Honors Chem U9: Equilibrium & Acid Base Reactions
1/96
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
What does stoichiometry allow us to do?
make predictions about how much product can be made from a specific amount of reactants
What do we assume with stoichiometry?
we assume reactions will proceed towards completion, but many reactions don't go to completion
Why does stoichiometry and reactions that do NOT go to completion work out?
we can NOT perform stoichiometric calculations since we are unable to predict from initial amount how many products can be formed
What is chemical equilibrium?
reactions that only partially react, or reactions that work in reverse
What is a reversible reaction?
chemical reaction that can occur in both the forward and the reverse directions, represented with a double arrow (⇌)
If some reactions and chemical proccesses have the ability to be reversed, what does that mean?
the original products become "reactants" and the original reactants become "products"
Some reactions are NOT reversible (ex: you cannot unburn a match) but what about the chemical process of water turning into vapor, then condensing back into liquid water?
as long as there is sufficient energy, the reverse reaction is possible
How are reversible reactions indicated?
we show that a reaction is reversible with an arrow that is pointing in either direction, showing that the reaction has the capability of flowing in the forward or reverse direction
What is equilibrium?
where the forward and reverse reaction occur simultaneously
What else can chemical equilibrium be defined as (equilibrium-wise)?
state in which the forward and reverse reactions balance each other out because they take place at EQUAL rates
just because both reactions are possible does NOT mean the concentrations are equal!
Does chemical equilibrium mean that a reaction stops?
does NOT mean the reaction stops; it means that products and reactants start to form at the same rate, so it appears that there is no net movement
The rate of a chemical reaction is an ___________ of the ___________ at which reactants are converted into products.
indication; speed
The forward reaction will have a _________ rate associated with it, and the reverse reaction will have _________ as well.
specific; its own
When the rate of the forward reaction is ________ to the rate of the reverse reaction, we say that the reactions have reached a state of _____________.
equal; chemical equilibrium
As fast as the forward reaction is __________ products the reverse reaction is __________ reactants, so it appears like there is no net change in the _________ on either side of the reaction.
forming; reforming; concentration
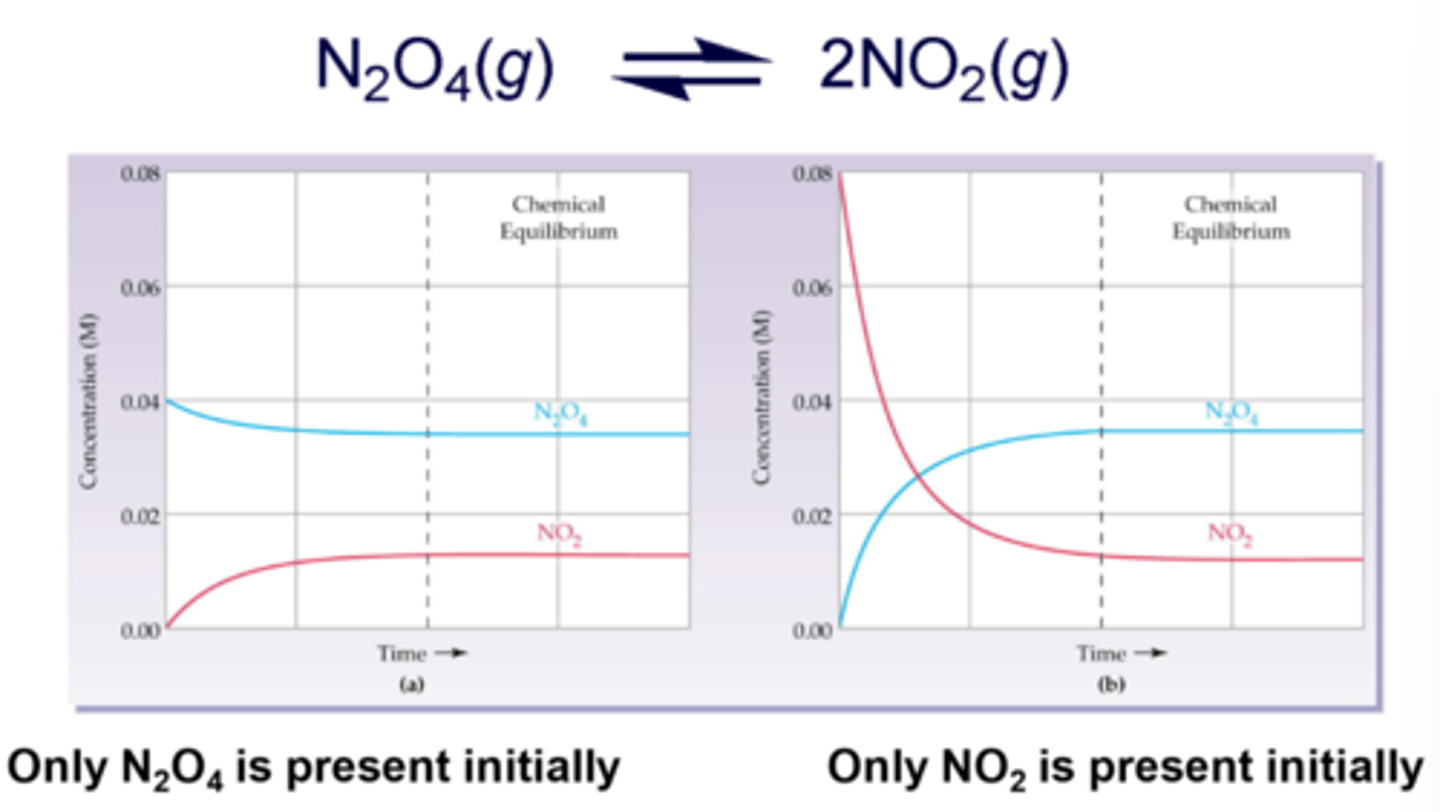
What does "product favoring" mean?
forward reaction occurs more readily before equilibrium is established, meaning that the concentration of products will be higher
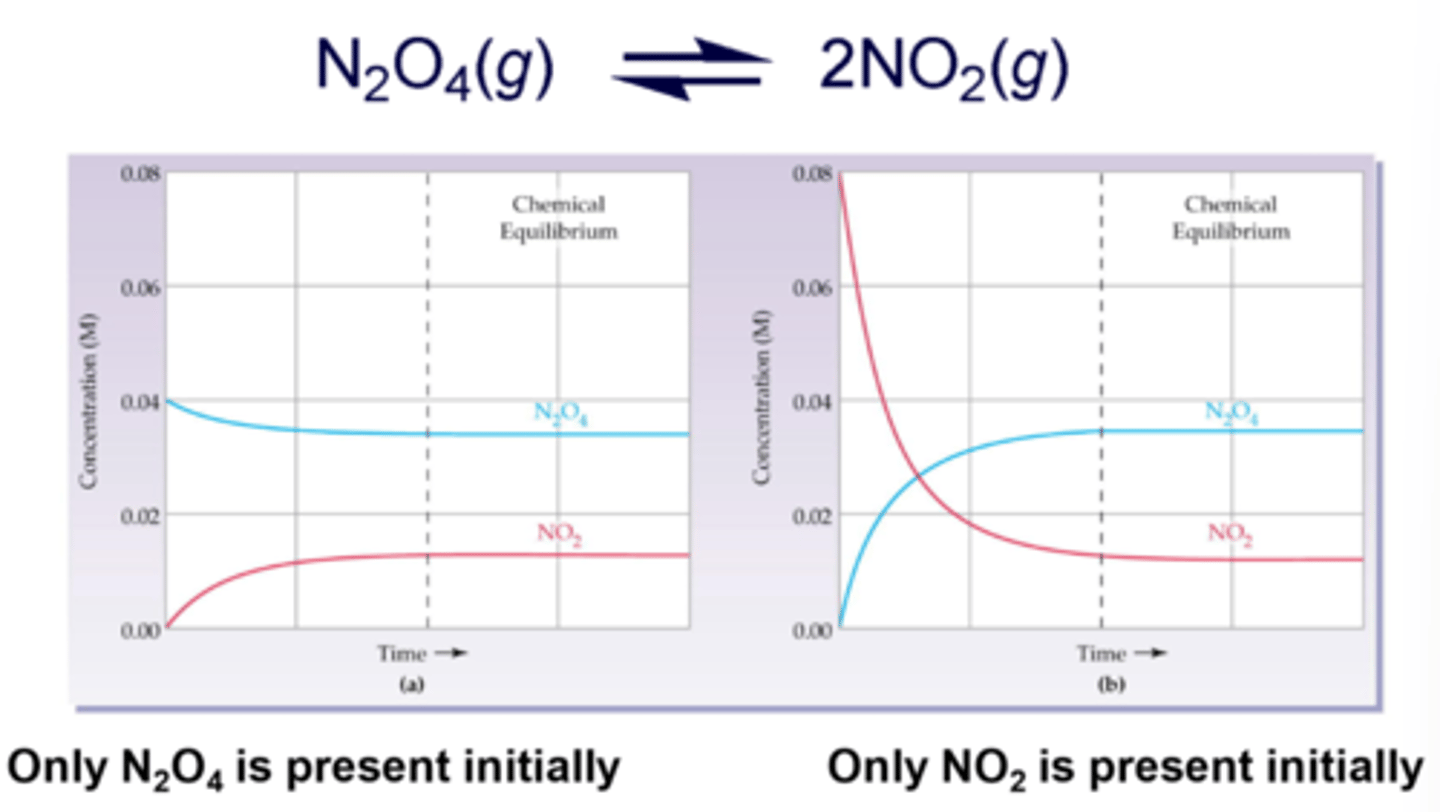
What does "reactant favoring" mean?
either the forward reaction does NOT work well (usually an energy issue), or that the reverse reaction occurs more readily, meaning that the concentration of reactants will be higher
What is the equilibrium constant, K?
unitless value that is a ratio of products to reactants accounting for stoichiometric relationships
What is the equilibrium constant, K, used for?
to calculate concentrations of reactants and products at equilibrium
What does the value of K tell you?
if reactants or products are favored
*K > 1 = product favored
K = 1 = equilibrium
K < 1 = reactant favored
Equilibrium expressions are written to show what K is equal to for each reaction, and these expressions include ONLY what species?
gaseous and aqueous
What are NOT included in equilibrium expressions, and properties do NOT change if volume is changed (unlike g or aq)?
solids and liquids
What is Kc?
equilibrium constant for concentration in molarity (M)
What is Kp?
equilibrium constant for pressure (usually in atm)
What is Kw?
equilibrium constant for the dissociation of water in molarity (M)
What is Ka?
equilibrium constant for the dissociation of acid in molarity (M)
What is Kb?
equilibrium constant for the dissociation of base in molarity (M)
What does it mean if the K value is greater than 1?
products are favored since the numerator will be greater; indicating the forward reaction occurs more until equilibrium is established
What does it mean if the K value is less than 1?
reactants are favored since the denominator will be greater; indicating that the reverse reaction occurs more until the equilibrium is established or if the forward reaction does NOT produce many products
What is phaseequilibria?
system with two or more phases existing simultaneously
(ex: ice cube at 32C or 0F; ice melts and freezes at the same rate)
What is physical equilibria?
system where the same substance exists between two or more different physical properties
(ex: ice cube at 32C or 0F; since its freezing and melting at the same rate, the properties between solid and liquid, like density, are in equilibrium)
What is solution equilibrium?
no more solute appears to dissolve and a state of dynamic equilibrium exists between solute and solvent
(ex: rate of solid dissolving equals the rate of solid forming from ions in a saturated solution)
What is Le Chatelier's Principle?
if a stress is applied to a system at equilibrium, the system shifts in the direction that relieves the stress
What is stress?
any change in a system that upsets equilibrium
If equilibrium shifts left...
producing more reactants reduces the stress
If equilibrium shifts rights...
producing more products reduces stress
What is the Haber Process?
process for synthesizing ammonia from hydrogen and nitrogen gas
Why does the labeled stress change the other components of the reaction on the graph?
because the system is attempting to counteract the disruption and restore equilibrium, as described by Le Chatelier's Principle
What factors affect how a reactions' equilibrium will shift?
- changing concentrations (adding/removing reactants or products)
- changing pressure
- changing temperature
- catalysts*
By adding or increasing the amount of reactants...
- increases chance of collisions
- forward reaction happens more often
- equilibrium shifts to the right
By adding or increasing the amount of products...
- increases chance of collisions
- reverse reaction happens more often
- equilibrium shifts to the left
By removing reactants/products...
reactants/products will need to react to restore what is missing to compensate; equilibrium shifts to the side of removal
By increasing pressure...
(looking for gases, and depends on the # of moles of reactants/products)
equilibrium shifts towards sides with less moles of gas; can better handle the extra pressure
By reducing pressure...
(looking for gases, and depends on the # of moles of reactants/products)
equilibrium shifts towards side with more moles of gas; without the excess pressure, equilibrium shifts back
Why does volume affect pressure and how?
since pressure is formed from the gas molecules colliding with and exerting pressure on the container; if volume decreases, pressure increases; if volume increases, pressure decreases
By increasing temperature...
*endo = heat + A + B ⇌ C + D
exo = A + B ⇌ C + D + heat
- favors the endothermic reaction
- endothermic forward = shifts towards products
- exothermic forward = shifts towards reactants
By decreasing temperature...
*endo = heat + A + B ⇌ C + D
exo = A + B ⇌ C + D + heat
- favors the exothermic reaction
- endothermic forward = shifts towards reactants
- exothermic forward = shifts towards products
Are catalysts a part of the chemical reaction?
no, all they do is reduce the activation energy required for the reaction to occur; it will affect the reactants and products side in the same way with NO shift or effect in equilibrium
What are the properties of acids?
- corrosive (burns you!)
- tastes sour
- has a sticky feel
- turns litmus paper red
- reacts with bases to form water and salt
(ex: citrus fruits, stomach acid, soda, etc.)
What are binary acids?
hydrogen + nonmetal; balanced like an ionic compound so you need enough hydrogens to balance the nonmetal charge; receives "hydro-" prefix with an "-ic" ending, followed by the word "acid"
(ex: Hydrobromic acid)
What are ternary acids?
hydrogen + polyatomic; also balanced like an ionic compound, but do NOT get the "hydro" prefix; either "-ic" if compound is "-ate", and "-ous" if compound is "-ite", followed by the word "acid"
(ex: H2SO4 = Sulfuric (sulfate) acid ; HClO2 = Chlorous (chlorite) acid)
What are the properties of bases?
- corrosive (burns you!)
- tastes bitter
- has a soapy feel
- turns litmus paper blue
- reacts with acids to form water and salt
(ex: bleach, milk of magnesia, soap, toothpaste, etc.)
What are indicators?
substance that gives a visible sign, usually a color change, of the presence or absence of hydrogen or hydroxide ions in various concentrations
What are a few types of indicators?
- universal indicator
- methylene blue
- litmus paper
- phenolphthalein
What are the three models for acids and bases?
- Arrhenius Model
- Bronsted-Lowry
- Lewis Model
What is an acid (Arrhenius Model)?
substance that contains hydrogen and ionizes/dissociates to produce hydrogen ions in aqueous solutions
What is an base (Arrhenius Model)?
substance that contains hydroxide and ionizes/dissociates to produce a hydroxide ion in aqueous solutions
What is dissociation?
physical separation of ions in an aqueous solution
(ex: NaCl breaking apart in solution)
How can percent dissociation be calculated?
by dividing the mass of the dissociated ions by the total mass x 100
What is ionization?
production of new chemical species from dissociated ions
(ex: formation of Na+ and Cl- from NaCl)
What are some problems with the Arrhenius Model?
some compounds produce basic solutions but do NOT contain hydroxide, unlike other models that can explain it fully
(ex: ammonia = NH3)
How can you describe the Bronsted-Lowry Model?
- based solely on the hydrogen ion, H+
- views acids as hydrogen ion donors
- views bases as hydrogen ion acceptors
What is self-ionization?
neutral compound reacts with itself to form ions
What is H3O+ called?
hydronium ion; hydrogen and hydronium ion are used interchangeably to represent acid concentration
What is a conjugate acid?
species produced when a base accepts a hydrogen ion
What is a conjugate base?
species produced when an acid donates a hydrogen ion
What is a conjugate acid/base pair?
consists of two substances related to each other (and look relatively alike) by the donating and accepting of a single hydrogen ion
What is amphoteric?
substances such as water that can act as both acids and bases
What is a proton?
hydrogen ion does NOT have an electron or neutron, so it is named as a different particle
What is a monoprotic acid?
has one ionizable hydrogen
(ex: HBr)
What is a diprotic acid?
has two ionizable hydrogens
(ex: H2SO4)
What is a polyprotic acid?
can be used for any acid that has more than two ionizable hydrogens
(ex: H3PO4)
What does the strength of an acid depend on?
how well it dissociates or breaks apart
What are strong acids/bases?
acids/bases that dissociate completely
What are weak acids/bases?
acids/bases that only partially ionizes
What applies to conjugates with dissociation?
if the acid/base is strong, then its conjugate will be weak; if the acid/base is weak, then its conjugate will be strong
What are the six strong acids?
- Hydrochloric acid (HCl)
- Nitric acid (HNO3)
- Sulfuric acid (H2SO4)
- Hydrobromic acid (HBr)
- Hydroiodic acid (HI)
- Perchloric acid (HClO4)
What are the strong bases (group 1 & 2 hydroxides)?
- Sodium hydroxide (NaOH)
- Lithium hydroxide (LiOH)
- Potassium hydroxide (KOH)
- Calcium hydroxide (Ca(OH)2)
- etc...
What is the pH scale?
scale from 0-14 indicating how acidic or basic a solution is; direct relationship between the negative logarithm of the hydrogen ion concentration expressed in molarity
When is a solution considered acidic?
when the value of pH is between 0 - 6.9; more H+ than OH-
When is a solution considered neutral?
when the value of pH is 7; equal H+ and OH-
When is a solution considered basic?
when the value of pH is between 7.1 - 14; more OH- than H+
What does the pH scale mean?
concentration of hydrogen ions decreases as the scale goes up, becoming less acidic; concentration of hydroxide increases as the scale goes up, becoming more basic
What is water dissociation written as?
- 2H2O (l) ⇌ H3O+ (aq) + OH- (aq)
- *H2O (l) ⇌ H+ (aq) + OH- (aq)
The closer the exponent is to base 10, such as 10^-1, the ______________ it is for both pH and pOH.
more concentrated
What is neutralization?
chemical reaction between an acid and a base which results in no excess hydrogen or hydroxide ions; acids and bases react to form water and salt (NOT always NaCl)
What is titration?
slow addition of an acid or base to a solution of unknown concentration until neutralization is reached
What is a titrant?
solution that is added
What is an analyte?
unknown solution that titrant is added to
What is a buret?
long piece of glassware with a valve
What is the first step for performing titrations?
titrant is place in the buret and slowly dropped into a flask of the analyte with an indicator
What is the second step for performing titrations?
when the indicator changes colors, the titration is complete and the volume of the titrant used is read from the buret and used to make calculations
What is the third and last step for performing titrations?
as end point happens quickly, when the color changes, the equivalence point is reached
What does the equivalence point indicate?
when moles of H+ and moles of OH- are equal; determined by the use of an indicator
What does the end point indicate?
when an indicator changes color; indicators are chosen so that the end point happens at or near the equivalence point
What is needed for titration calculations?
balanced neutralization reaction and the molarity formula
What are the titration calculation steps?
1. Write the balanced chemical reaction
2. Utilizing the known concentration and volume, find the moles
3. Use stoichiometry to convert moles of titrant to moles of analyte
4. Use calculated moles and volume of unknown to find concentration in molarity for analyte