Reactions + Reagents/Products (Alkene Nucleophiles)
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Alkene Nucleophiles —> Acid Initiated
Electrons from double bond = Nucleophile —> attack acidic H from top/bottom = racemic
Negative Conjugate Base group attach to most substituted carbon
Stabilizing due to hyperconjugation
Hydration
Reagents: H2SO4, H2O excess
Products: OH on most substituted carbon of once-alkene and H on least substituted carbon of once-alkene
Addition to ALKYNE
Reagent: H-X (excess) [X: Br, Cl, etc)
Product: Hydrogens Attach to one carbon of once-alkyne, 2 X atoms bonded to carbon of once alkyne
Hydration of Alkyne
Reagents: H2SO4, H2O
Products: RC=OMe
Carbocation Rearrangements —> Ring Expansion
Reagents: H-X [X:Cl, Br, I] strong acid
Product: Ring expanded one more side
Carbocation Rearrangement —> rearranged to form more substituted carbocation
Reagents: H2SO4, H2O
Products: Carbon ring containing alkene —> Alkene replaced with Me group on one carbon (shifted Me from different carbon ring “alkyl migration”) and Hydrogen the other; Migrated Alkyl replaced with OH group
Rearrangement for resonance stability
Reagents: H-I (Strong Acid)
Product: Alkene group —> H on least substituted carbon, H from neighboring beta carbon shifts onto other carbon forming cation on beta carbon which attracts -I
Alkene Nucleophiles Electrophile Inititated
result in ANTI addition (Added Electrophile and Nucleophile groups anti of each other)
What was trans originally is ANTI
what was cis originally is SYN
Electrophile attract electrons from double bond and donate lone pair to one of the carbons in double bond to form 3 member ring
Dihalogenation
Reagent: X2 [X: Cl, Br]
Product: Anti X with same orientation on each carbon
Halohydrin Formation
Reagents: Br2/I2/Cl2, H2O
Product: Anti X and OH groups
EPOXIDATION
reagent: 1. mCPBA, CH2Cl2 2. NaOH, H2O
Product: Anti OH groups
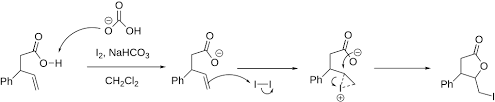
Iodolactones
Reagent: 1. I2, H2O
Products: see image
Alkene Nucleophiles —> Syn Addition
Added groups on same side
What was trans originally is ANTI
what was cis originally is SYN
Hydroboration-Oxidation
Reagents: 1. BH3, THF (solvent) 2. NaOH, H2O2, H2O
Product: Cis Groups of H and OH
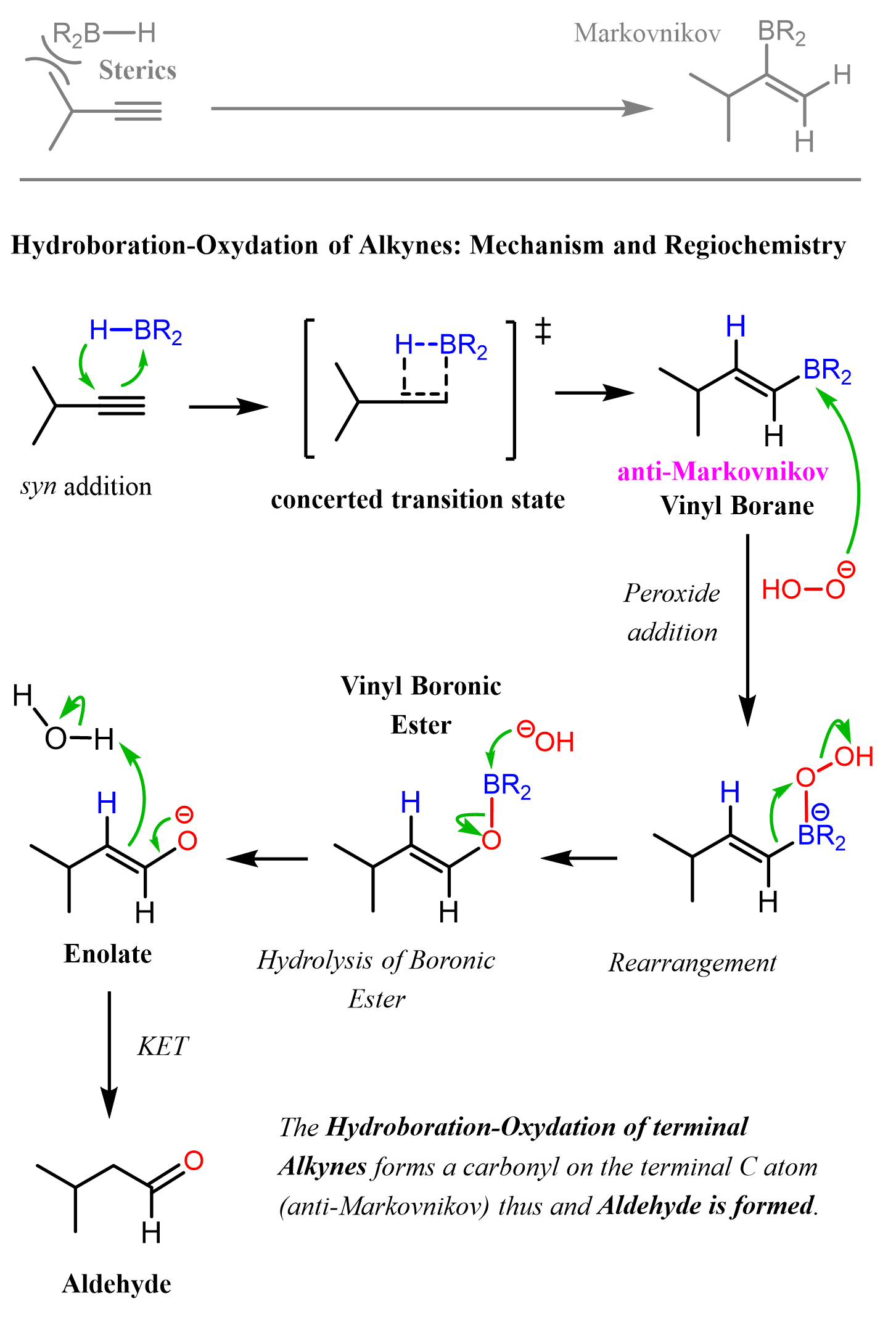
Alkyne Hydroboration-Oxidation
Reagents: 1. BH3, THF 2. NaOH, H2O2, H2O
Product: Alkyne —> Aldehyde
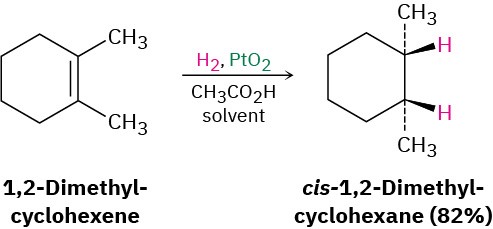
Hydrogenation
Reagents: H2, EtOH, Pd/C or Pd/I
Product: Cis H atoms bonded to each carbon
Alkyne Hydrogenation
Reagents:
H2, EtOH, Pd/C —> Goes all the way to Alkane
H2, EtOH, Pd/C, Pb+2, Purine —> goes only to alkene
Dihydroxylation
Reagent: OsO4, NaHSO3, H2O
Products: Cis groups of OH
Ozonolysis
Reagent: 1. O3, CH2Cl2 2. Me2S
Product: 2 Ketones
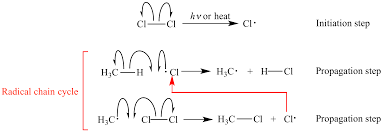
Radicals
Unpaired electrons formed by homolyzing a weak bond with heat or light
STABILIZERS:
Sterics (More steric hinderance around radical = more stable)
Electronegativity (less Electronegative the atom = more stable)
Hyperconjugation (the more substituted the atom = more stable)
Resonance (more resonance it can participate in = more stable; sp2 electrons)
Radical Chain Reaction
Reagent: RO-OR or X2 (Cl,Br)
Initiation: Homolyze weak bond of reagent with light/heat to create radical
Propogation: React with alkene
Termination: remaining radicals form a bond to end the reaction