titration 1 : To use a standard solution of anhydrous sodium carbonate to standardise a solution hydrochloric acid
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
theory
By titrating a provided hydrochloric acid (HCl) solution of unknown concentration against a standard solution of sodium carbonate (Na2CO3), the concentration of the hydrochloric acid can be determined.
procedure
1) Filling the burette with hydrochloride acid solution to be standardised
2) Making the anhydrous sodium carbonate up into a standard solution
3) Transferring exact volume of sodium carbonate solution into a conical flask
4) carry out titration
suitable indicator for this titration
methyl orange - is a strong acid-weak base titration, the end point occurs below a pH of 7 , Methyl orange changes colour between pH 3 and 5 coinciding with this end point
colour change observed at end point
yellow to red / pink
titration equation
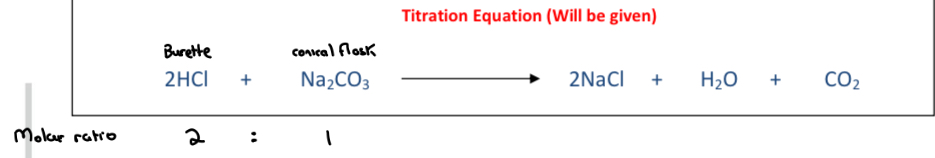
Explain why a standard solution of hydrochloric acid CANNOT be directly made up i.e. why must hydrochloric acid be standardised by titration?
Hydrochloric acid is not a primary standard – it cannot be obtained in pure state and hydrogen chloride is a gas at room temperature
Explain why a standard solution can be directly made up from anhydrous sodium carbonate
• Anhydrous sodium carbonate is a primary standard - it is pure, is stable in air, has a high molar mass and can dissolve easily in water to directly make up a solution of accurately known concentration (a standard solution)