Ecology Final Exam Flashcards (Exam Questions)
1/223
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
224 Terms
Which of the following chemical bonds is strongest in cells?
Covalent
There are ___ common amino acids.
20
Name 2 basic amino acids
Lysine and Arginine
Name the purine necleotides
Adenine and Guanine
How many nanometers are in 1.78 centimeters?
17,800,000 cm
Name 2 of the uncharged, polar amino acids
Serine and threonine
In a polar covalent bond consisting of hydrogen and oxygen, the _______.
Oxygen has a partial negative charge because it is more electronegative than hydrogen
A monosaccharide with 4 carbons will have ____ oxygens and ____ hydrogens.
4 and 8
What living things are the universal building blocks of all living things?
Cells
Which fatty acid type contains NO double bonds?
Saturated
Define a protein domain:
a distinct, stable, and functional unit within a protein’s tertiary structure that can evolve, function, and exist independently of the rest of the protein chain
key characteristics:
self-stabilizing
functional unit
evolutionary “Lego Bricks”
What would the resolution of a microscope using a light source with a wavelength of 400 nm and a numerical aperture of 1 be?
200 nm
d = lambda (wavelength)/ 2(NA) [Numerical Aperture of the lens]
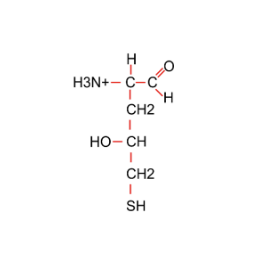
Which of the following chemical groups is not present in the compound in the figure?
Carboxylic acid

A derivation of the nucleotide adenine forms the energy molecule ____.
ATP
Which of the following is/are present in all cells?
All of the above are in all cells
Which of the following polysaccharides is made by humans?
Glycogen
Which of the following are true of ALL proteins?
None of the above is correct
Name the 3 domains of life.
Prokaryotic cells, eukaryotic cells, and archaea
Name the 4 macromolecules of life:
Carbohydrates, Proteins, Lipids, and Nucleic Acids
Beta-sheets in which the amino (N)-termini of the beta-strands are aligned are described as ____.
Parallel
Which amino acid is incompatible with the formation of alpha helices?
Proline
The reason is due to lack of hydrogen bonding.
The earliest light microscopes were noted in the historical record in the _____.
1600s
How many valence electrons are in carbon?
4
The percentage of water in cells ranges from _____ percent.
70%-95%
If the pH of cells the normal range of approximately 7.2, the ______ group in amino acids can donate a hydrogen to lower the pH.
Carboxyl Group
The carboxyl groups is an acid. At pH 7.2, it has already donated its proton to the environment, existing in its conjugate base form, -COO^-
Which of the following is correct about alpha helices?
They are stabilized by hydrogen bonding.
Sucrose is a disaccharide made of ___.
Fructose and Glucose
Which of the following elements make up 96% of all living matter?
Calcium
Carbon
Hydrogen
Nitrogen
Oxygen
Carbon, Hydrogen, Nitrogen, and Oxygen
Scientists estimate that prokaryotes were first found on earth approximately how many billion years ago?
3.5
The amino acid sequence of a polypeptide is its ___ structure.
Primary
Which type of microscopy requires coating the specimen with heavy metal?
Scanning electron microscopy (SEM)
Name 4 things that all living things do.
Maintain homeostasis, using energy (metabolism), respond to stimuli (detecting and respond to changes in their environment), and reproduce and grow
Which of the following types of bonds act in protein folding?
All of the above
Which of the following atoms is most likely to form an anion in a dissociated ionic bond?
Fluorine
What is the carbohydrate in DNA, and how many carbons are in it?
deoxyribose and 5 carbons (pentose)
The majority of amino acids have side chains that are ____.
Nonpolar
At least some of the illumination source passes through specimens in all of the following types of microscopes, except?
Scanning Electron
When was the cell theory proposed?
1839
Who showed in the laboratory that spontaneous formation of organic compounds that could form the macromolecules of life could occur in conditions found on primitive Earth?
Theodor Schwann
Seeing things clearly using a microscope depends on all of the following, except _______.
Microscope Working Distance
Which of the following chemical bonds is due to the random, ever-changing accumulation of electrons in one part of a molecule?
Van der Waals interactions
Which of the following polymers are created by dehydration or condensation reactions?
All of the Above
A cell with endoplasmic reticulum most likely belongs to the domain ____.
Eukaryotes
An electron microscope would be needed to view which of the following?
Ribosomes
Which of molecules below demonstrates the ability of chains of carbons to form ring structures?
All of the Above
Name 4 types of proteins
Enzymatic, Structural, Transport, and Defensive
The lenses in an electron microscope are actually_____.
Magnets
The cell theory was proposed by _______.
Schleiden and Schwann
Which of the following is a part of all amino acids?
Amino Group
Two microscopes with the same lenses are using different light sources. Microscope A is using visible light. Microscope B is using ultraviolet light that has a wavelength of 350 nm. Which of the following is true?
The resolution of microscope B is better than microscope A.
The simplest amino acid is ____, and its R group is ___.
Glycine . . . a hydrogen atom
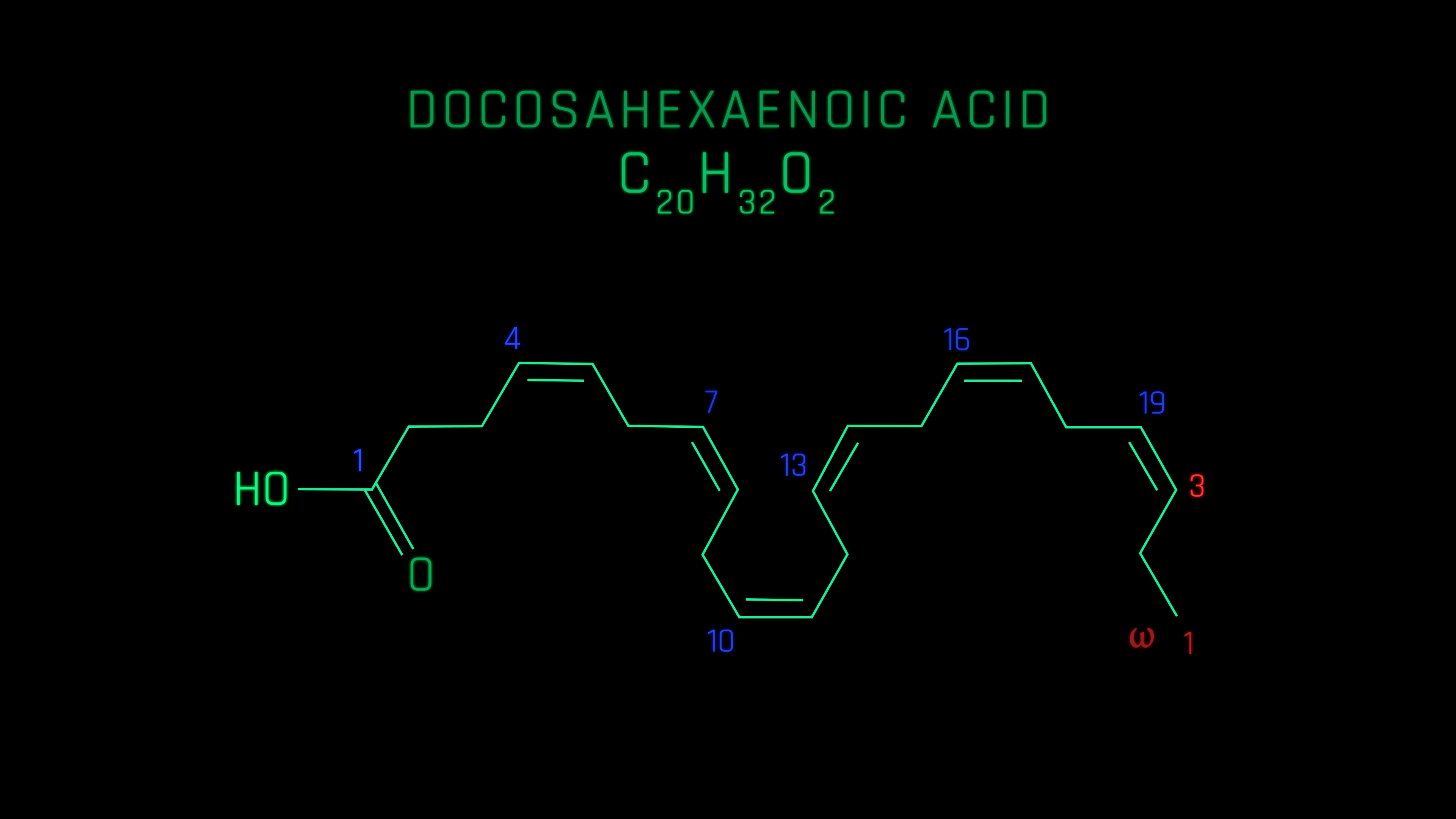
How many double bonds are in docasahexaenoic acid (DHA)?
6 double bonds
What organelles have a cisternae?
Golgi apparatus, Nucleus, and the Endoplasmic Reticulum
Which of the following is correct?
None of the above
Which of the listed organelles makes proteins that are secreted from the cell? (indicate all that apply)
Rough Endoplasmic Reticulum
Which of the following is correct about transmembrane proteins that form channels for hydrophilic substances to pass?
None of the Above
Which of the following can most easily pass through the phospholipid bilayer of cells without the assistance of proteins?
Steriods
Serial endosymbiosis suggests what?
Mitochondria formed because aerobic bacteria united with pre-eukaryotic cells
Who in the 1920s used bacteria to provide evidence that there was a "transforming principle."?
Frederick Griffith
Part of the mitotic spindle attaches at the ____ of chromosomes.
Centromeres
Catalase is an oxidative enzyme that can be found in what?
Peroxisomes
Histone modifying enzymes add what to histones?
All of the Above
Why is the movement of sodium ions into human cells more powerful than the movement of potassium ions out of the cells?
Sodium is moving down its concentration gradient
The most widely used and abundant activated carrier is what?
ATP
Who authored the seminal 1953 Nature paper on the structure of DNA?
James Watson and Francis Crick
Which of the following chemical interactions is most important in formation of the phospholipid bilayer of around cells?
Hydrophobic?
What reinforces the plasma membrane, giving cells their shapes?
cytoskeleton
What is involved in condensing the interphase chromosomes?
Cohesins
What do chromatin remodeling complexes do?
Use energy from ATP to change the position of nucleosomes
Which of the following movements is done by phospholipids in cellular membranes?
All of the Above
The resting membrane potential of most human cells is about ___.
-70 mV
Histone H1
shortens the linker regions between nucleosomes
Who was involved in determining that DNA is the genetic material?
Fred Griffith, Martha Chase, and Oswald Avery
Who used a phage virus to definitively dertermine that DNA is the genetic material?
Alfred Hershey and Martha Chase
What determines if a ribosome will attach to the endoplasmic reticulum by interacting with proteins in the ER membrane?
polypeptide
Proteins that form channels for the osmosis of water in some cells are called?
aquaporins
___ transfers phospholipids randomly from the cytosolic to the lumen side of its phosphoipid bilayer and is found in the ___.
Scramblase … Smooth ER
Movement of a large 100 kD protein into the nucleus will involve which of the following?
All of the above
Organisms in what domains typically have linear DNA?
Eukaryotes
When the cytokine IL-10 is shipped from the Golgi apparatus, it is immediately released from the cell. This is an example of ___.
Constitutive secretion
In nucleosomes, how many nucleotides pairs wrap around the histone core?
147
What occurs during the depolarization stage of an action potential?
Sodium ions enter the cell.
Gradient-driven antiports do which of the following?
Use the diffusion of one substance into the cell to provide energy for moving a second substance out of the cell
Which parts of nucleotides are directly involved in the phosphodiester bond of the backbone of nucleic acids? Be very specific: the ___ bonds with the ____.
Phosphate group … 3’ Carbon
Protein B has a lipid extension that interacts with fatty acid tails in the cell membrane. Is protein B an integral or peripheral membrane proteins?
Correct answer:
Integral
How many hydron bonds are between guanine and its correct base-pairing partner in DNA?
3
What part of the cell is not a part of the endomembrane system?
Mitochondria
Describe DNA
One turn of the DNA helix is about 3.4 nm
At normal body temperatures, cholesterol makes cell membranes
Less fluid
Which of the following statements about the plasma membrane of cells is correct?
None of the above
Very condensed chromatin can be found in all of the following, EXCEPT ____.
Euchromatin
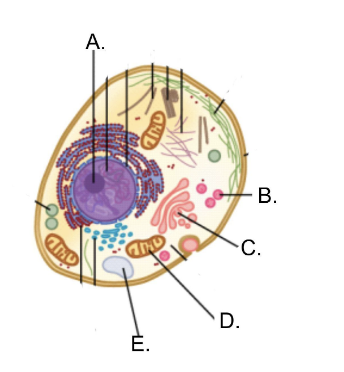
Label the parts of the cell:
A: Nucleolus
B: Lysosomes
C: Golgi Apparatus
D: Mitochondrion
E: Peroxisome
Cells shrivel when place in what kind of environments?
Hypertonic
What happens to the volume to surface area ratio in eukaryotes compared to prokaryotes?
Increases
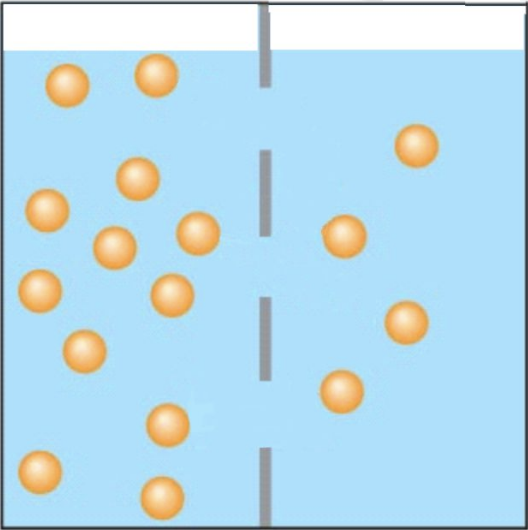
If only osmosis can occur in the pictured situation, what will move across the membrane and which way will it go?
The blue water will move to the left.
Who produced the x-ray diffraction data on the structure of DNA?
Rosalind Franklin
A saline solution has a concentration of 5mM. Is it hypotonic, hypertonic, or isotonic?
Cannot be determined with the given information
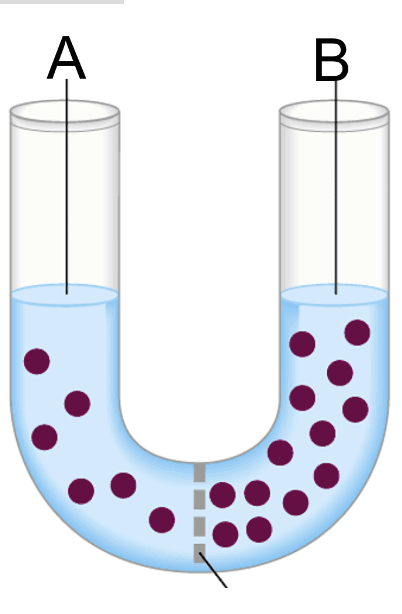
In the pictured situation, the solutes are the dots. Which side is the hypertonic solution?
B
Proteins make up how much mass of most plasma membranes?
50%
Name the three parts of a DNA nucleotide:
Nitrogenous Base, a 5 carbon sugar, phosphate group