3.1.12.1 Brønsted–Lowry acid–base equilibria in aqueous solution
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
What is a Bronsted-Lowry acid?
Proton donor
Releases H⁺ ions when mixed with H₂O
3 types of bases
Metal …….. eg NaO
Meta ……….. eg) NaOH
Metal ……….. eg) Na_2CO_3
oxides
hydroxides
carbonates
Base vs alkalis solubility
Alkali = A base that is soluble in water. All alkalis are bases
Bases = Insoluble in water
Are most hydroxides soluble or insoluble in water ?
Most hydroxides are SOLUBLE in water so are alkaline
E.g. the alkaline earth metals NaOH (aq)
Common acids, bases and alkalis examples: (they can be in/soluble)
Acids: HCl, H₂SO₄, HNO₃
(Insoluble) Bases: Zn(OH)₂
(Soluble) Alkalis: NaOH, KOH, NH₃
What is a Bronsted-Lowry acid?
and what does it do in water
Proton donor
Releases H⁺ ions when mixed with H₂O
Which arrow sign should be put for weak acids and strong acids?
Weak acids: ⇌ (shows partially dissociation)
Strong acids : → (shows full dissociation)
What is a Bronsted-Lowry base ?
Proton acceptor
Can H₂O act as a base?
Yes
How are strong and weak acids/ bases different?
Strong acids/bases COMPLETELY dissociate/ ionise in aqueous solutions to release H⁺ ions
Weak acids/bases only SLIGHTLY dissociate/ ionise to release small amounts of H⁺ ions
Name a few common weak acids
Ethanoic acid (CH₃COOH)
Phosphoric acid (H₃PO₄)
and other carboxylic acids
Where does the equilibrium lie with weak acids?
Towards the LEFT so the backwards reaction is favoured so not many H⁺ is produced
Name strong acids
HCl hydrochloric acid
H₂SO₄ sulphuric acid
HNO₃ nitric acid
Where does the equilibrium lie with strong acids?
Towards the RIGHT so the forward reaction is favoured so lots of H⁺ is produced
Name Strong Bases
Group 1 metal hydroxides
NaOH, LiOH, KOH, Ca(OH)₂, Ba(OH)₂
Where does the equilibrium lie with strong bases ?
Towards the RIGHT so the forward reaction is favoured so lots of OH⁻ is produced
Name some weak bases
Ammonia (NH₃), Amines
Where does the equilibrium lie with weak bases?
Towards the LEFT so the backwards reaction is favoured so not many OH⁻ ions are produced
Conjugate acid
Each acid is linked to a conjugate base on the other side of the equation
Base + H⁺ ⇌ Conjugate acid
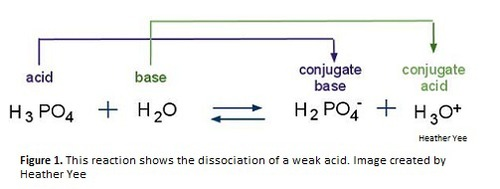
Conjugate base
Each base is linked to a conjugate acid on the other side of the equation
Acid ⇌ Conjugate base + H⁺
What is the conjugate acid of OH⁻ ?
OH⁻ + H⁺ ⇌ H₂O
OH⁻ and H₂O are called a conjugate acid-base pair
What is the conjugate base of HF ?
F⁻
HF ⇌ F⁻ + H⁺
HF and F⁻ are called a conjugate acid-base pair
Whenever a substance acts as a Brønsted-Lowry base, we call the product it forms its...
Conjugate acid
Whenever a substance acts as a Brønsted-Lowry acid, we call the product it forms its...
Conjugate base
Conjugate pairs according to Brønsted-Lowry model
Every acid-base reaction has 2 conjugate pairs
NH₃ (aq) + H₂O (l) ⇌ NH₄⁺ (aq) + OH⁻ (aq)
Conjugate pairs: (NH₃ and NH₄⁺) & (H₂O and OH⁻)
What happens when 2 acids are mixed together?
The stronger acid will act as a Brønsted-Lowry acid, donating a proton
The weaker acid will act as a Brønsted-Lowry base, accepting a proton
Complete the equation for HCl (aq) + CH₃COOH (aq)
HCl (aq) + CH₃COOH (aq) ⇌ Cl⁻ (aq) + CH₃C(OH)₂⁺ (aq)
The HCl is a stronger acid so acts as a proton donor
The ethanoic acid is a weaker acid so acts as a proton acceptor
H₂SO₄ (aq) + CH₃COOH (aq) → HSO₄⁻ (aq) + CH₃C(OH)₂⁺ (aq)
Identify the acid and base according to the Brønsted-Lowry model.
Brønsted-Lowry acid: H₂SO₄
Brønsted-Lowry base: CH₃COOH
Identify the acid and base according to the Brønsted-Lowry model.
NH₃ + NH₃ → NH₄⁺ + NH₂⁻
Give your answers as compound names, rather than chemical formulae.
Brønsted-Lowry acid: ammonia 1
Brønsted-Lowry base: ammonia 2
Ammonium ions can be used as a weak acid in organic reactions. Write an equation for the dissociation of this acid.
NH₄⁺ ⇌ H⁺ + NH₃
Consider the following acid-base reaction:
C₅H₅N + HCl → C₅H₅NH⁺ + Cl⁻
What are the conjugate pairs ?
Conjugate acid = HCl, Conjugate base = Cl⁻
Conjugate acid = C₅H₅NH⁺, Conjugate base = C₅H₅N
Conjugate acid = HCl, Conjugate base = Cl⁻
Conjugate acid = C₅H₅NH⁺, Conjugate base = C₅H₅N
What is a monoprotic acid ? Give 3 examples
● An acid that donates ONE H⁺ ion for every acid molecule.
● So the concentration of the H⁺ ions is equal to the concentration of the acid HCl, HNO₃, HBr
What is a diprotic acid ?
● An acid that donates 2 H⁺ ions for every acid molecule.
● So The concentration of H⁺ ions is 2x the concentration of the acid [H⁺]= 2[Acid]
E.g. H₂SO₄
How do diprotic acids dissociate? Use H₂SO₄ as an example.
First: H₂SO₄ → H⁺ + HSO₄⁻
Second: HSO₄⁻ ⇌ H⁺ + SO₄²⁻
Dissociates 1 proton at a time, and the 1st proton dissociates more fully than the 2nd
Acidity
The higher the concentration of H⁺ the higher the acidity