Lab 2 Reducing Benzil Using Sodium Borohydride
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
What are the two main reducers
LiAlH4 and NaBH4. LiAlH4 reduces aldehydes, ketones, carboxylic acids, esters, nitriles, and amides while NaBH4 only reduces aldehydes and ketones.
what is the aqueous acid for in reductions
to free the product from a complex mixture. Also to protonate the O-
Why didn’t we use LiAlH4 in ether if it reduces everything?
The hydrogen atoms in LiAlH4 carry significant negative charge so they will react violently with any protic solvents such as OH and H2O to create flammable H2. So LiAlH4 must be used in an anhydrous solvent such as diethyl ether or tetrahydrofuran.
why did we use NaBH4
it will react very slowly with protic solvents so we can still use alcohol as a solvent. NaBH4 will decompose in the presence off acid groups so the acids first have to neutralized with sodium hydroxide.
how many molecules of a ketone or aldehyde can one reducing agent reduce
it can reduce 4 molecules of the ketone or the aldehyde as each reducing agent has 4 hydrogens it can use ti deliver the hydride ion. but this dependent on the number of carbonyls like if there are two carbonyls then only 2 molecules of the molecule will be reduced.
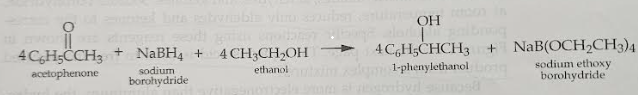
mechanism of the reduction
The carbonyl carbon will take the hydrogen from NaBH4 because it is so negative then the electrons will be pushed up to the O and it will be O-. Then the O will take the hydrogen from ethanol and then the ethanol will be negative so the it will attach the NaBH3 because that is positive.
what happens with the stereochemistry of the product?
If only one O is reduced then there will be (+)- benzoin and (-)- benzoin. If both carbonyls get reduced then there will be meso-hydrobenzoin or (+)-hydrobenzoin and (-)-hydrobenzoin. This is because the NaBH4 can attack from either side of the carbonyl.
how to distinguish between the different stereoisomers
The melting point of (±)-hydrobenxoin will have a melting point of 122-123. To distinguish between (±)- benzoin and meso-hydrobenzoin a TLC could be run, as the two alcohols in the meso compound will make it have a lower Rf value. The IR spectrum for benzoin would show a carbonyl peak whereas the meso-hydrobenzoin would only have alcohol peaks. The mixture melting point can be done by mixing some of the product with meso-hydrobenzoin and then recording the MP and then doing the same with (±)- benzoin as the melting point will be lower for the mixture that is incorrect.
What was the reaction
The carbon from the carbonyl in Benzil is attached by the H in NaBH4 and then the product is reprotonated with the ethanol.
diastereomers
stereoisomers that are not mirror images and have different physical properties. Like (±)-hydrobenzoin and meso-hydrobenzoin. like (±)- hydrobenzoin has a melting point at 122-123 and meso-hydrobenzoin has a melting point of 135-137.