L25 - Metabolism in Obesity and Aging
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
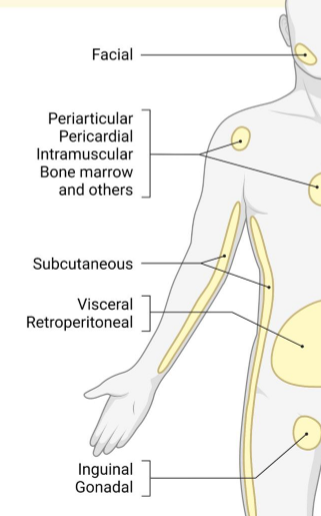
what is white adipose tissue
tissue where lipid is stored - this is facial, bone marrow, subcutaneous, visceral, gonadal, and some white adipose tissue depots are more highly associated with increased risk for metabolic disease
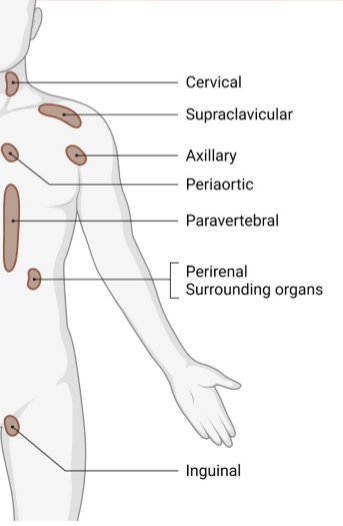
what is brown adipose tissue
tissue that has high thermogenic potential, so it has the ability to break down fat and glucose as heat instead of storing energy - this maintains body temperature, and consists of cervical, axillary, inguinal, and supraclavicular tissue
what is thermogenesis
dissipation of energy through the production of heat by uncoupling the proton gradient
what is AMPK
adenosine monophosphate-dependent kinase which is a highly conserved fuel sensor and energy gauge that regulates cellular activity based on the balance of ATP, ADP, and AMP
when is AMPK active
when the cell has a low energy charge, and it is activated by an increase in AMP and ADP levels
what are the allosteric activators of AMPK
AMP acts as an allosteric activator, increasing AMPK activity by up to 2,000 fold
what are the inhibitors of AMPK
high levels of ATP
what are the physiological triggers of AMPK
energy starvation, cellular stress, exercise, fasting, or cold exposure
what catabolic pathways are regulated by AMPK
catabolism gets turned on to promote pathways that generate ATP - beta-oxidation (fatty acid breakdown), glucose uptake via GLUT1 and GLUT4, glycolysis, and mitochondrial biogenesis
what anabolic pathways are regulated by AMPK
anabolism is turned off by AMPK to inhibit pathways that consume ATP to promote energy storage - fatty acid synthesis by phosphorylating and inactivating ACC, lipogenesis, gluconeogenesis glucose production in the liver, and glycogen synthesis
what is insulin resistance
a clinical sign of metabolic disease characterized by a cell’s failure to respond properly to insulin
how does insulin resistance reduce cellular response
in this state, cells (adipocytes) show lower glucose uptake and lower lipogenesis
how does insulin resistance impair glucose transport
it is specifically associated with impaired GLUT4 translocation, meaning the transporters that move glucose into the cell do no function correctly
how does insulin-resistance affect the physical cells
insulin-resistant adipocytes are typically larger (hypertrophy) with disorganized cortical actin and unilocular-like lipid droplets
what are the systemic effects of insulin resistance
along with inflammation, it promotes low levels of lipolysis, leading to free fatty acids being stored in other organs, which causes lopotoxicity
how does insulin resistance affect development
it often develops during obesity due to extended overnutrition and a consistently high energy charge in the body
how can metformin work to counteract insulin resistance and improve metabolic health
Metformin can act as the first-line treatment drug for Type II diabetes that works primarily by stimulating AMPK activity by inhibiting Complex I in the mitochondria - this reduces ATP production and increases AMP/ATP ratio which directly activates AMPK
how does metformin affect the liver vs muscles
in the liver, it reduces the release of glucose and inhibits gluconeogenesis while in the muscles, it improves glucose uptake
what are the clinical results of metformin on diabetes
this treatment reduces blood glucose levels, improves insulin sensitivity, and does not cause weight gain
how does AMPK activation work to counteract insulin resistance and metabolic health
by acting as a cellular fuel gauge, and getting activated when the cell’s energy charge is low - when activated, AMPK shifts the cell’s metabolism from storing energy to generating it
how does AMPK activation increase glucose uptake
it stimulates GLUT4 and GLUT1 transporters to bring more glucose into the cell
how does AMPK activation promote fat burning
it inhibits ACC which reduces fatty acid synthesis and increases beta-oxidation, which is fat burning
how does AMPK activation increase browning of adipocytes
browning is where white fat storage cells take on characteristics of brown fat cells (heat production), increasing mitochondrial activity and thermogenesis
what is cellular aging
the time-dependent accumulation of biochemical and molecular “wear and tear” that eventually leads to a loss of cell function
what are the sources of cellular damage in cellular aging
aging begins with the accumulation of damage from endogenous (internal) and exogenous (external locations)
what are the endogenous sources of cellular damage
normal metabolic by-products like ROS, errors in DNA replication, and hydrolysis
what are the exogenous sources of cellular damage
environmental factors like UV light, ionizing radiation, and chemicals like cigarette smoke
what are the molecular consequences of cellular aging
DNA mutations through the oxidation of guanine to 8-oxoguanine (if not repaired, leads to mispairing G:C becoming T:A during replication), mitochondrial dysfunction which leads to further ROS accumulation and a damaging cycle, loss of proteostasis (misfolded protein accumulation), and epigenetic alterations
what is cellular senescence
when damage becomes too extensive for repair mechanisms like base excision repair to handle - consists of cell cycle arrest which stops dividing to prevent passing on the mutations, apoptosis resistance where these “zombie cells” don’t die but persist and accumulate in tissues, and also systemic pathology where the accumulation of these cells leads to functional decline, tissue atrophy, inflammation, and metabolic diseases
what are the therapeutic strategies for cellular aging
stimulating repair by returning metabolism to normal by enhancing the cell’s natural repair pathways, and by senolytics which is a new class of drugs designed to kill senescent cells by turning on their apoptotic pathways, clearing them from the body
what is the 8-oxoguanine repair
OGG1 is a key example of a repair enzyme, and if it is missing (like in knockout mice), the mouse becomes heavier and develops hyperinsulinemia, showing a direct link between failed DNA repair and metabolic disease
what causes the DNA damage of oxidation of guanine to 8-oxoguanine
reactive oxygen species
how does the body repair oxidation of guanine to 8-oxoguanine
via base excision repair to fix the specific lesion by recognizing it with the enzyme 8-Oxoguanine Glycosylase and cleaving it from the sugar-phosphate backbone - this leaves behind an AP site where other enzymes then nick the DNA strand at this location, and DNA polymerase inserts the correct guanine base - DNA ligase then seals the backbone up
why is it important that OGG1 repairs 8-oxoG
if failed to repair, the cell might pair it with adenine instead of cytosine during replication, leading to a permanent mutation (G:C to T:A transversion) that can drive aging or metabolic disease
how can DNA damage/repair contribute to cellular aging
via the accumulation of unrepaired lesions that shift the cell from a functional state to a dysfunctional one - aging is the result if the body’s repair systems being outpaced by biochemical wear and tear, eventually leading to a buildup of “zombie” cells that compromise the whole organism
how does induction of cellular senescence lead to cellular aging
chronic, unrepaired DNA damage acts as a primary trigger for the cellular cell to enter senescence which is a state where the cell permanently stops dividing to prevent damaged DNA from being copied (preventing cancer) but refuses to die
how does tissue dysfunction lead to DNA damage and then cellular aging
as these “senescent” cells accumulate over time, they take up space without performing their normal duties which leads to tissue atrophy and a decline in organ function, which are the physical hallmarks of aging