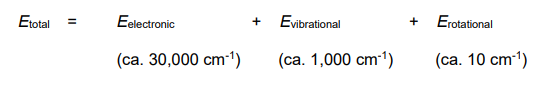Electronic Spectroscopy
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
What is electronic spectroscopy
Study of how EM radiation interacts with a molecules to change its electronic energy
When observing in the UV-vis region we look at wavelength rather than wavenumber or frequency
Given the three properties of electrons key for spectroscopy
Quantized , at discrete energy levels
Widely Spaced, large differences in energy
Give electronic transitions which are in the UV visible region
What is a chromophore
A molecule or group that absorbs in the UV-visible region
If one colour is absorbed the complementary colour will be observed
Give the Beer-Lambert Law
A = ε c l
Absorbance , A, no units
Concentration, c, mol dm-³
Pathlength, l, cm
Molar absorption coefficient, ε, dm3 mol-1 cm-1
For specific molecules at specific wave length
Give Absorbance equation using only pathlengths
A = log10 (l0/l)
What is an electronic transition
An electron moves from one molecular orbital to another
What is gross selection rule for electronic transitions
Gross selection rule
for absorption or emission, there must be a change in dipole as the electron moves and a large dipole change will give a strong band
What is specific selection rule for electronic transitions
Specific molecule determines the specific MOs involved
specific energy gaps i.e. absorption λs
specific band strengths i.e. ε values
Give typical lambda max values for different electronic transitions
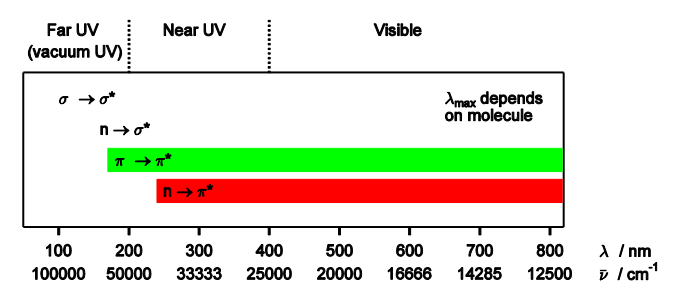
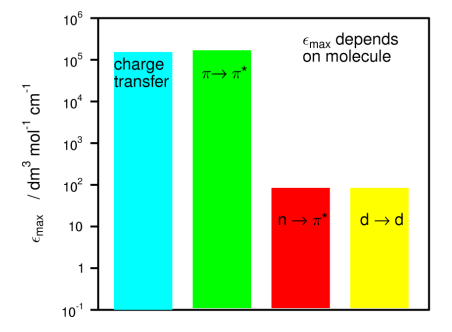
Give two rare transitions we may also see
Charge transfer transitions (donor -> acceptor groups)
d-> d transitions (Transition metal groups)
Give typical range of εmax values
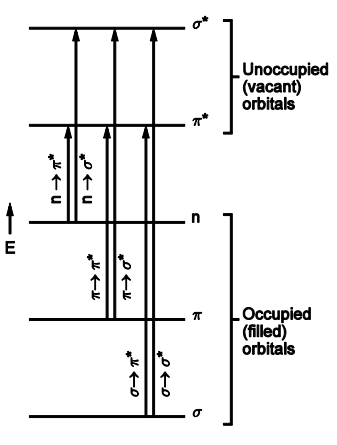
Why do we see many transitions within one molecule
Several different occupied MOs
Several different unoccupied MOs
As such we can see several different transitions as there are different MOs that can transition into each other
We will also see several different bands in the spectrum as they correspond to the electrons jumping between the several different MOs
draw energy level diagram for the typical transitions
Effect of conjugation on wavelength
As conjugation increases π → π* absorption band shifts to longer wavelength leading to a lower energy state
What can we use electronic spec to measure
Concentrations
Chromophores
What is an Isosbestic point
Constant absorbance at a specific λ as two species interconvert
occurs when two interconverting species have the same ε at a specific λ
What is the Born-Oppenheimer approximation
Electronic,Vibrational and Rotational energies are very different. Each can be considered independent of the others
They can also all transition simultaneously