Alkanes and alkenes
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Explain what type of hydrocarbon alkanes and alkenes are
Alkanes are saturated hydrocarbons because they contain only C−C single bonds and have no double or triple bonds AND (very important for M2) with no additional SOMETHING Q NO ME ACUERDO
Alkanes are unsaturated hydrocarbons because they contain a double C=C bond
What is the general formula for alkanes?
CnH2n+2
The first __ alkanes are gases at room temp (around 25 °C)
All other alkanes you are likely to see at IGCSE are ___
Four
Liquids
Members of a homologous series have similar chemical properties because__
What is the chemical property of alkanes?
The have the same functional group (+ bonding within the molecules)
They are fairly unreactive organic compounds — often thought of as being quite inert
Explain why alkanes are quite inert
They only have C−C single bonds and C−H bonds which are strong bonds
Key point
What is the actual meaning of inert when we refer to ‘alkanes being quite inert’?
The alkanes are not inert in the sense that they don’t react with anything, like neon, but they are not very reactive for organic compounds and only really undergo 2 reactions
What are the two reactions of alkanes?
Combustion and substitution
Why is highly unlikely that in combustion reactions a product is hydrogen?
Because hydrogen is very flammable so you will always obtain water, even if there is insufficient oxygen
Substitution reaction
What is the difference when you need to react an alkane with a halogen vs an alkene with a halogen?
Describe why it is a substitution reaction
Alkanes will only react the presence of ultraviolet radiation (e.g from sunlight). Alkenes will react without the need for UV light
A hydrogen atom in the alkane is replaced by a halogen atom
What is mono-substitution?
A substitution reaction in which only one hydrogen atom in an alkane is replaced by a halogen atom
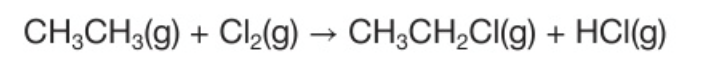
When ethene and chlorine are exposed to UV light you get a mixture of chloroethane and hydrogen chloride gases
Why is the HCl*
What is the name given to the products formed from substitution reactions of alkanes with halogens?
How is the product named?
Halogenoalkanes
According to the format x-haloalkane, where x indicates the position of the halogen atom in the longest carbon chain.
What are the uses of halogenoalkanes?
They have many uses as solvents, refrigerants, propellants and pharmaceuticals
Savemyexams
CFC stands for chlorofluorocarbon, an important class of halogenoalkanes, previously used in fridges and air conditioners, but gradually being phased out because of the damage they cause to the ozone layer.
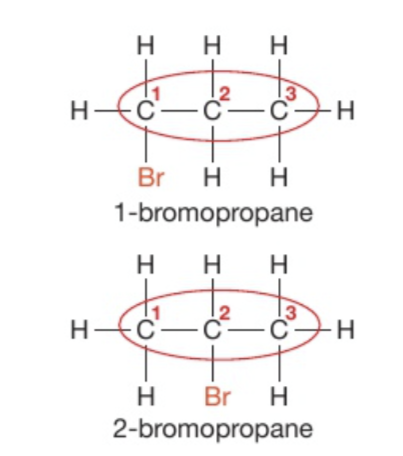
When propane (and alkanes a partir de este) reacts with bromine will be able to form 2 organic products when reacted with a halogen like bromine even when only mono-substitution occurs.
What are the names of both products? What is the link between both products?
Propane
The two products are structural isomers of bromopropane, they have the same molecular formula but different structural formulae
General formula of alkenes? What is their functional group?
CnH2n → therefore all their empirical formulae is CH2
Functional group is C=C
Physical properties of alkenes?
Similar to those of the alkanes. REMEMBER: all the members of the homologous series show a gradation (trend) in their physical properties
For both alkanes and alkenes, those with up to four carbon atoms are gases at room temp
The next dozen or so are liquids
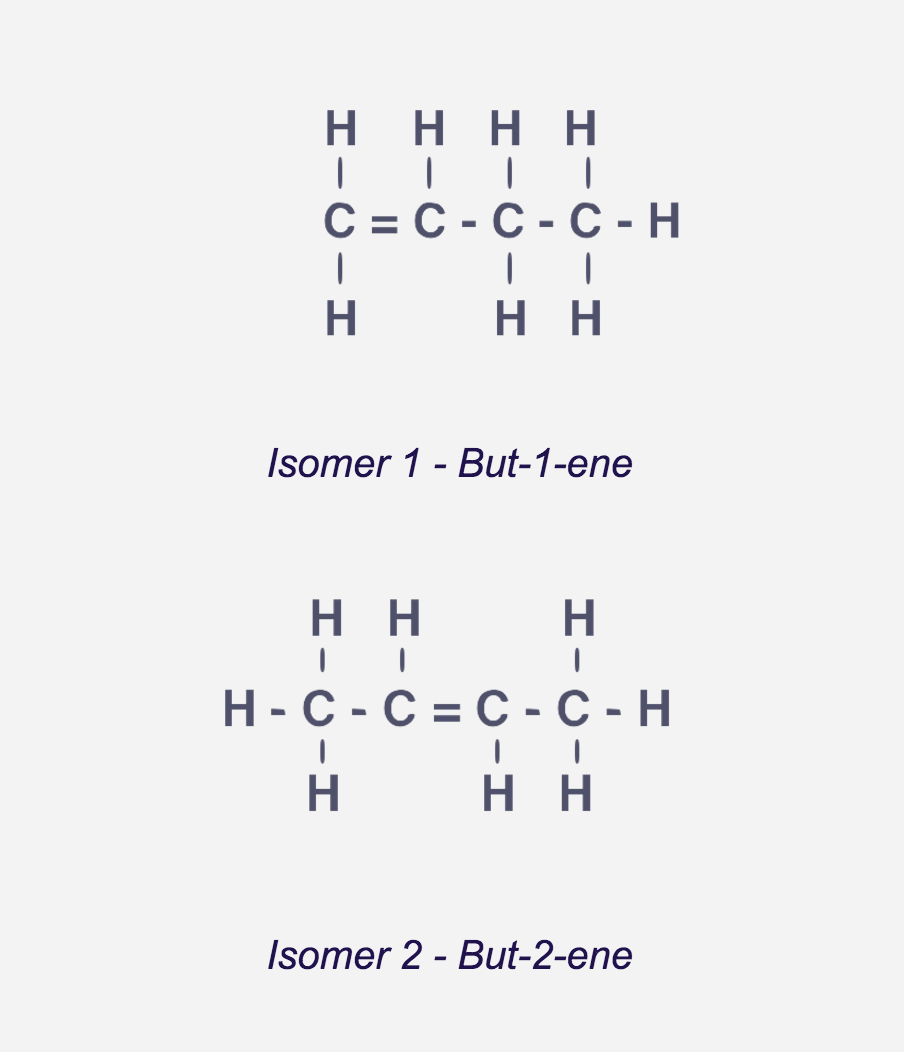
What are the unbranched chain isomers of alkenes?
They refer to the different structural formula they can have depending on the different position the double bond has
Alkenes are much more/less reactive than alkanes
What is a type of reaction that alkenes but not alkanes can undergo? Why?
Alkenes are much more reactive than alkanes
Alkenes undergo addition reactions. The presence of the C=C double bond allows alkenes to react in ways that alkanes cannot
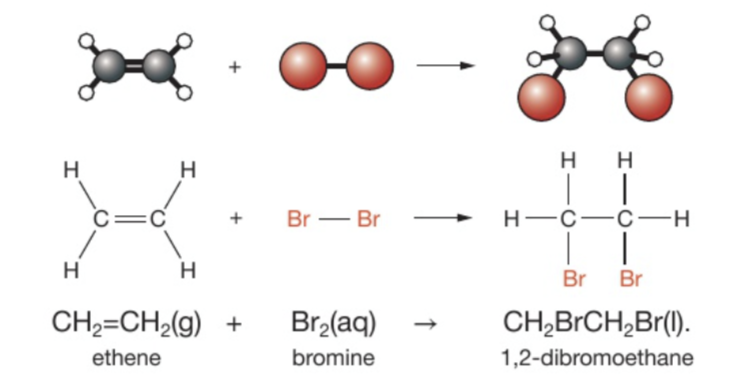
Describe the reaction between alkenes and bromine: e.g ethene and bromine, pero es lo mismo para cualquier otro alkene creo
What type of reaction?
Requirements?
Products? The general name when bromine adds to alkane and for this specific one
Observations when a reaction has occurred?
It is an addition reaction
Bromine adds to ethene (o cualquier otro alkene) without the need for heat, light or a catalyst
The product name is a dibromoalkane (changes from -ene a -ane CAREFUL WITH ENDINGS). Of this specific reaction we get 1,2-dibromoethane
If there is a reaction it turns from orange (because bromine water is orange) to colourless because 1,2-dibromoethane has formed (it is a colourless liquid)
How can you distinguish an alkane from an alkene (test for unsaturated compounds)? Why does it allow alkenes to do this but alkanes not?
Bromine water is an orange solution of bromine
With an alkane, it will remain as an orange solution as alkanes do not have double bonds
The alkene will decolorise the bromine water and turn it colourless
The alkenes have C=C double bond allows alkenes to react in ways that alkanes cannot. This double bond makes them more reactive than alkanes as this double bond contains an area of high electron density.