CHM 230 Exam 3
0.0(0)
Studied by 5 peopleCard Sorting
1/34
Earn XP
Description and Tags
Last updated 1:45 PM on 3/31/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
1
New cards
How do you find Q
use inital molarities to find K
2
New cards
If Q> K the equilibrium shifts
left
3
New cards
If Q < K the equilibrium shifts
right
4
New cards
If Q = K then the system is
at equilibrium
5
New cards
What is the relationship between delta G and K
delta G= -RT \* lnK
6
New cards
the position of K is dependent on
temperature
7
New cards
an acid is a compound that _____ H+
donates
8
New cards
a base is a compound that _____ H+
accepts
9
New cards
ph=
\-log\[H3O+\]
10
New cards
\[H30+\]=
10^-pH
11
New cards
What is the equation for the autodissociation of water?
2H20-→ H3O+ + OH-
12
New cards
kw = (number)
1x 10^-14
13
New cards
kw = (expression)
\[H30+\] x \[OH-\]
14
New cards
what = 14
pH + pOH
15
New cards
ability to donate H+ is dictated by
Ka
16
New cards
What are the 7 strong acids
HCl, HBr, HI , HNO3, H2SO4, HCLO3, HCLO4
17
New cards
by definition a strong acid has ( )= ( )
\[HA\]=\[H3O+\]
18
New cards
What is the acid dissociation expression
HA (aq) + H20 (l) -→ H30+ (aq) + A- (aq)
19
New cards
Weak acids have a Ka
20
New cards
higher Ka =
stronger acid
21
New cards
When do you not need to solve a quadratic
\-when x to intial M is less than 5%
\-or when intial M: K is > 1000
\-or when intial M: K is > 1000
22
New cards
When does the assumption fail
\-when Ka gets close to 1
\-when HA gets small
\-when HA gets small
23
New cards
% ionization=
amount H30+/ M\[HA\]
24
New cards
What is the conjugate base of hydocyanic acid, HCN
CN-1
25
New cards
What is the conjugate acid of hydrogen sulfate anion HSO4-1
H2SO4
26
New cards
Which of these weak acids is the strongest?
\-propanic acid Ka= 1.4 x 10^-5
\-phenol Ka= 1.3 x 10 ^-7
\-benzoic acid Ka= 6.8 x 10^-5
\-ammonium cation, Ka= 5.6 x 10^-10
\-propanic acid Ka= 1.4 x 10^-5
\-phenol Ka= 1.3 x 10 ^-7
\-benzoic acid Ka= 6.8 x 10^-5
\-ammonium cation, Ka= 5.6 x 10^-10
benzoic acid
27
New cards
Which acid is the strongest
\-HCLO4
\-HNO3
\-HCLO2
\-HBrO3
\-HCLO4
\-HNO3
\-HCLO2
\-HBrO3
HClO4
28
New cards
Which of these ionic compounds will create a neutral solution when dissolved in water?
\-ammonium nitrate
\-sodium perchlorate
\-sodium carbonate
\-iron (II) chloride
\-ammonium nitrate
\-sodium perchlorate
\-sodium carbonate
\-iron (II) chloride
sodium perchlorate
29
New cards
Which of these ionic compounds will create a BASIC solution when dissolved in water?
\--ammonium nitrate
\-sodium perchlorate
\-sodium carbonate
\-iron (II) chloride
\--ammonium nitrate
\-sodium perchlorate
\-sodium carbonate
\-iron (II) chloride
sodium carbonate
30
New cards
Which of these could you add to solution of HCN to make a buffer?
\-potassium nitrate
\-lithium cyanide
\-water
\-sulfuric acid
\-potassium nitrate
\-lithium cyanide
\-water
\-sulfuric acid
lithium cyanide
31
New cards
Suppose you need to make a buffer with a pH= 8.50
Which of these systems would be the best choice?
\-pyruvic acid/pyruvate pka= 2.55
\-acetic acid/acetate pka=4.74
\-phenol/phenolate pka= 9.89
\-boric acid/borate pka= 9.27
\
Which of these systems would be the best choice?
\-pyruvic acid/pyruvate pka= 2.55
\-acetic acid/acetate pka=4.74
\-phenol/phenolate pka= 9.89
\-boric acid/borate pka= 9.27
\
\-boric acid/borate pka= 9.27
32
New cards
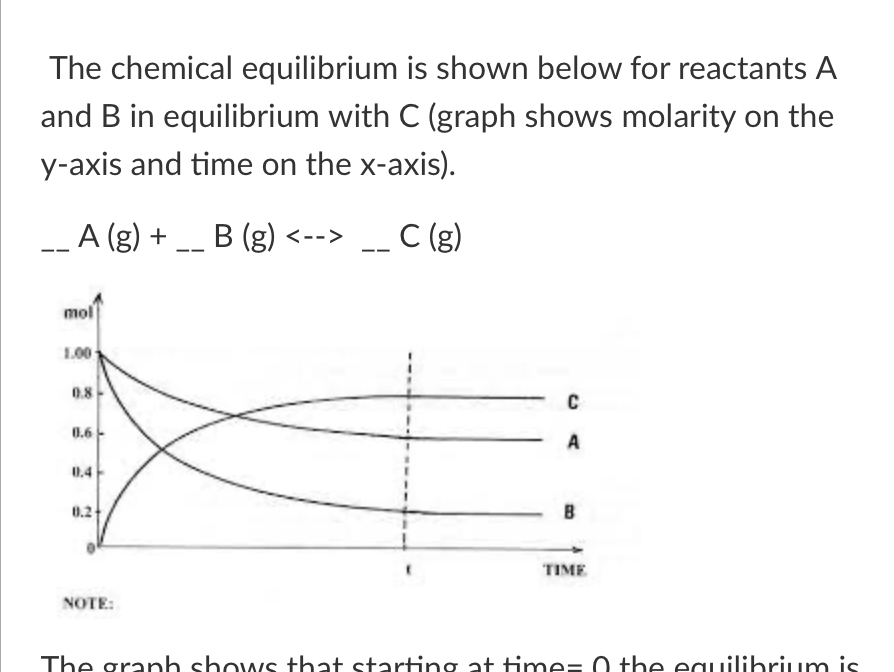
The graph shows that starting at time=0 the equilibrium is moving…
to the right (toward products)
33
New cards
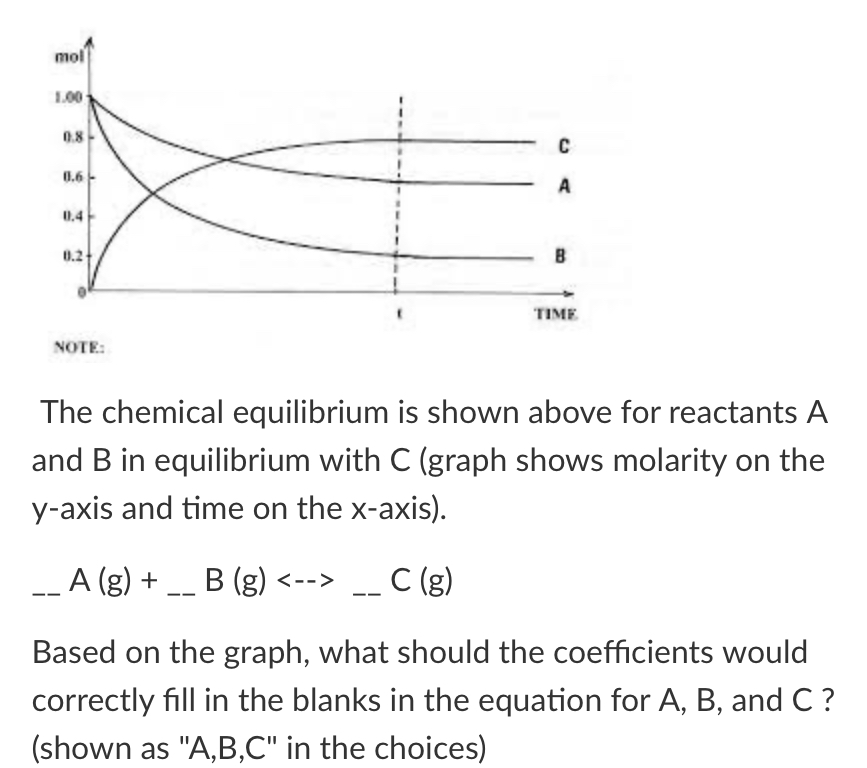
Based on the graph what should be the coefficients for ABC
1,2,2
34
New cards

Which reaction has the LARGEST value for equilibium constant K
A+D→ AD
35
New cards

Which reaction has the SMALLEST value for equilibrium constant K
A+C-→ AC