Chapter 18: Ethers + Epoxides, Thiols + Sulfides
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
ether
R-O-R
epoxide
thiol
R-SH
sulfide
R-S-R
ether nomenclature
groups attached to oxygen + ether
alkoxy substituent
ether properties
C-O-C bond is almost tetrahedral (112 degrees)
sp3 carbon
C-O has slight dipole movement
lower boiling points than alcohols, higher boiling points than hydrocarbons
william ether synthesis
less subsituted is better alkyl halide
more substituted + more hindered is better alkoixde ion
not 3 alcohols
williamson ether vs alkoxymercuration
WE
less bulky
akoxy
bulky
needs to be able to form C=C from ether
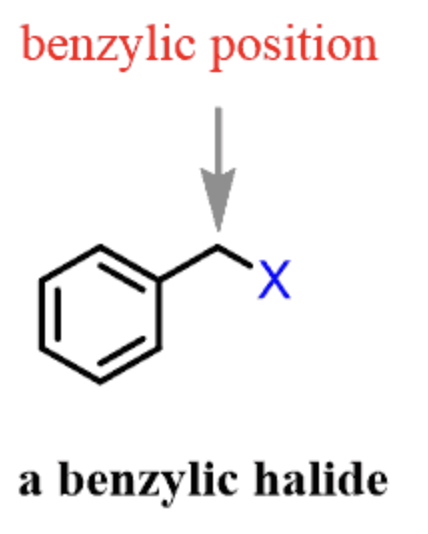
benzylic
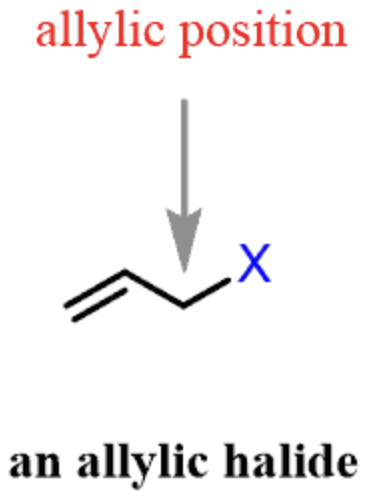
allyl
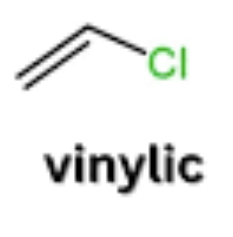
vinyl
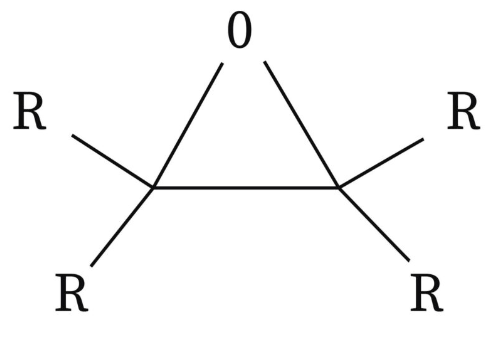
epoxide
3-membered cyclic ethers
always named as substituents - “epoxy”
reaction of alkenes w/mCPBA
reactivity of alkene increases w/increasing alkyl substitution
1 equiv = only 1 C=C reacts
ring opening under acidic conditions
1 + 2 substitution: nucleophile attack occurs primarily at the less substituted site
3: nucleophile attack occurs at the more highly substituted site
both products form
nucleophile (Br) and OH are trans
Br is at more hindered site
inversion occurs
ring opening under basic conditions
Sn2, reaction occurs at less highly substituted position
inversion of stereo!
NMR
epoxides have a slightly higher field (more right) than ethers due to ring strain
thiol
R-SH
thiol nomenclature
add thiol to name of parent hydrocarbon
substituent is mercapto
sulfides nomenclature
R-S-R
sulfide is ending
subsistent is alkylthio