A&P I- Lecture Exam #1
1/217
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
218 Terms
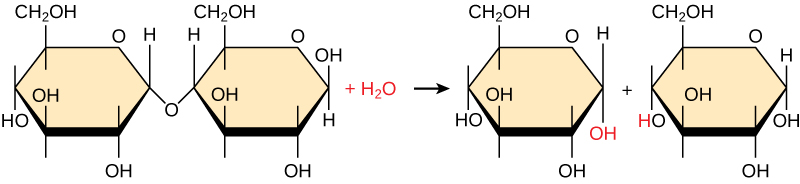
Which of the following is true for the reaction shown below (select all that apply)?
the molecule on the LEFT side of the equation is a POLYMER
the glucose molecules pictures on the RIGHT side of the image are MONOMERS
the glucose molecules pictures on the RIGHT side of the image are the PRODUCTS
this is a hydrolysis reaction
this this is a decomposition reaction
the atoms on the large molecule on the LEFT are joined by COVALENT bonds
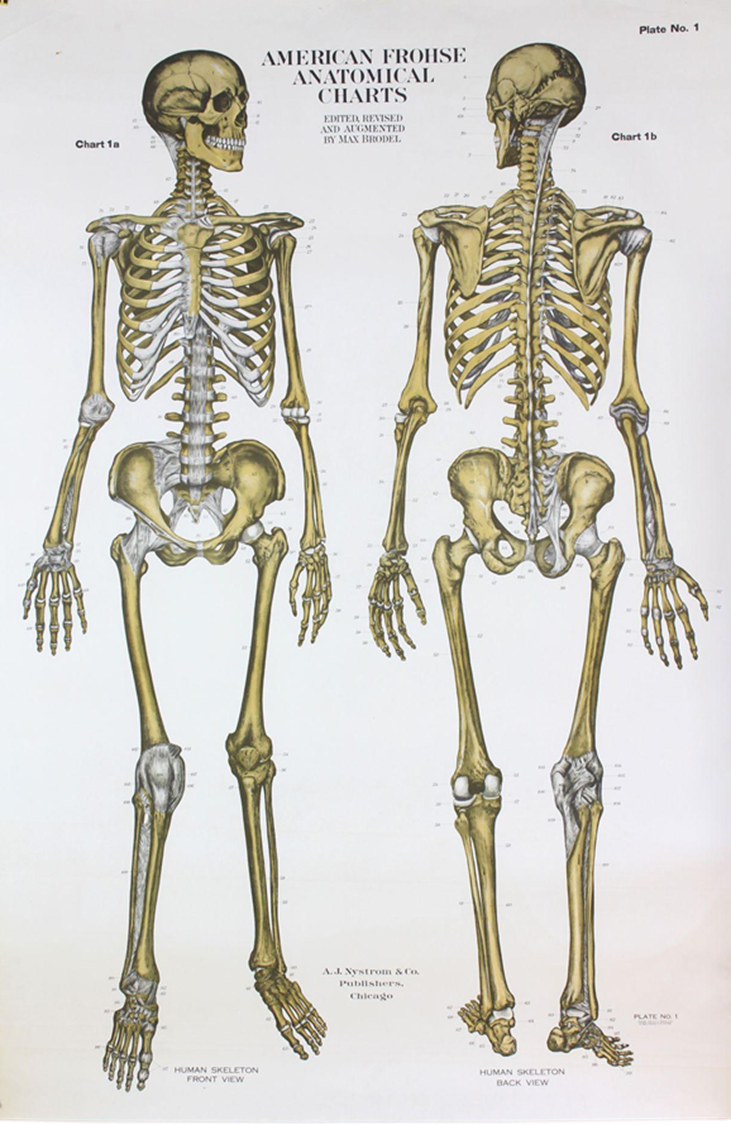
What level of hierarchical structure does the following image belong to?
organ system level
Which of the following is an example of a normal physiologic process that uses a positive feedback loop?
childbirth/labor
What level of structural hierarchy would you expect to include the heart, blood vessels, and blood?
organ system level
ATP is _____.
used to transfer energy within a cell
What is true about electrons in a polar covalent bond?
Electrons are shared in a mutually stabilizing arrangement resulting in a molecule where there are partial positive or negative regions.
Electrons are not involved in the formation of covalent bonds.
Which body cavity contains all of the others listed?
ventral body cavity
The rough ER has its name due to what associated structures?
a. lysosomes
b. ribosomes
d. proteins
Uracil ________.
a. contains nitrogen
b. is a pyrimidine
c. is found in RNA
d. all of the above
NOT C!!!
A phospholipid ______.
a. can donate both cations and anions in solution
d. has both polar and nonpolar regions
The activity of body system must be coordinated for homeostasis
True
Which of the following is true of an acid?
lower than 7 on pH scale
considered proton donors
the strength is determined by how many cations are released
ionize in water and release hydrogen cations
Which of the following specialties might focus on studying all the structures of the ankle and foot?
regional anatomy
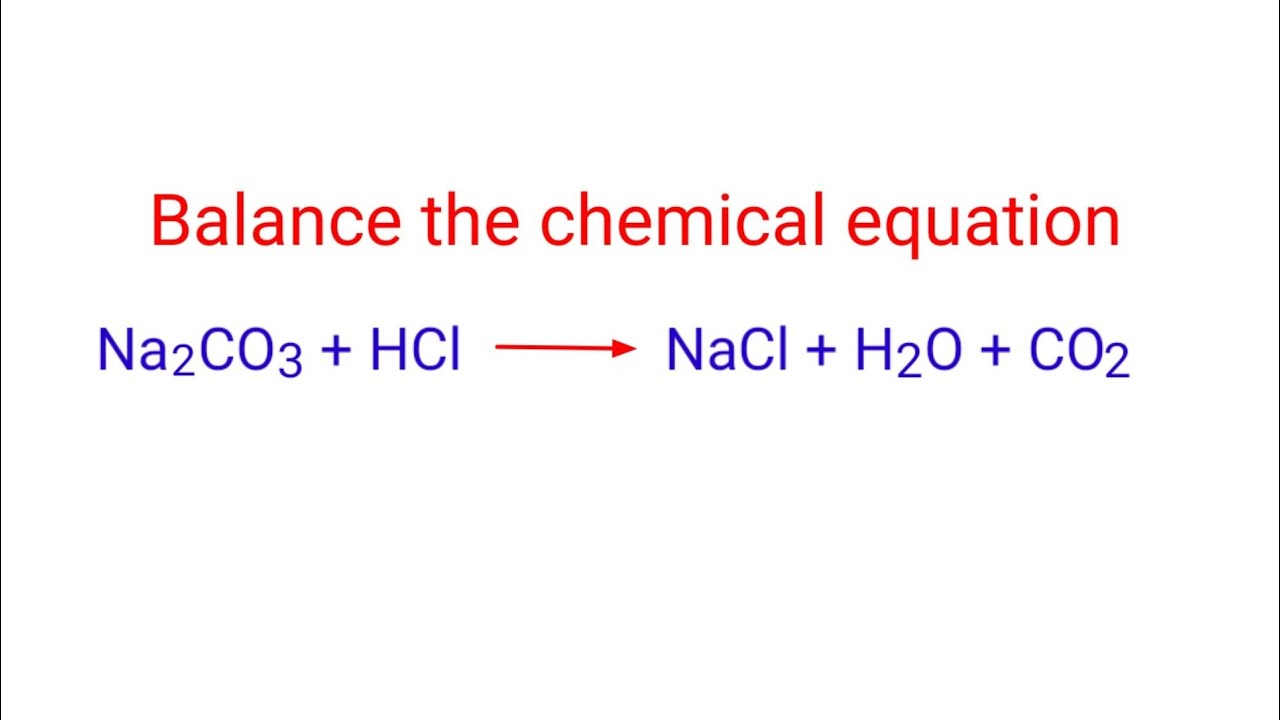
Which of the following is the acid?
HCl
Match term with description (about bonds and charges)
Anion- an atom or molecule with a NEGATIVE charge
Atom- the SMALLEST unit of an element that can participate in chemical reactions
Cation- an atom or molecule with a POSITIVE charge
Electron- NEGATIVELY charged component that orbits the NUCLEUS
Ion- any atom or molecule that has a CHARGE
Molecule- two or more atoms JOINED by an energy relationship
Neutron- NEUTRALLY charged component of the NUCLEUS
Proton- POSITIVELY charge component of the NUCLEUS
A cell with a predominance of free ribosomes is most likely ________.
primarily producing proteins in the cytosol
A molecule of ammonia contains 1 atom of nitrogen and 3 atoms of hydrogen. These are linked with _____.
polar covalent bonds
Large numbers of ribosomes are present in cells that specialize in producing which of the following molecules?
proteins
Why do denatured proteins have reduced or lost function?
due to loss of functional shape
A pentose sugar is part of the monomer used to build which type of macromolecule?
nucleic acids
Why is each element unique with respect to its chemical properties? Each element has a distinctive ________.
number of protons
Together, just four elements make up more than 95 percent of the body's mass. These include ________.
oxygen, carbon, hydrogen, nitrogen
Where in the body do you expect to normally find interstitial fluid?
between cells within tissues
A patient is diagnosed with metabolic acidosis, a condition in which the pH of the blood is more acidic than normal. As the blood and interstitial fluid becomes more acidic, chemoreceptors sensitive to hydrogen cation concentration in the medulla oblongata relay this information to the respiratory centers within the medulla oblongata. These respiratory centers then coordinate the activity of inspiratory and expiratory muscles to change the respiratory rate and depth, resulting in an increase in tidal volume. As the volume of air moved increases, the body is able to off-gas more carbon dioxide and increase the pH of the blood.
Which of the following describes the pattern in the scenario above?
negative feedback
Which of the following organ systems has a major function of enabling movement and maintaining body temperature?
muscular
What is the difference between covalent bonds and ionic bonds?
Covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the ELETRICAL ATTRACTION between charged atoms
Which of the following factors affect the rate of a chemical reaction?
all of the above
A collection of similar tissues that performs a specific function is an ________.
organ
Which of the following is the strongest evidence that protein structure and function are correlated?
Denatured (unfolded) proteins do not function normally
Enzymes catalyze reactions and are consumed (used up) in that reaction.
false
Which of the following body regions corresponds to the term oris?
mouth
If the amount of sodium in the blood decreases, what would a negative feedback control mechanism be expected to do?
increase the amount of sodium in the blood
A _______ is the structure responsible for taking information about a variable and comparing it to the set point and normal range.
control center
The nucleus and mitochondria share which of the following features?
a. protein-lined membrane pores
c. the synthesis of ribosomes
NOT D or B!!!
The body system responsible for structural support and movement is the _______.
skeletal system
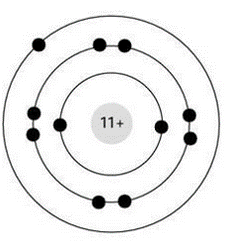
Select all of the following which describe the atom pictured below
sodium atom and electrically neutral
Which SPECIFIC body cavity contains the intestines?
abdominal cavity
Place the mixtures in order of most heterogenous (#1) to least heterogenous (#3)
suspension
colloid
solution
What component of amino acid structure varies among different amino acids?
the components of the R-group
There are 20 different amino acids. What makes one amino acid different from another?
Different side chains (R-groups) attached to an α carbon
What term is used to describe the steady-state value for any variable that the body attempts to maintain?
set point
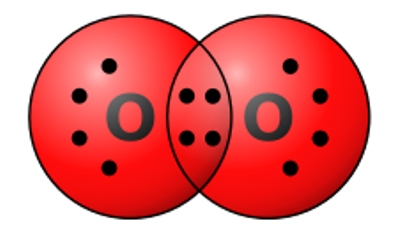
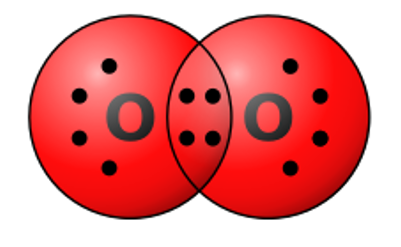
Select all of the following which are true for the molecule pictured below.
the molecule is nonpolar, the molecular structure includes nonpolar covalent bonds, the molecule is a molecule of oxygen
A patient is diagnosed with metabolic acidosis, a condition in which the pH of the blood is more acidic than normal. As the blood and interstitial fluid becomes more acidic, chemoreceptors sensitive to hydrogen cation concentration in the medulla oblongata relay this information to the respiratory centers within the medulla oblongata. These respiratory centers then coordinate the activity of inspiratory and expiratory muscles to change the respiratory rate and depth, resulting in an increase in tidal volume. As the volume of air moved increases, the body is able to off-gas more carbon dioxide and increase the pH of the blood.
What is the stimulus which occurred in the above scenario?
metabolic acidosis
Which of the following combinations of atoms is most likely to result in a chemical reaction?
hydrogen and hydrogen
Physiology is the study of…
how organisms function
The lumbar region is ______.
superior to the popliteal region
Nitrogen (N) is more electronegative than hydrogen (H). Which of the following is a correct statement about the atoms in ammonia (NH3)?
Each hydrogen atom has a partial positive charge; the nitrogen atom has a partial negative charge
Which SPECIFIC body cavity contains the lungs?
pleural cavity
C6H12O6 is the chemical formula for a ________.
hexose monosaccharide
The shape of a protein determines its function(s)
true
Which of the following is the part of metabolism that requires energy for synthesizing (making) body structures?
anabolism
The partial negative charge at one end of a water molecule is attracted to the partial positive charge of another water molecule. What is this attraction called?
a hydrogen bond
What is the role of a catalyst in a chemical reaction?
increase the rate of the reaction
Which of the following is the term for all of the body's chemical reactions including both break down and synthesis activities?
metabolism
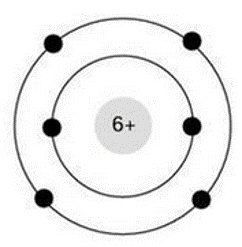
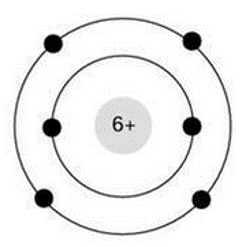
Select all of the following which describe the atom pictured below
carbon atom, electrically neutral
Which of the following is a function of life?
all of the above are functions of life
Eating a salt-rich meal increases blood volume and pressure, stretching blood vessel walls. Nerve signals sent to the brainstem stimulate changes in hormonal and neural signaling. The heart rate is slowed, blood vessel walls are relaxed, and the kidneys increase urinary salt. The blood pressure returns toward normal.
Identify the effectors in this homeostatic reflex:
heart, blood vessels, kidneys
Which of these are functions of the Golgi apparatus?
Modifying proteins synthesized on ribosomes associated with granular endoplasmic reticulum and allowing the distribution of modified proteins throughout the cell in vesicles
The ability of an enzyme's active sites to bind only substrates of compatible shape and charge is known as ________.
specificity
The rate of a reaction occurring in the absence of its enzyme is one molecule of product made per second. Which of the following is likely in the presence of its enzyme?
100 molecules per minute
After you eat lunch, nerve cells in your stomach respond to the distension (the stimulus) resulting from the food). They relay this information to ________.
a control center
Dermicidin is a an antimicrobial peptide (protein) produced by the stratified cuboidal epithelium of the sweat glands.
Place the steps for producing and secreting dermicidin in the correct sequential (numerical) order, starting with the DNA as number 1, and ending with exocytosis as number 10.
DNA contains a gene that codes for dermicidin
the gene for dermicidin is transcribes from DNA to messenger (mRNA)
the mRNA exits the nucleus
the mRNA enters the rough ER
ribosomes on the rough ER translate the mRNA to dermicidin
dermicidin is modifies within the Golgi apparatus
dermicidin is packages into a transport vesicle
dermicidin is packages into a secretory vesicle
the secretory vesicle fuses with the plasma membrane
hermicidin is secretes by exocytosis
GOT IT PARTIALLY WRONG!!
Which of the following body regions corresponds to the term oculus?
eye
Which concept is the defining feature of the discipline of physiology?
homeostasis
During childbirth, as a baby's head is pressed against the mother's cervix, signals are relayed via nerves from cervix to the mother's brain. The brain initiates the secretion of oxytocin (a hormone) from the mother's pituitary gland. Oxytocin stimulates further uterine contractions. As the uterus contracts even harder in response to oxytocin, the baby's head is pushed harder against the cervix; this stimulates yet more nerve signals to the mother's brain, resulting in yet more oxytocin secretion until finally the baby is pushed through the cervix. This process of childbirth is an example of
positive feedback
What is true about electrons in a nonpolar covalent bond?
Electrons are shared in a mutually stabilizing arrangement resulting in a molecule where there is no weak regional charge (no partial positive or negative regions)
Which of the following is the best definition of a set point?
The ideal value (number) of a physiological variable (like the temperature on a thermostat)
Which of the following organ systems has a major function of secreting hormones?
endocrine
You are observing the rate of an enzymatic reaction. You continue to add substrate in higher and higher concentrations, but eventually you see the rate of reaction plateau, no further increases in the rate of the reaction could be observed. Which of the following are plausible explanations for your observation (select all that apply)?
the enzyme has become saturated, the substrate solution has become contaminated with an allosteric inhibitor (a substance that makes the enzyme non-functional)
The molecular formula for glucose is C6H12O6. What would be the molecular formula for a molecule made by linking three glucose molecules together by dehydration reactions?
C18H32O16
Jenny mixes up a batch of pancake batter, then stirs in some chocolate chips. As she is waiting for the first few pancakes to cook, she notices the chocolate chips sinking to the bottom of the clear glass mixing bowl. The chocolate-chip batter is an example of a ________.
suspension
An ___________ is the structure responsible for carrying out the directions of the control center.
effector
The energy stored in a foot of snow on a steep roof is ________.
potential energy
A substance formed of crystals of equal numbers of cations and anions held together by ionic bonds is called a(n) ________.
salt
Spherocytosis is a human blood disorder associated with a defective cytoskeletal protein in the red blood cells (RBCs). What do you suspect is the consequence of such a defect?
Abnormally shaped RBCs
Which of the following is most likely to be found evenly distributed in water in a homogeneous solution?
sodium ions and chloride ions
The characteristic that gives an element its distinctive properties is its number of ________.
protons
Which of the following body regions corresponds to the term auris?
ear
Which of the following is NOT a potential function of proteins?
Energy storage that can be easily digested and metabolized
The active site of an enzyme is a functional binding site
true
A patient is diagnosed with metabolic acidosis, a condition in which the pH of the blood is more acidic than normal. As the blood and interstitial fluid becomes more acidic, chemoreceptors sensitive to hydrogen cation concentration in the medulla oblongata relay this information to the respiratory centers within the medulla oblongata. These respiratory centers then coordinate the activity of inspiratory and expiratory muscles to change the respiratory rate and depth, resulting in an increase in tidal volume. As the volume of air moved increases, the body is able to off-gas more carbon dioxide and increase the pH of the blood.
What is/are the effector(s) in the above scenario?
Inspiratory and expiratory muscles
Which of the following structures covers/touches the heart?
Visceral pericardium
CH4 is methane. This compound is ________.
organic
The term "metabolism"
refers to the synthesis and breakdown of organic molecules involved in cell structure and function.
The bonding of calcium, phosphorus, and other elements produces mineral crystals that are found in bone. This is an example of a(n) ________ reaction.
synthesis
When an atom donates an electron to another atom, it becomes
an ion
Which of the following is a feature common to all three components of the cytoskeleton?
b. They are all polymers of protein subunits.
c. They are all characterized by roughly the same diameter.
What are the reactants?
HCl+ NaHCO3 —> NaCl +H2O + CO2
HCl and NaCO3
The bonding of two amino acid molecules to form a larger molecule requires the ________.
release of a water molecule
Which type of interaction stabilizes the α-helix and the β-pleated sheet structures of proteins?
hydrogen bonds
Which organelles would you expect to be especially numerous in cells such as muscle cells that have a tremendous demand for energy in the form of ATP?
mitochondria
A patient is diagnosed with metabolic acidosis, a condition in which the pH of the blood is more acidic than normal. As the blood and interstitial fluid becomes more acidic, chemoreceptors sensitive to hydrogen cation concentration in the medulla oblongata relay this information to the respiratory centers within the medulla oblongata. These respiratory centers then coordinate the activity of inspiratory and expiratory muscles to change the respiratory rate and depth, resulting in an increase in tidal volume. As the volume of air moved increases, the body is able to off-gas more carbon dioxide and increase the pH of the blood.
What do you expect to occur when the pH of blood and interstitial fluid return to normal values?
The respiratory rate and depth return to resting/normal values
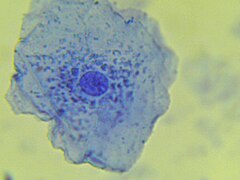
What level of hierarchical structure does the following image belong to?
cellular level
Which of the following attaches the lungs to the thoracic wall?
Parietal pleura
The sharing of electrons between atoms results in the formation of which of the following?
covalent bonds
Stimulation of the control center responsible for cooling off the body would cause which of the following to happen:
sweat glands to increase their output
Which of the following describes an ionic bond?
an electrostatic force of attraction between cations and anions due to apposite charges
The heart is within the _____.
pericardial cavity
What organic compound do brain cells primarily rely on for fuel?
glucose
In which organelle is most of the ATP generated in a cell?
mitochondria