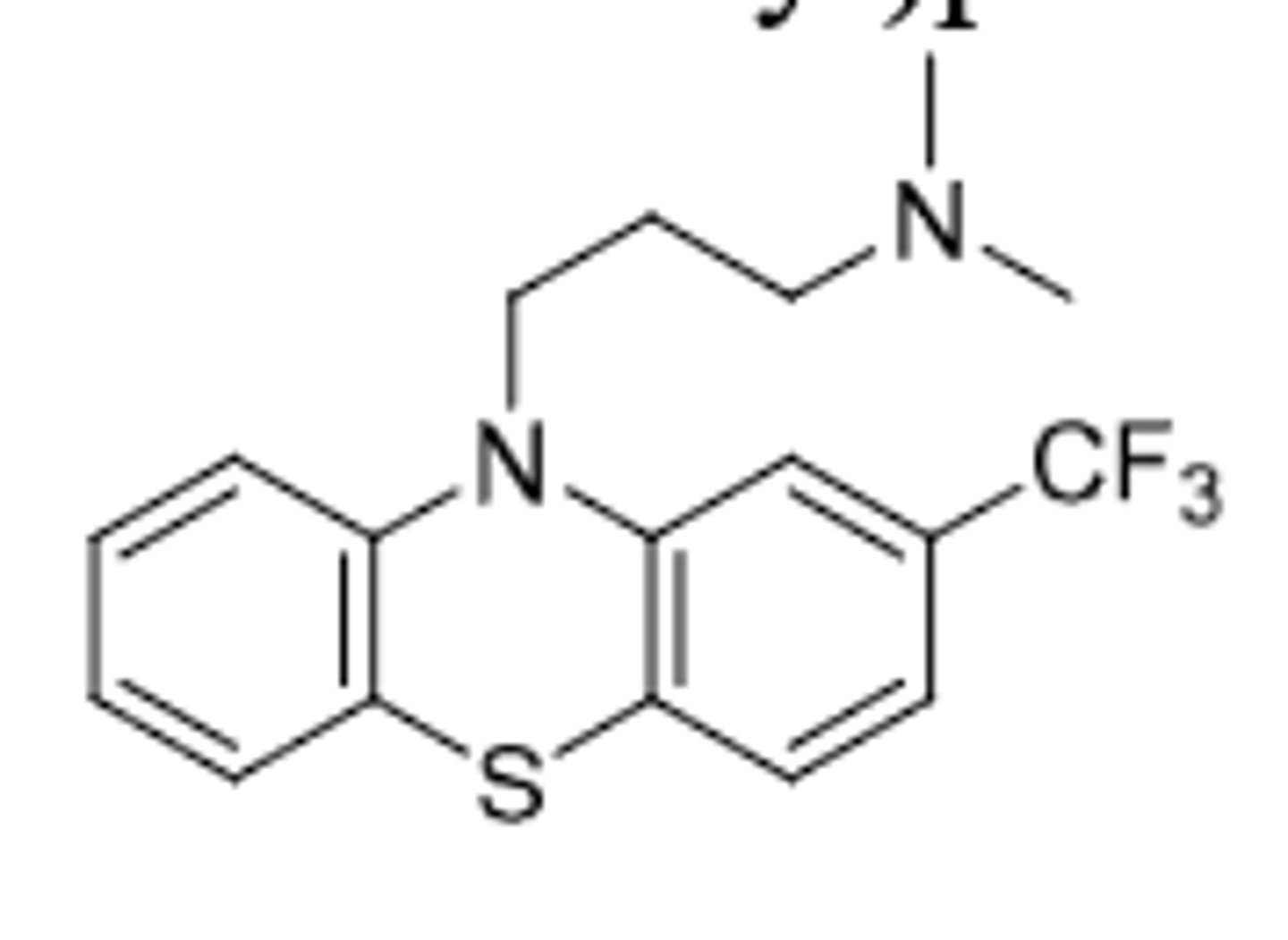
Triflupromazine
-aliphatic phenothiazine
-D2 antagonist --> Antipsychotic and control of N/V (same as chlorpromazine)
-only difference from chlorpromazine is EWG --> the CF3 (increased lipophilicity)
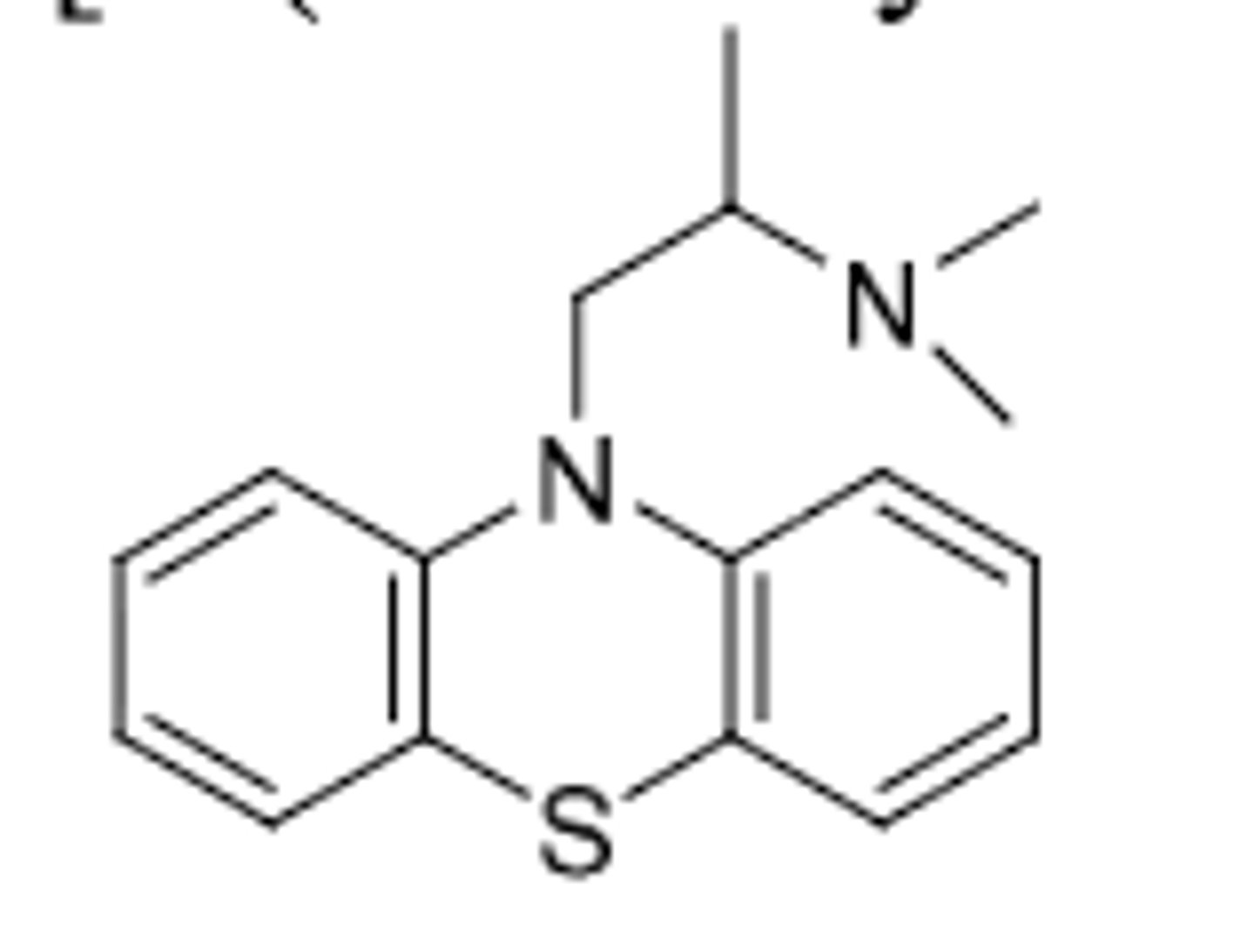
Promethazine
-aliphatic phenothiazine
-Very weak antipsychotic (missing structural features including only 2C linker and no EWG)
-1/10 activity of chlorpromazine
-used for control of motion sickness and post operative patients (anti emetic effects); also has antihistamine activity
-NOT used for psychosis as antipsychotic
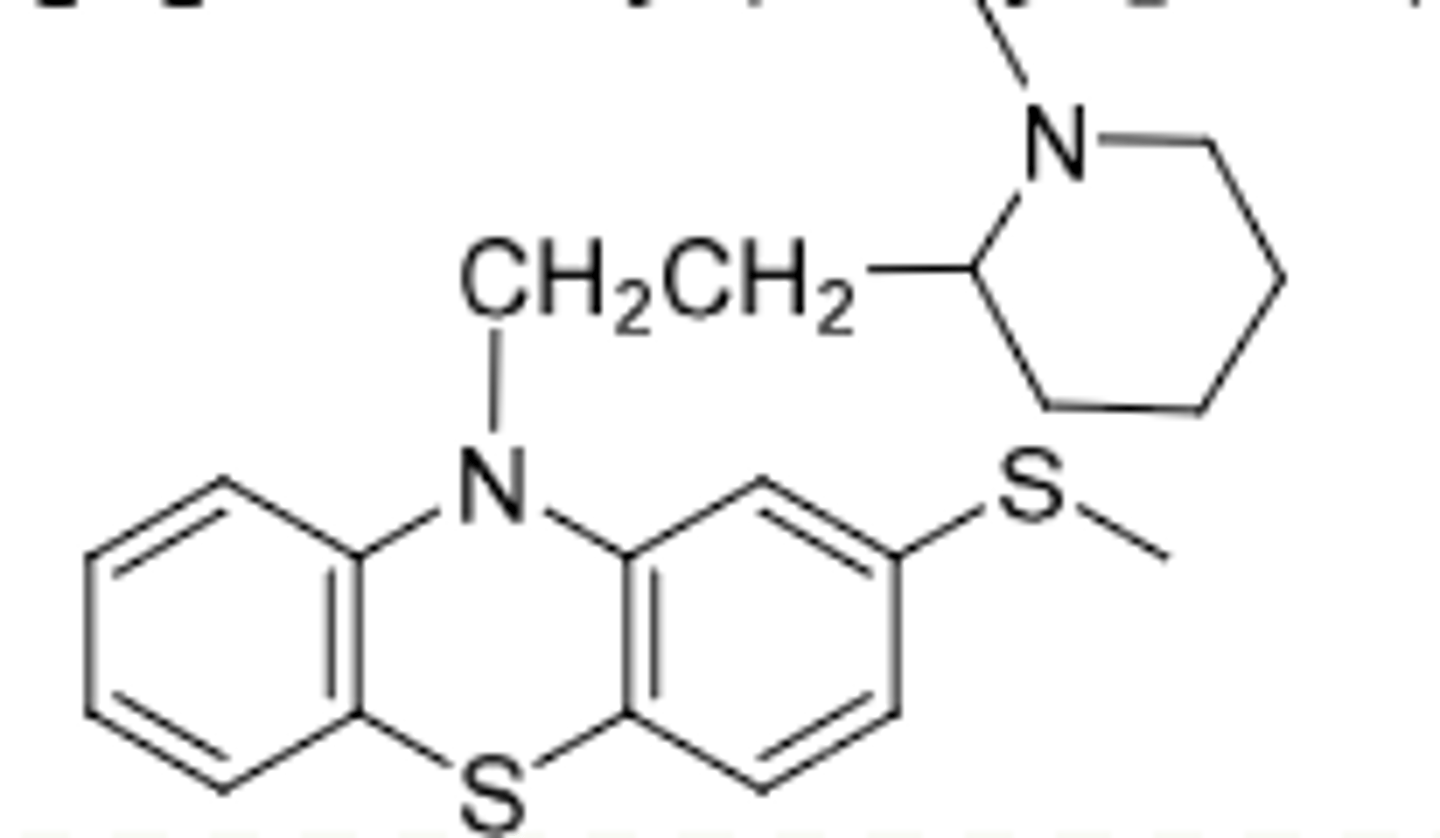
Thioridazine
-Piperidine phenothiazine
-weak antipsychotic (EWG is not strong) but can be oxidized --> metabolites are more potent antipsychotics (they are their own drugs too)
-NOT A PRODRUG THOUGH B/C PARENT IS ACTIVE
-CYP2D6 metabolized
-Sulfoxide (mesoridazine), 8x more potent than parent; still clinically used today
-Sulfone (sulfuridazine) is 2x more potent thamn parent; withdrawn due to cardiac side effects
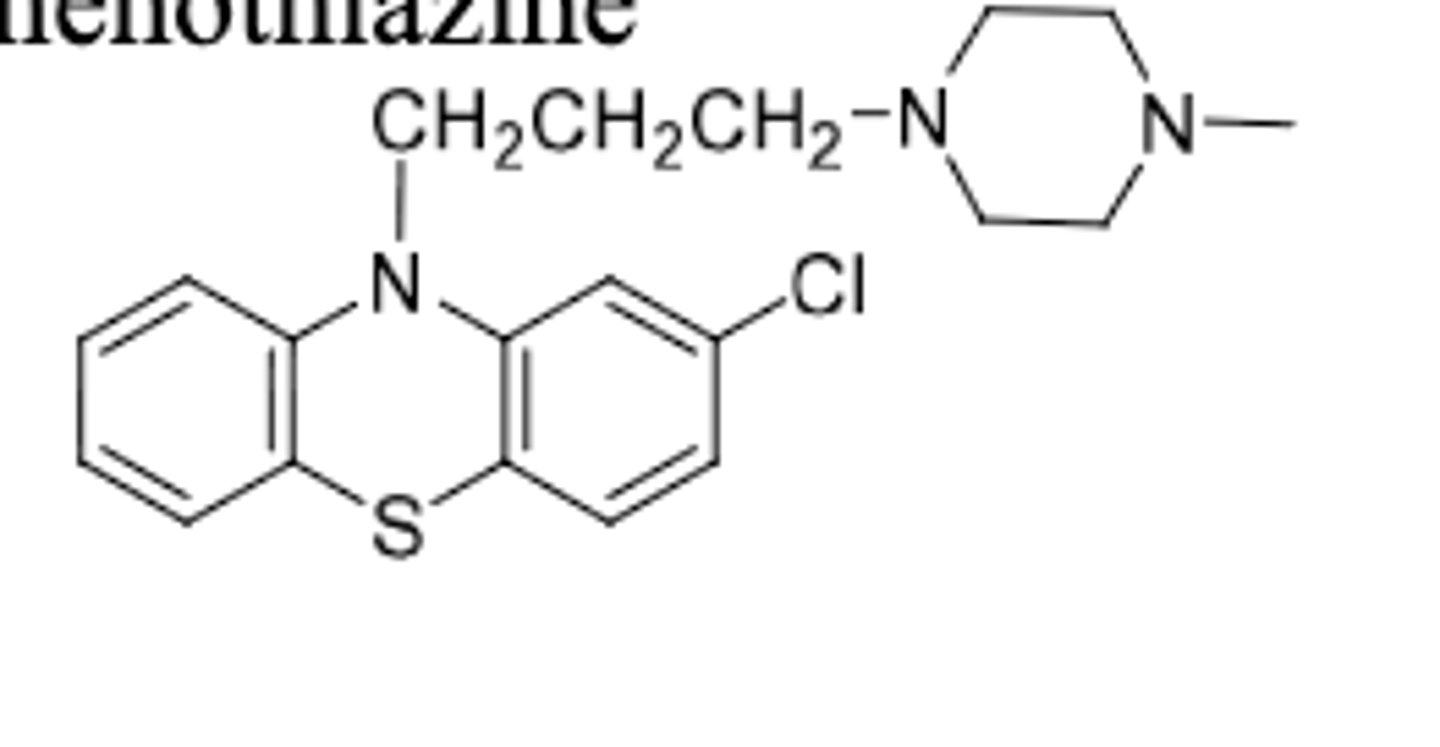
Trifluoperazine
-piperazine phenothiazine (more potent than diethyl amine side chain due to more CNS activity)
-used for antipsychotic
-used for schizophrenia and short term treatment of non psychotic anxiety
Prochlorperazine
-piperazine phenothiazine
-antipsychotic and antiemetic
-also has N/V use (unlike trifluoperazine)
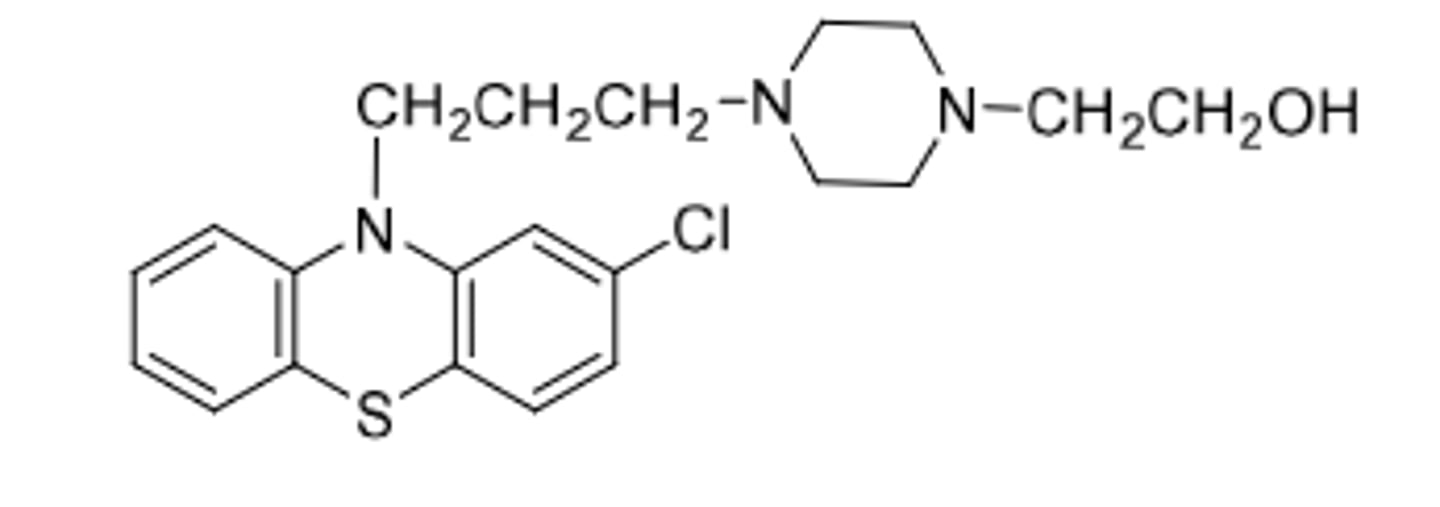
Perphenazine
-piperazineethanol phenothiazine
-antipsychotic
-can be given IV to control N/V
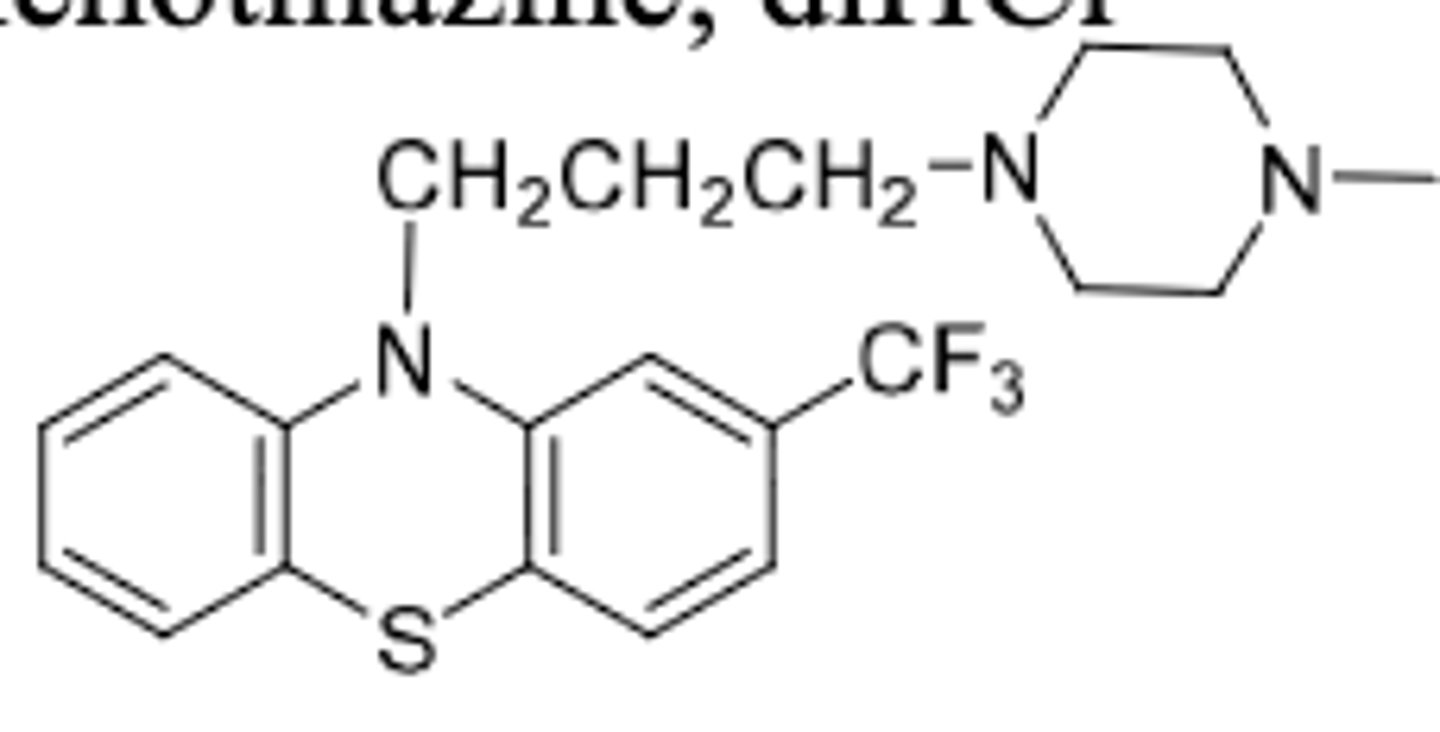
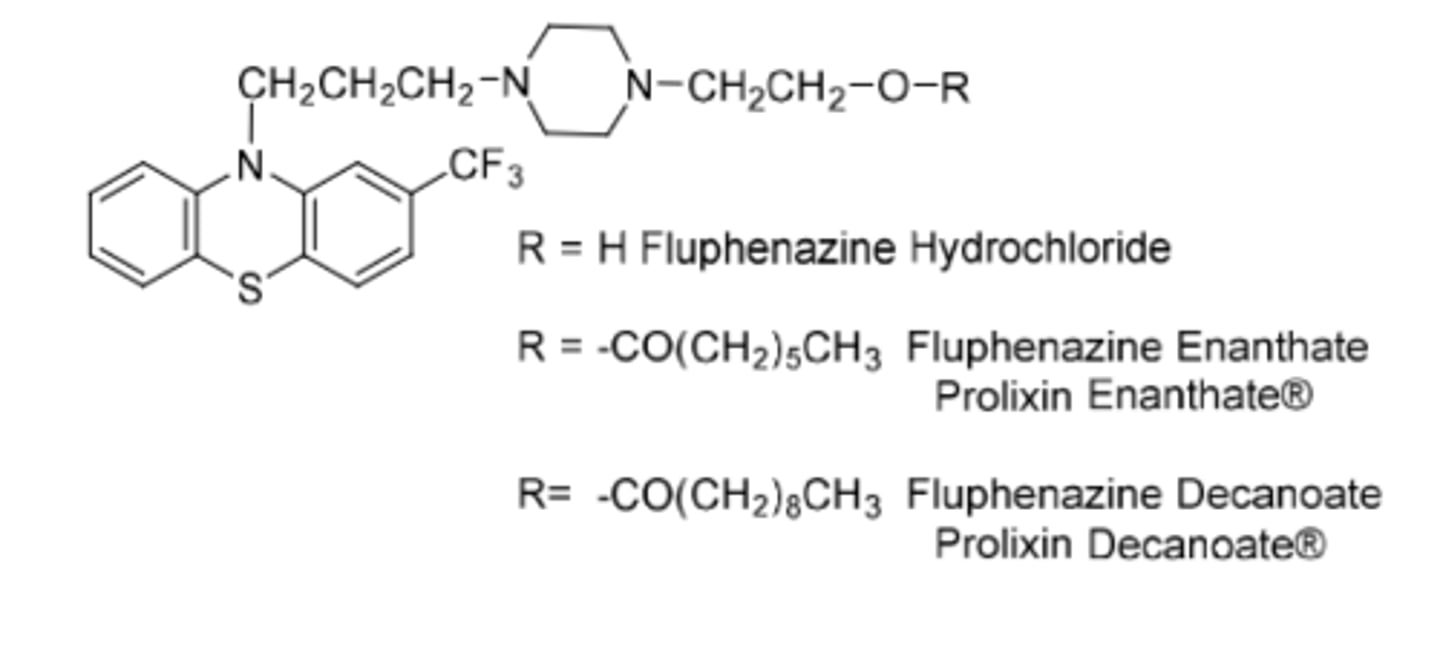
Fluphenazine
-piperazineethanol phenothiazine
-to treat schizophrenia
Original: (R=H)
-needs to be adminsitered 3-4x per day
-t1/2 = 33h
Enenthate:
-R=7 carbon fatty acid
-once every 2 weeks
Decanoate:
-R=10 carbon fatty acid
-once every 3-4 weeks
Prodrug forms: Suspend in oil, inject into patients muscles, drug slowly releases from injection site into blood --> fatty acid is attached to hydroxy by ester functional group --> esterases in blood release fluphenazine over time
-t1/2 = 163-132h
-used for chronic treatment of schizophrenia
-long acting parenteral that solved the initial adherence problem
Thiothixene
-isosteric phenothiazine
-double bond is bioisosteric replacement
-work on D2 receptors
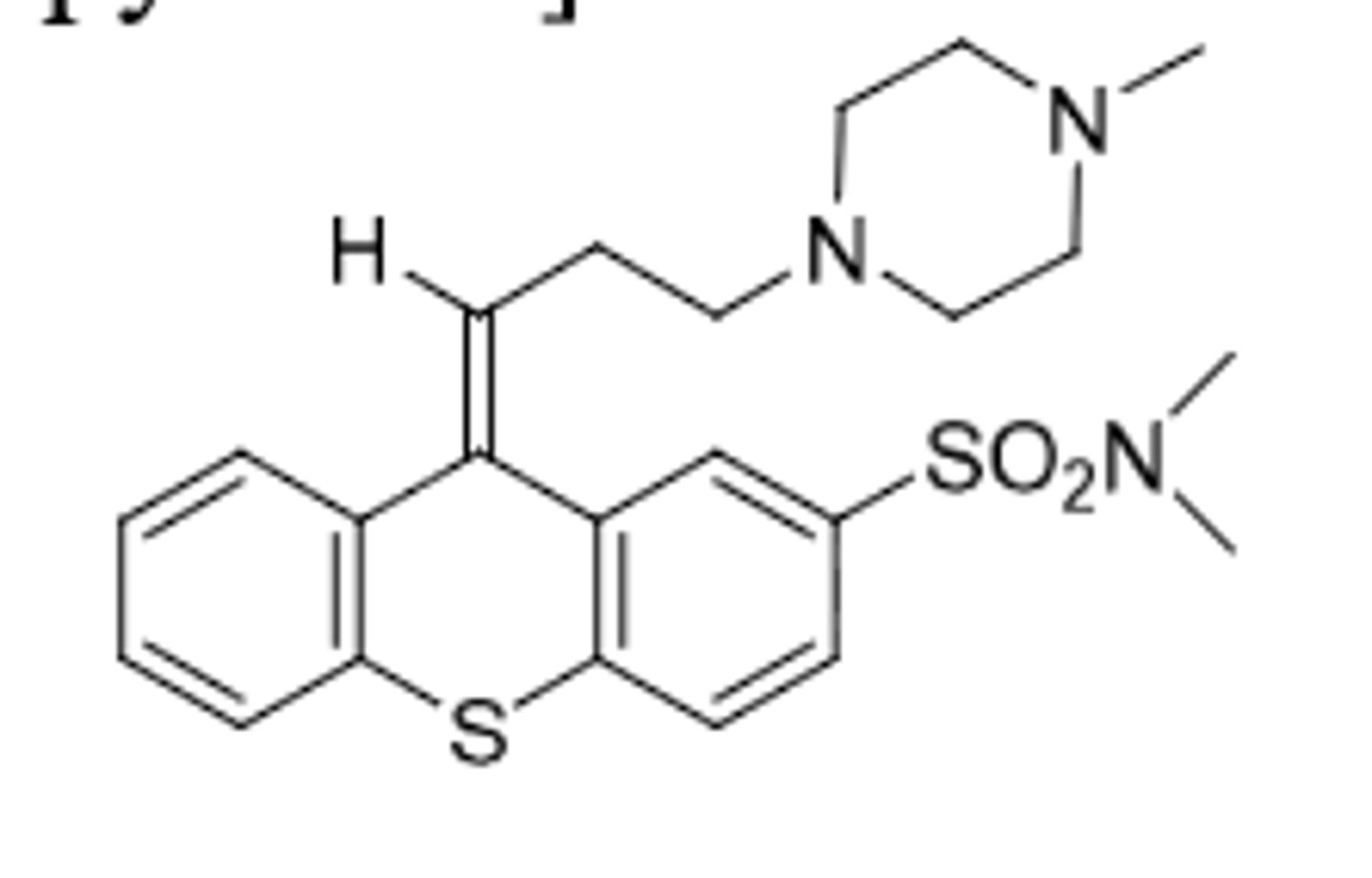
Molindone
-indole antipsychotic for management of schizophrena
-unknown MOA
-clinical profile similar to piperazine phenothiazines
-very extensive metabolism
-Are in general more potent than the phenothiazines and isosteric phenothiazines
-Cause greater CNS side-effects but less peripheral anticholinergic side-effects than the phenothiazines
Mimicking dopamine to block D2 for antipsychotic and antiemetic effects
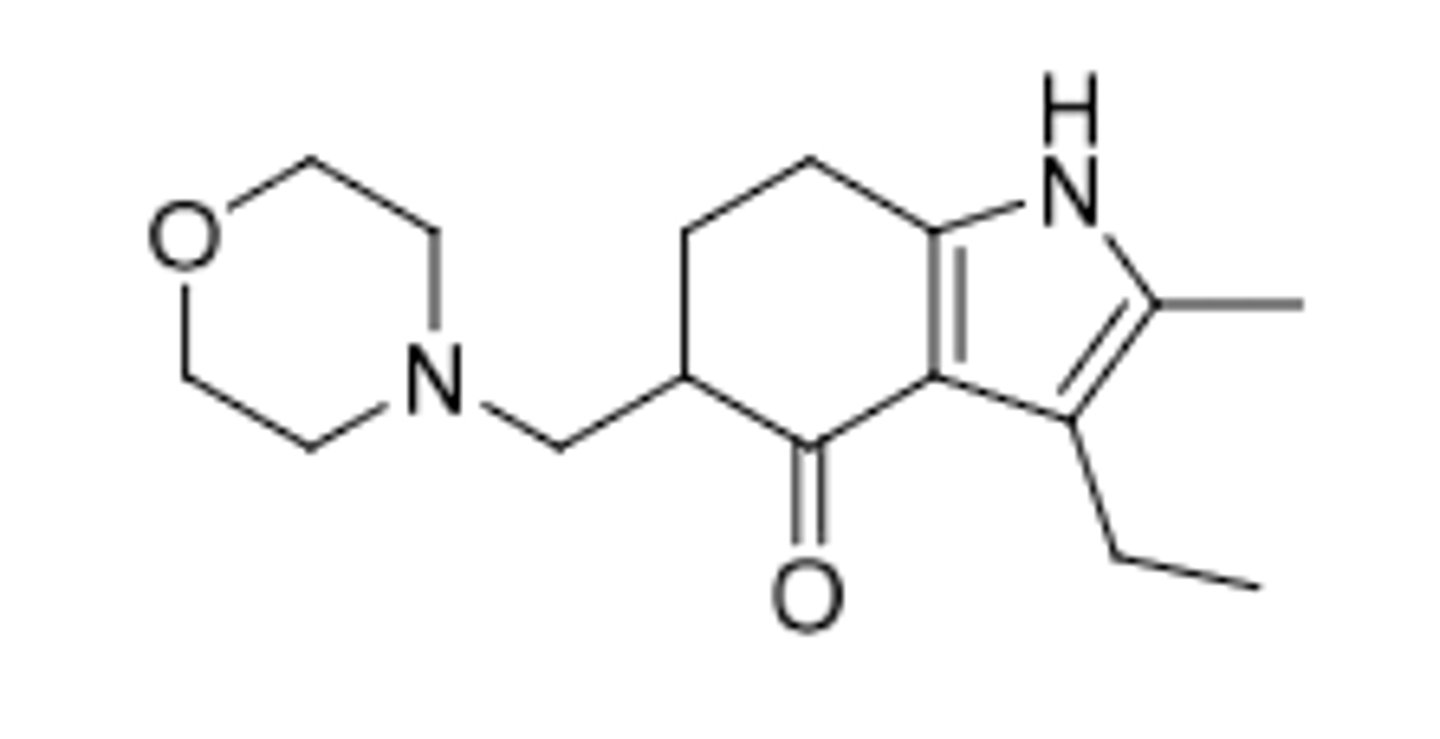
Butyrophenones
Haloperidol
-butyrophenone
-D2 inverse agonist
-antipsychotic used for schizophrenia and tourettes syndrome
-Attaching 10 carbon fatty acid (decanoate) to OH creates prodrug --> dissolve in sesame oil, IM injection given monthly (drug slowly released from injection site by estereases to remove fatty acid in blood and produce haloperidol)
metabolism:
-OH conjugation (50-60%)
-dealkylation (20-30%)
-reduction of ketone to alcohol (23%)
Droperidol
-butyrophenone
-Potent D2 antagonist
-half life only 2 hours --> not used for schizophrenia
-used as antiemetic for N/V in surgical procedures or diagnostic procedures or chemotherapy
-injection only
Metabolism: reduction, double bond, hydroxylation
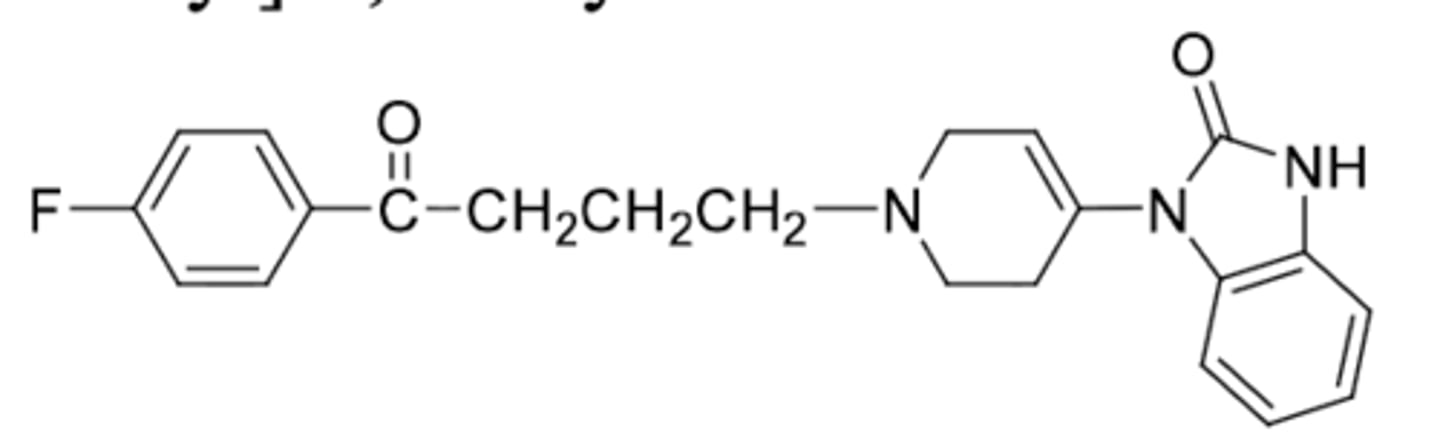
Pimozide
-butyrophenone (with diphenyl methyl instead of phenyl ketone)
-used for suppression of ticks in tourettes disorder
-Lithium ion can be absorbed and distributed into CNS
-works by inhibiting NE reuptake and increasing serotonin receptor sensitivity
-Positively charged cation eliminated in urine (similar to sodium ions) --> risk of sodium ion depletion because it inhibits reabsorption of sodium ions
-treatment of manic episodes in bipolar disorder
Lithium Carbonate, Lithium Citrate
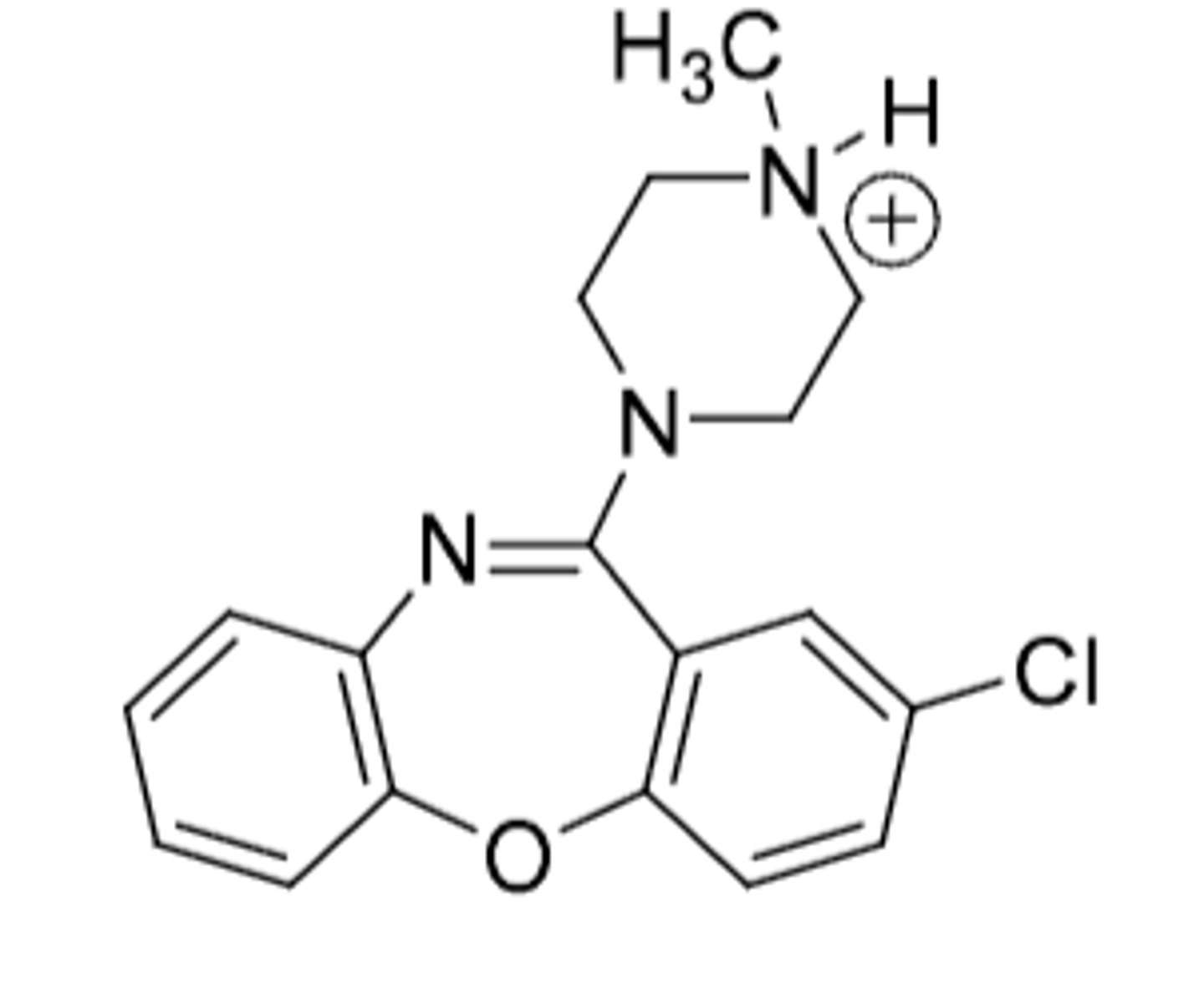
Loxapine
-used for schizophrenia (unknown MOA of how)
-demethylated on quaternary N to form TCA amoxapine
First generation antipsychotics only treat positive aspect of schizophrena (hallucinations)
First generation antipsychotics have extrapyrimidal symptoms (EPS) side effects and movement disorders such as TD (tardine dyskinesia)
The negative aspects of schizophrenia, such as withdrawal from society and depression are NOT treated with 1st gen drugs
All second generation antipsychotics have 5HT1 agonist activity to help with the treatment of the negative aspects of schizophrenia and help reduce the EPS side effects
First-gen vs. Second-gen antipsychotics
Clozapine was the first available drug to treat both positive and negative aspects of schizophrenia, but the significant risk for agrunulocytosis (drop in WBC, can be fatal)
Lower risk of EPS of TD symptoms
Risperidone (Risperidal)
-second generation antipsychotic
-oral solution or disintegrating tablet
-t1/2=3-14h (taken multiple times per day)
-microencapsulated polymeric formulation (encapsulated in PLG copolymer) --> Risperidal consta - extends half life of risperidone by crosslinked ester bonds to be slowly hydrolyzed and released over time
Hydroxy metabolite of risperidone (OH on rightmost ring) is active --> Paliperidone (Invega)
-t1/2 = 23-30h
-hard shell, nonabsorbable tablet for once daily dosing
Invega hydroxy group can be metabolized (acetylation)
-Palmitate --> 16 Carbon fatty acid --> prodrug form
-Palmitate forms nanocrystals to suspend in aqueous solutions
-increase size of nanocrystal = more drug in injection = less frequent dosing
1. Invega sustena = once per month
2. Invega trinza = once every 3 months
3. Invega halfyera = once every 6 months
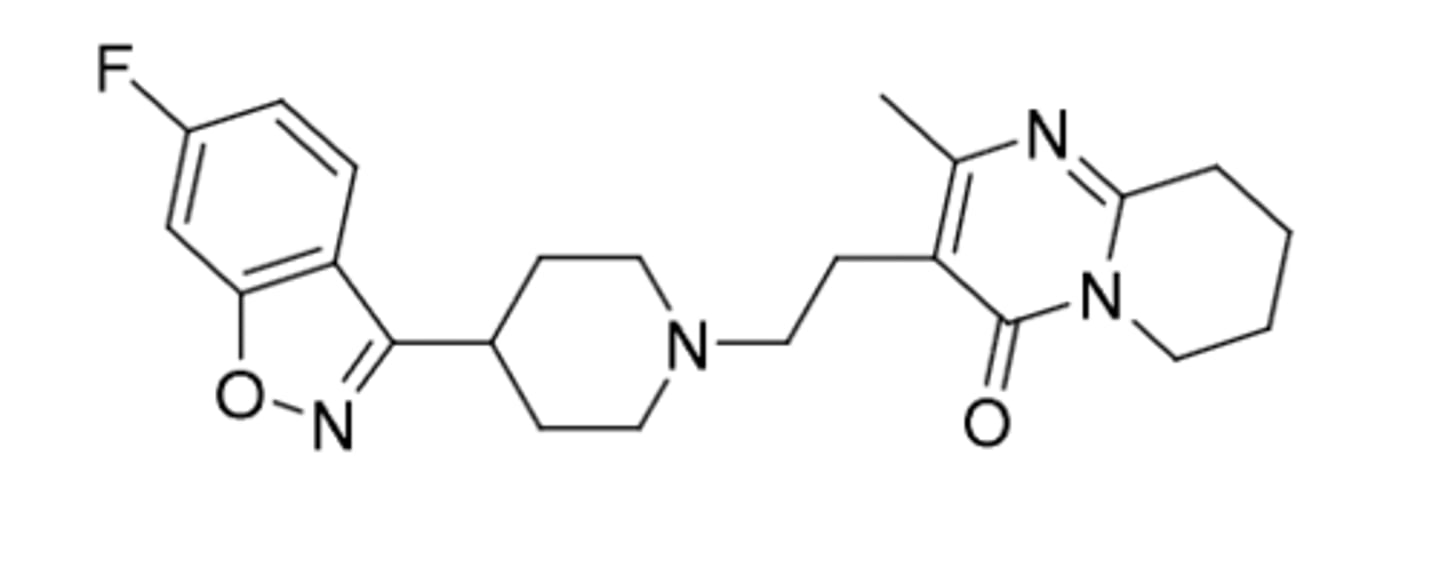
Iloperidone
-second generation antipsychotic
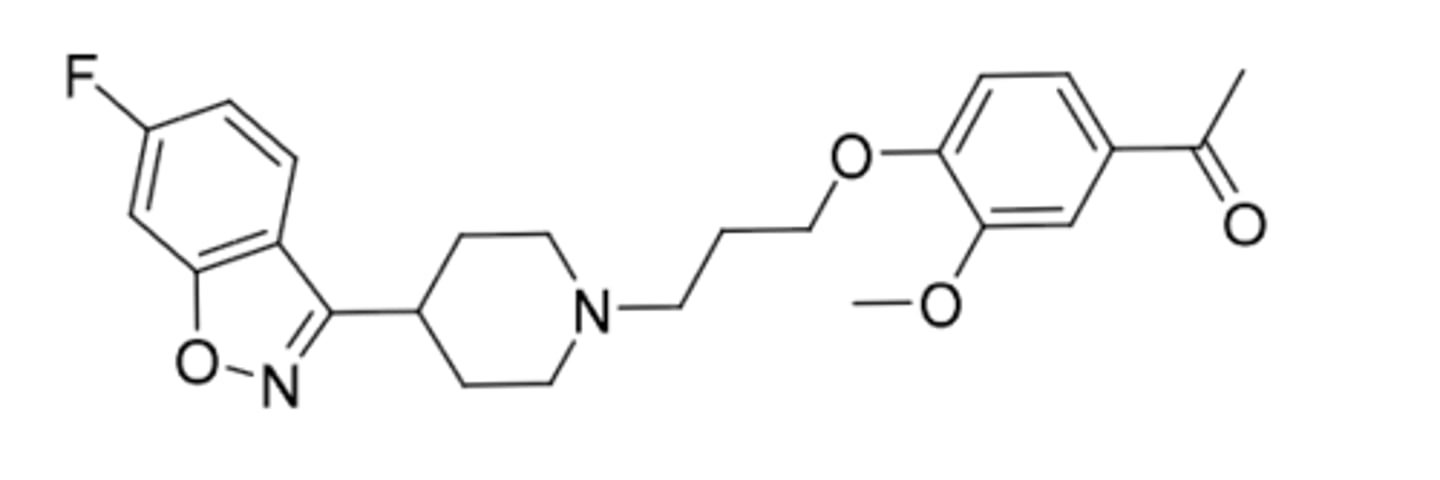
Ziprasidone
-second generatioon antipsychotic
-benzoisothiazole bioisosteric replacement for benzoisoxazole in risperidone
-blocking D2, 5HT2 antagonist, 5HTA1 agonism
-food effect(can increase absorption when taken with food)
-indicated for schizophrenia and bipolar disorder
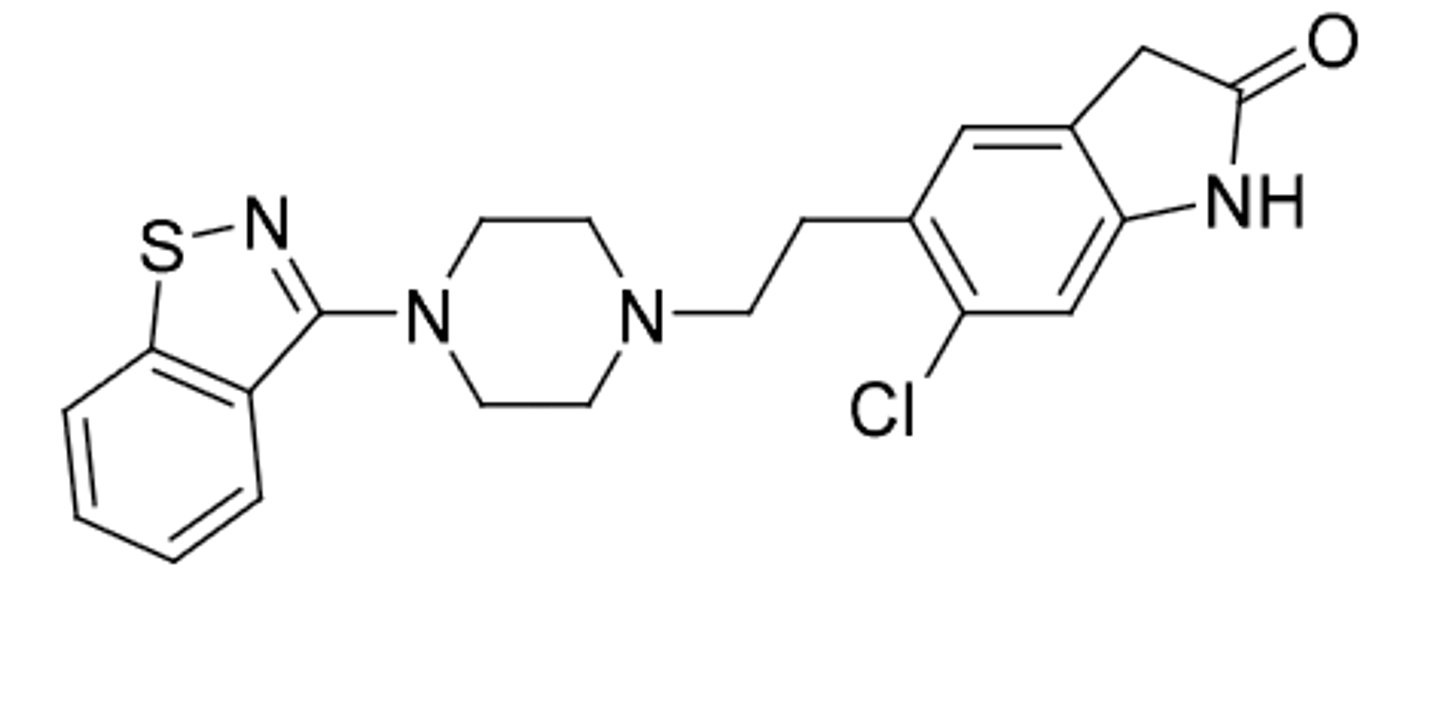
Lurasidone (same as ziprasidone)
-second generatioon antipsychotic
-benzoisothiazole bioisosteric replacement for benzoisoxazole in risperidone
-blocking D2, 5HT2 antagonist, 5HTA1 agonism
-food effect(can increase absorption when taken with food)
-indicated for schizophrenia and bipolar disorder
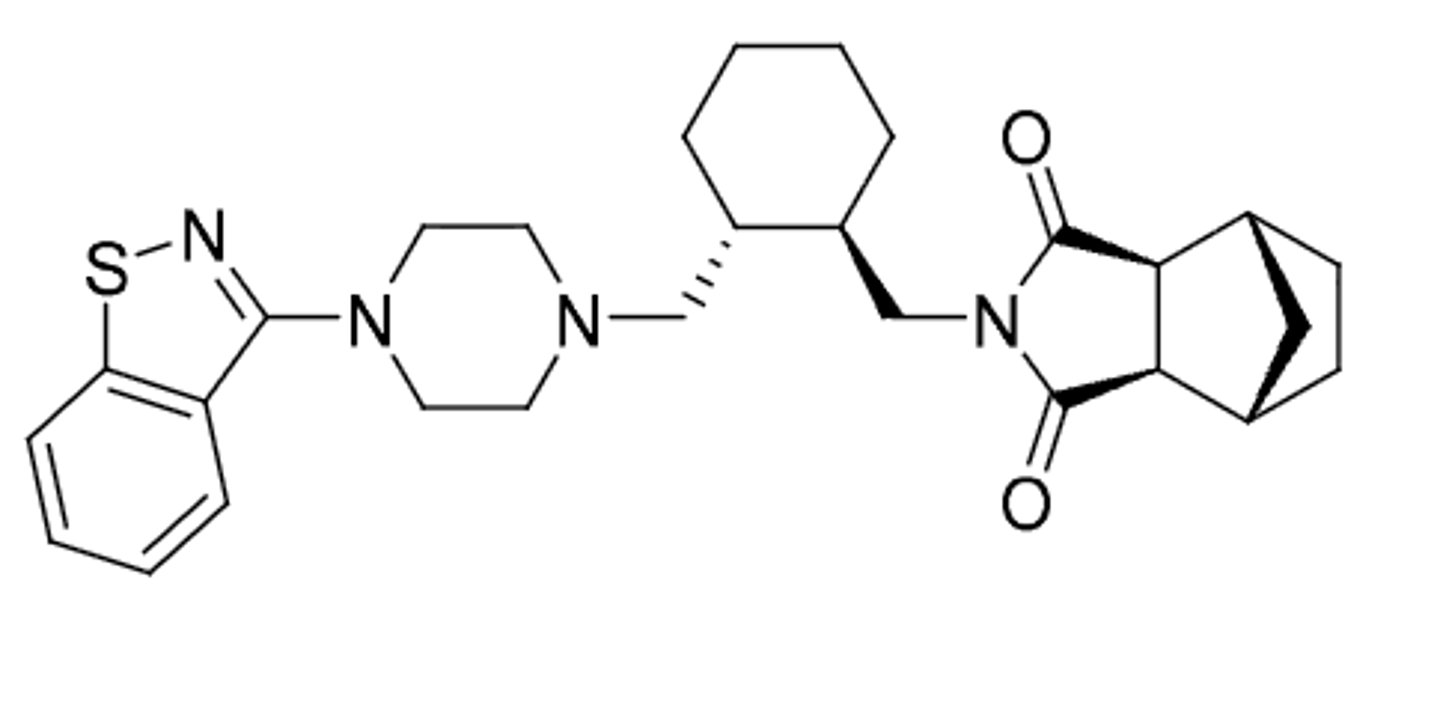
Olanzapine
-treats + and - aspects of schizophrenia (second gen antipsychotic)
-similar structure to clozapine (bioisosteric replacement of phenyl ring on right) but NO agranulcytosis
-used for bipolar disorder
-fewer Parkinson-like symptoms
-produces very little sedation
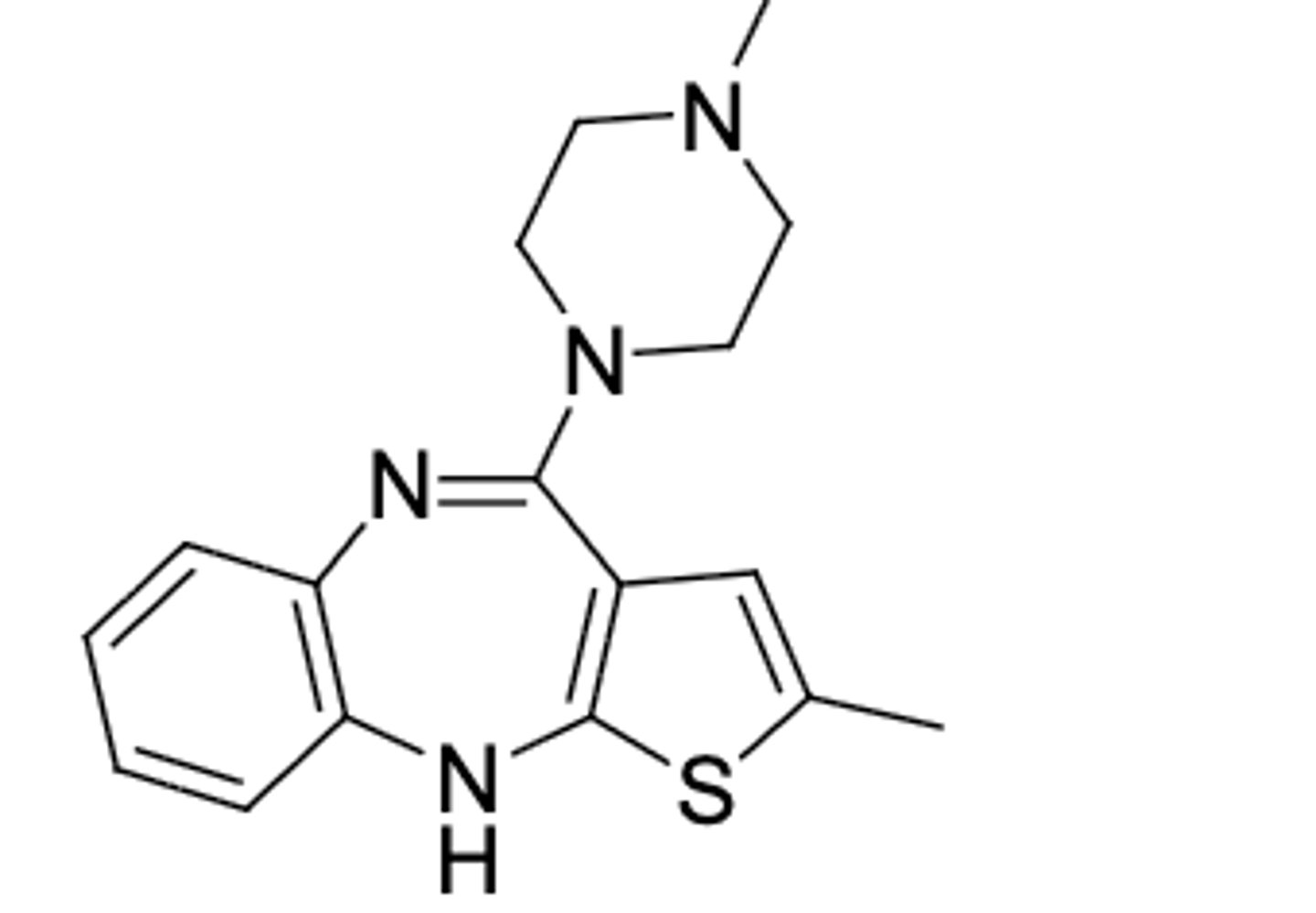
Quetiapine
-second gen antipsychotic (dopamine and serotonergic activity)
-used for management of psychotic disorders including schizophrenia
-can be oxidized at S or OH leading to inactivation
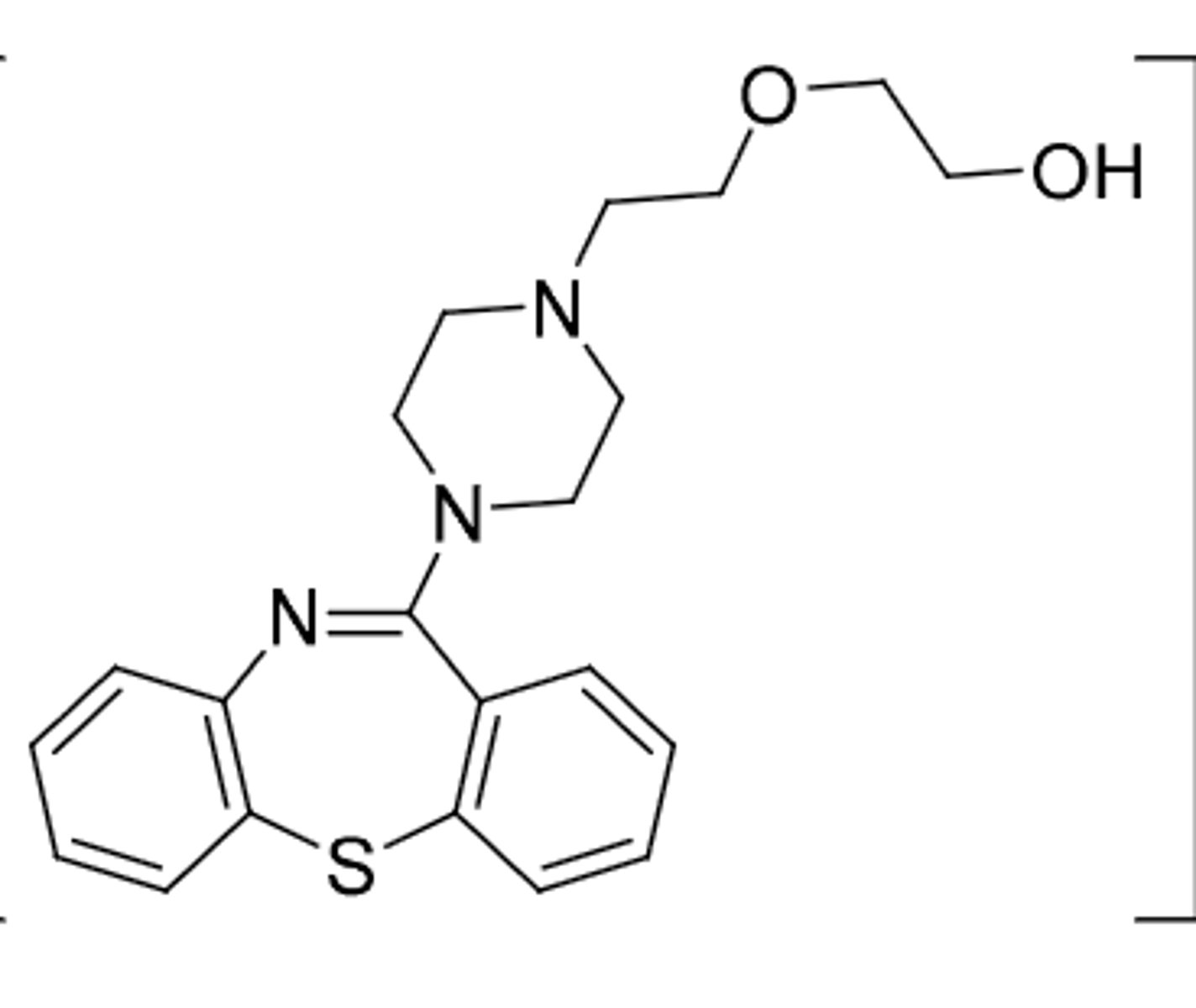
Apriprazole (Abilify)
-2nd gen antipsychotic
-partial agonist at D2, partial agonist at 5HT1A, antagonist at 5HT2A
-used to treat schizophrenia, bipolar disorder, and tourettes (2nd gen antipsychotic)
-requires 1-2 weeks of administration before effect is seen
-t1/2 = 75h
Metabolism:
1. inative dealkylation
2. ACTIVE - can be oxidized to remove 2H on rightmost ring to form active didehydro metabolite (t1/2 = 94h)
3. hydroxylation
Prodrug form: Apriprprazole Lauroxil (Aristada)
-ester + 12C fatty acid linker
-long duration of action, lipid soluble prodrug in sterile emulsion
-injected into patient every 4-6 weeks
Apriprazole mantena
-ER formulation of parent drug molecule in micron particle size in aqeuous solution for monthly IM injection
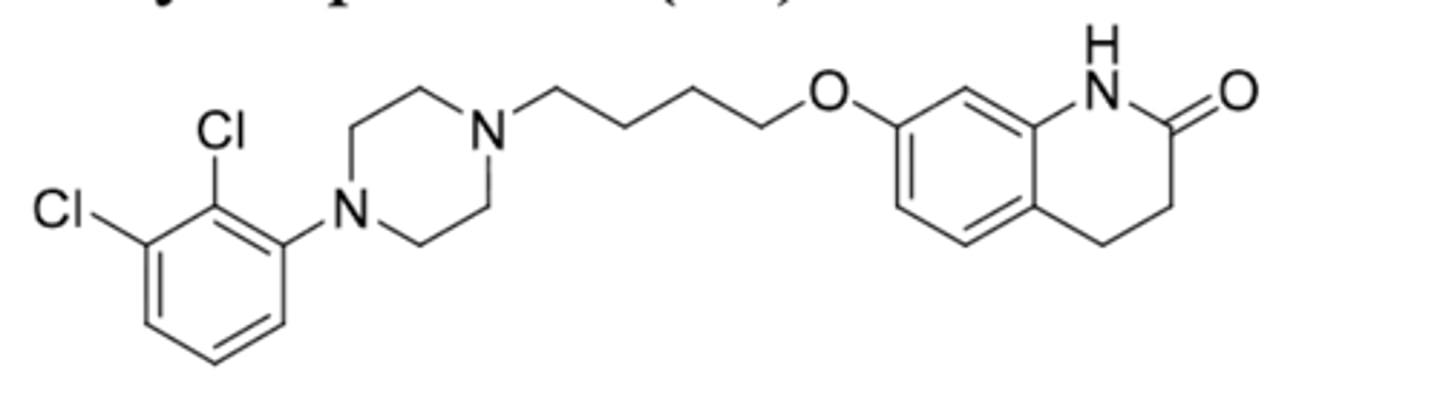
Brexpiprazole
-2nd gen antipsychotic
-very similar to apriprazole
-partial agonism at D2, partial agonism at 5HT1A, and antagonism at 5HT2A
-used for MDD, schizophrenia, and agitation in Alzheimers
-t1/2 = 91h, F=95%
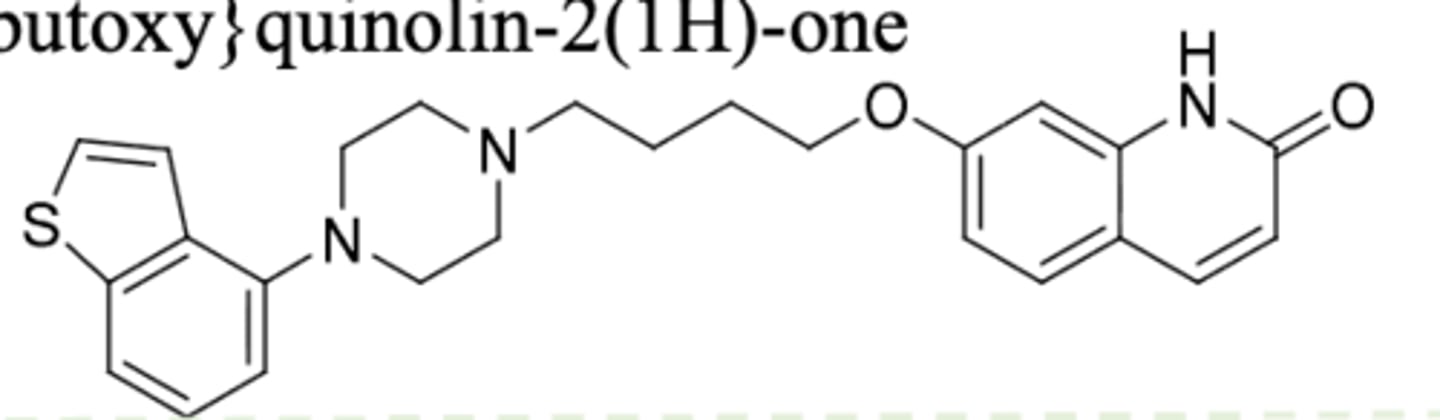
Cariprazine (Vraylar)
-2nd gen antipsychotic
-partial agonist at D2 and 5HT1A, antagonist at 5HT2A, AND 5HT2B antagonist activity
-demethylation on tertiary N to give active metabolites
-t1/2 = 2-4 days
= approved for schizophrenia, bipolar disorder, and MDD
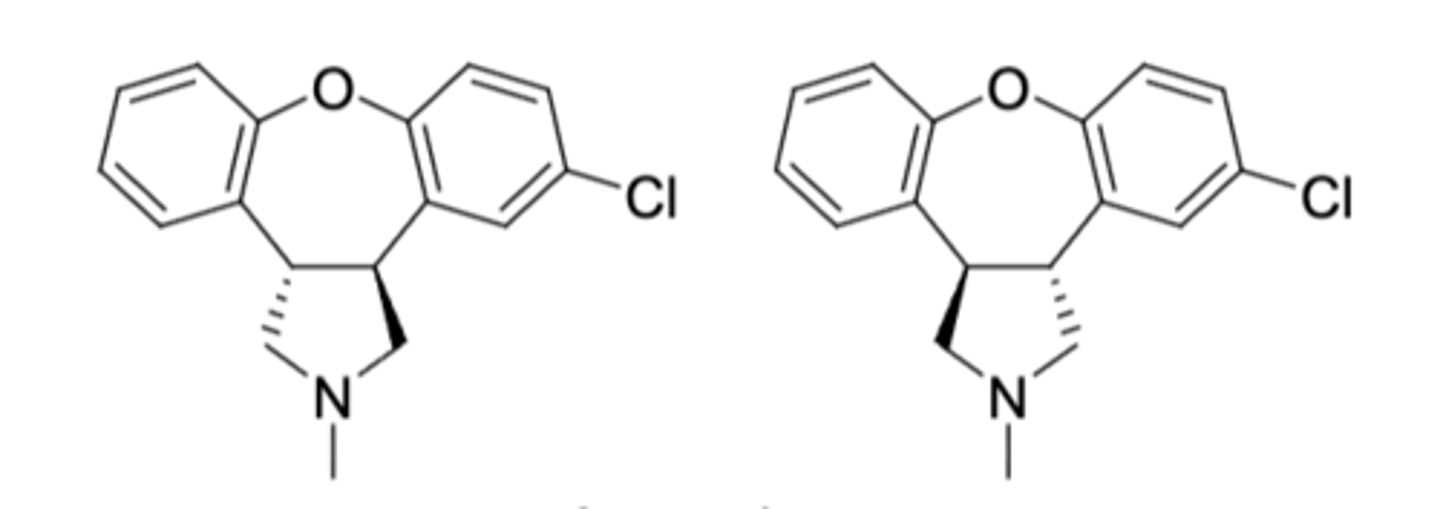
Asenapine (Saphris)
-2nd gen antipsychotic with dopaminergic and serotonergic activity
-mixture of 2 enantiomers for reatment of acute manic and mixed episodes associaed with bipolar disorder and schizophrenia
-sublingual tablet (nota bsorbed through GI tract --> avoids first pass metabolism)
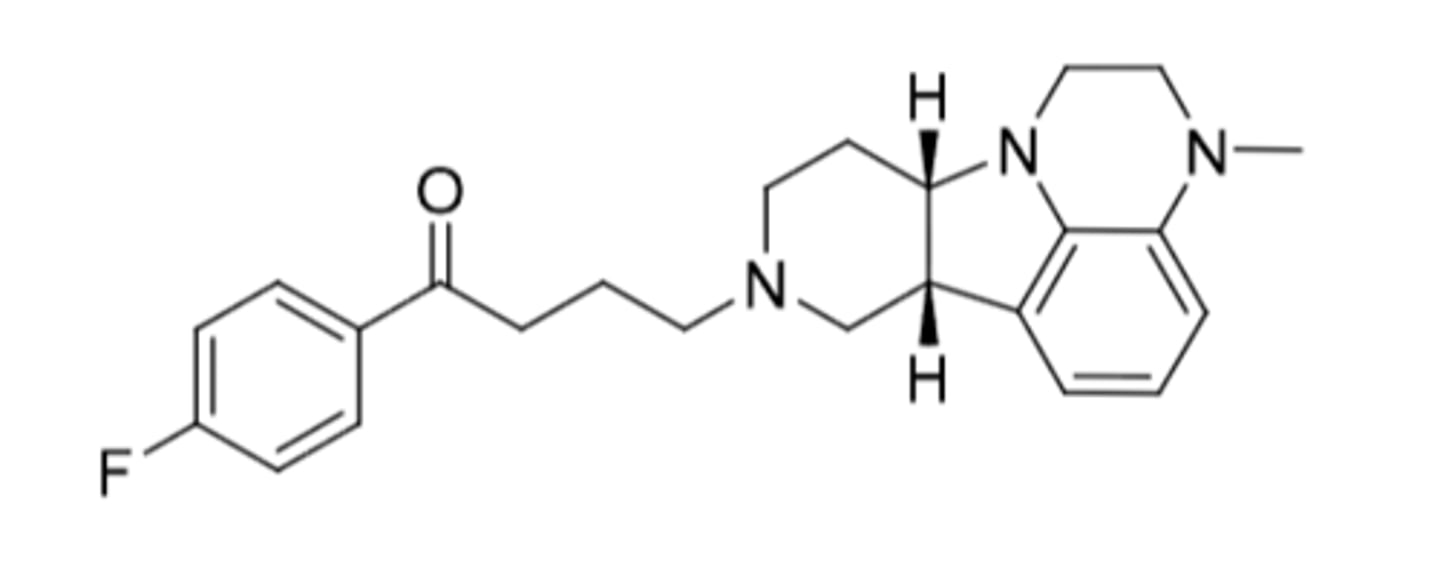
Lumateperone
-2nd gen butyrphenone (added serotonergic activity)
-metabolized by CYP3A4 to 20+ different metabolites
-CI: cannot drink grapfruit juice (CYP3A4 inhibition --> extends half life)
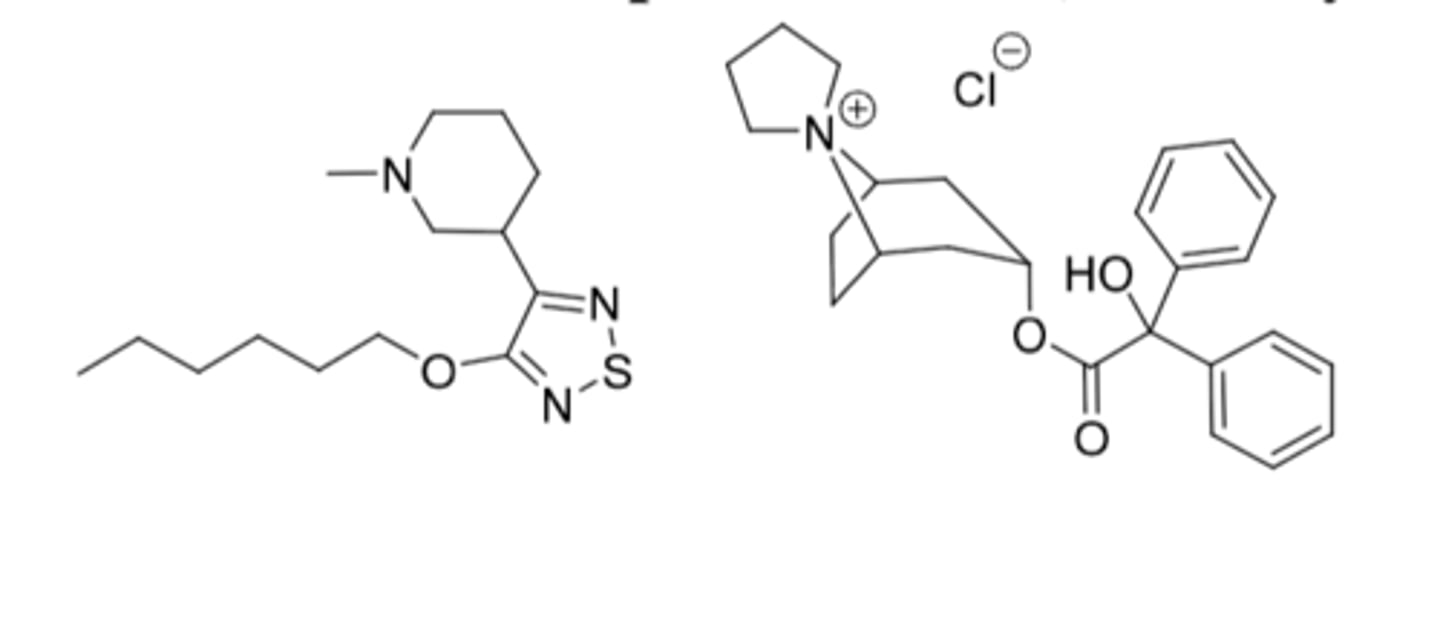
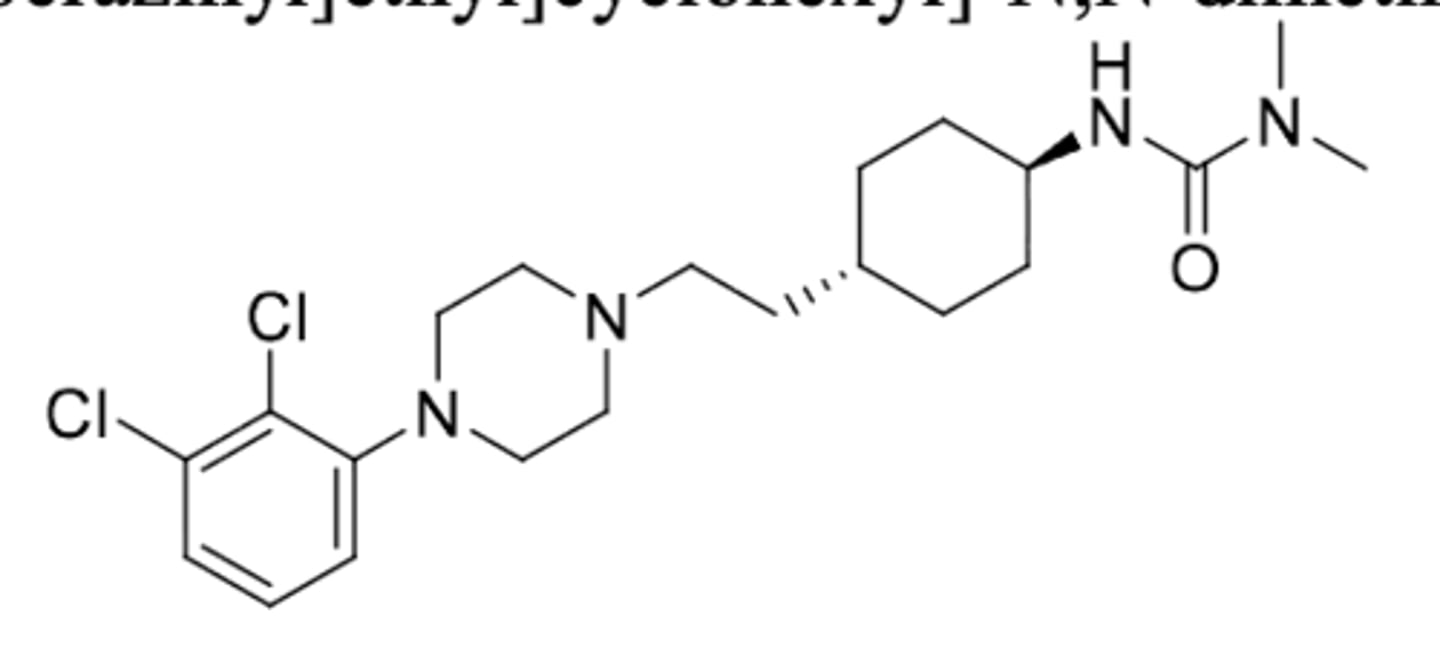
Xanomeline and trospium chloride, Cobenfy®
-Approved for schizophrenia in adults
Xanomeline
-Muscarinic receptor agonist at M1 and M4 receptors
-6C alkyl chain to cross BBB
-M1: improve cognitive function and negative aspects of schizophrenia
-M4: decrease release of dopamine and improve positive aspect of schizophrenia
-side effects: GI (N/V, indigestion, constipation) and CV (increase BP and HR)
Trospium
-Muscarinic receptor antagonist
-Doesn't get into CNS because it has a permanent positive charge
-counters periphery effects of xanomeline because trospium only stays in periphery