Chemistry Mock Final
1/144
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
145 Terms
Atom (in terms of subatomic particles)
The smallest particle of an element that contains electrons surrounding a nucleus containing electrons and protons
Molecule (try hard version)
A group of two or more atoms chemically combined to form an identifiable unit which retains the properties and composition of the substance
Subatomic Particle Masses
Protons: 1
Electrons: 1/1840
Neutrons: 1
Atomic Number
number of proton in the nucleus of an atom
Mass number
number of neutrons and protons in the nucleus
Isotope
atoms of the same element, with the same number of protons but different number of neutrons
Relative atomic mass
The average mass of an atom of an element, taking into account the abundance of all its isotopes. With reference to 1/12th the mass of Carbon-12
Relative Atomic Mass formula
(% of isotope A x mass of isotope A) + (% of isotope B x mass of isotope B) / 100
Sub-atomic Particle Positions
Protons and Neutrons: in nucleus
Electrons: in shells
Sub-atomic Particles Relative Charges
Protons: +1
Electrons: -1
Neutrons: 0
Element
a substance made of atoms that all contain the same number of protons and can’t be split into anything simpler
there are 118 elements
ex: Copper, Iron, Arsenic
Atom
Smallest part of an element that has the element's properties.
Compound
a pure substance made of two or more different elements chemically bonded
∞ number of compounds
can’t be separated by physical means
ex: NaCl, MgO
Mixture
combination of two or more substances mixed but not chemically bonded together.
can be separated by physical means like filtration/evaporation
ex: sand and water, oil and water
Molecule
One or more element chemically combined
Heating Curve
Graph showing how a substance changes state when heated
Cooling Curve
A graph showing how a substance changes state when cooled.
Filtration
Used to separate insoluble solid impurities from a mixture.
Fractional Distillation: Use
Used to separate miscible substances with different boiling points
Can separate more than 2 substances
Miscible Liquids
Liquids that mix
They form one layer
Eg: ethanol and water
Immiscible Liquids
Liquids that don't mix
They form more than one layer
Eg: Oil and water
Properties of the compound vs Properties of the element it's made of
Different
Pure salt from Rock Salt
Filtration and Crystallisation
Rock salt = Pure salt + insoluble impurities
Filtration: separates the insoluble impurities from the mixture
Element, Compound, Mixture Diagram
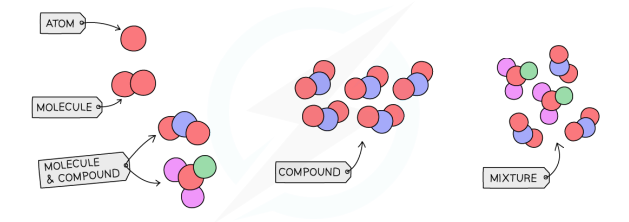
Pure Substance vs Mixture
Natural language: natural and clean, nothing added
Chemistry: pure substance = single element/compound with no other substances
ex: pure water has only H2O molecules
drinking water isn’t pure because it has additional substances like dissolved ions and chlorine
Distinguishing Purity
Pure substances melt and boil at specific temperatures ex, pure water m.p. = 0°C, b.p = 100°C
impure substances have a range of melting and boiling points because they consist of different substances
so, this data can be used to distinguish pure and impure substances
this also helps us assess the purity of drugs and foodstuffs
Assessing Purity: Process
using a melting point apparatus to heat a small portion of the sample and observe the exact melting point
compare to a data table
the closer the value is to the actual melting point, the purer the substance
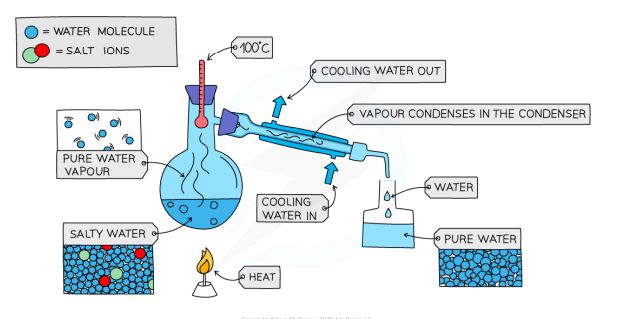
Simple Distillation: Process
separate a liquid and a soluble solid from a solution or a pure liquid from a mixture of liquids
the solution is heated, and the liquid evaporates
the vapour rises through the neck of the round-bottomed flask
the vapour passes through the condenser, where it condenses and cools to a pure liquid that is collected in a beaker
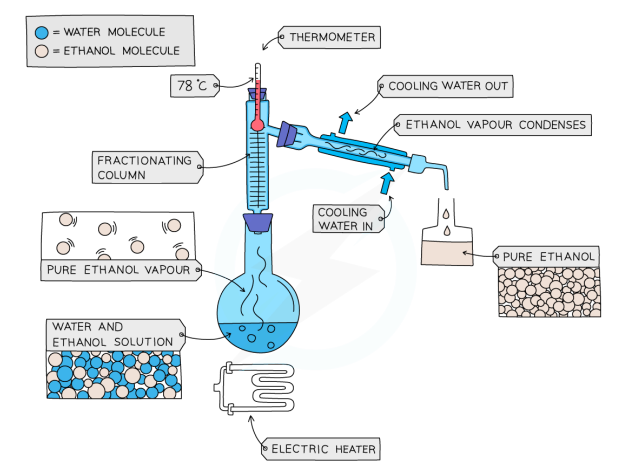
Fractional Distillation
used to separate two or more miscible liquids
solution is heated to the temperature of the substance with the lowest m.p
this substance will evaporate first, and vapours will pass through a condenser
the liquid will be condensed and collected in a beaker
once all of the substance is collected, the other liquid will be left behind
Fractional Distillation of Ethanol and Water
Ethanol b.p = 78 °C, Water b.p = 100 °C
mixture is heated until it reaches 78 c, and the ethanol boils and distills out of the mixture and condenses into the beaker
when the temperature starts to increase to 100 °C, heating should be stopped. Water and ethanol are now separated
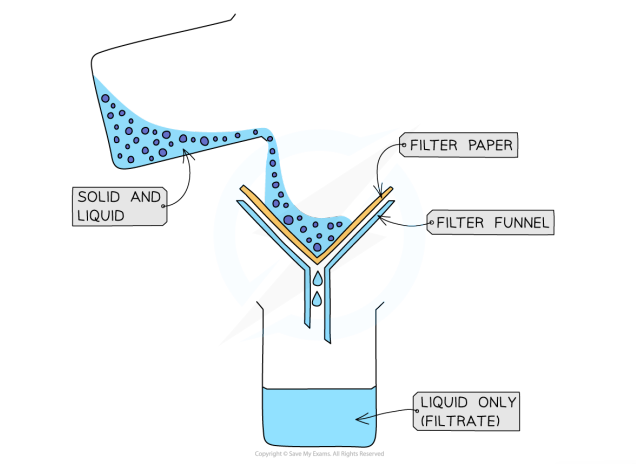
Filtration: Process
used to separate undissolved solids from a mixture of solids and a liquid (centrifugation can also be used for this mixture)
filter paper is placed in a filter funnel above a beaker
the mixture is poured in
the filter paper will only allow small liquid particles to pass through as filtrate
solid particles are too large, so the stay behind as filtrate
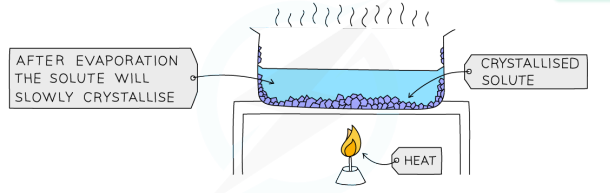
Crystalllisation
used to separate a dissolved solid from a solution, when the solute is more soluble in hot solvent than cold
solution is heated, allowing the solvent to evaporate and leave a saturated solution behind
test if the solution is saturated by dipping a glass rod into the solution (if the solution is saturated, crystals will form on the glass rod)
saturated solution will cool slowly
crystals will grow as solids come out of the solution due to decreasing solubility
crystals are collected by filtering, they are washed with cold distilled water to remove impurities and then allowed to dry
Paper Chromatography: Process
used to separate substances with different solubilities in a given solvent
a pencil line drawn on chromatography paper, and spots of the sample are placed on it (pencil is used as ink would run into the chromatogram along with the samples)
the paper is then lowered into the solvent container (the line must be above solvent line so samples don’t wash into the solvent container)
solvent travels up the paper by capillary action, taking the coloured substances with it
different substances have different solubilities, so will travel at different rates (this causes the substances to separate, higher solubility = travel further)
this will show the different components of the ink/dye
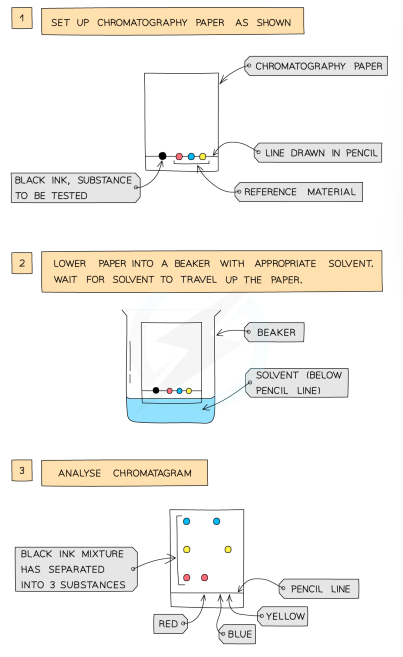
Interpreting Chromatograms
pure substances = only one spot
impure substance = multiple spots
same substance = identical chromatograms
mixture = separates to show different components as separate spots
use a known compound to identify spots
Rf Values
used to identify components of mixtures
always the same for a particular compound
solvent changed = Rf value changes
allows to identify compounds because it can be compared with known values
Rf = distance moved by substance / distance moved by solvent
Law of Conservation of Mass
Total mass of reactants is always equal to total mass of products
RFM Calculation
multiply each Ar by how much there is
add them together
Avogadro’s Number
6.023 × 1023
Moles and Mass
n = M/Mr
Calculating Molar Mass/ no. of moles
calculate Mr
divide mass given by Mr
Calculating Reacting Mass
find reacting moles (given or calculate with M / Mr)
find ratio between given substance and substance to find
find no. of moles
use no. of moles to find mass
Balancing Equations using Reacting Mass
write unbalanced equation
write down masses
calculate moles using mass and Mr
use moles to find ratio
use ratio to balance equation
Reasons for not getting 100% yield in a reaction
some reactants left behind
reaction may be reversible- high yield is impossible since products are continually turning back into reactants
some products lost during purification or separation stages like filtration or distillation
side reactions: substances reacting with gas in the air or impurity in reactant
products lost during transfer between containers
Thoretical Yield
amount of product that would be obtained under perfect practical and chemical conditions
calculated from balanced equation and the reacting masses
Actual Yield
recorded amount of product obtained
always less than theoretical yield
Percentage Yield: Purpose
compares actual yield to theoretical
for economic reasons, objective of every company is to have yield % as high as possible → to reduce costs & wastes and increase profits
good way to measure how successful a chemical process is
Calculating Percentage Yield
find actual yield (usually given)
find theoretical yield (calculate using moles and mass)
use equation: (actual/theoretical) x 100
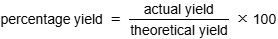
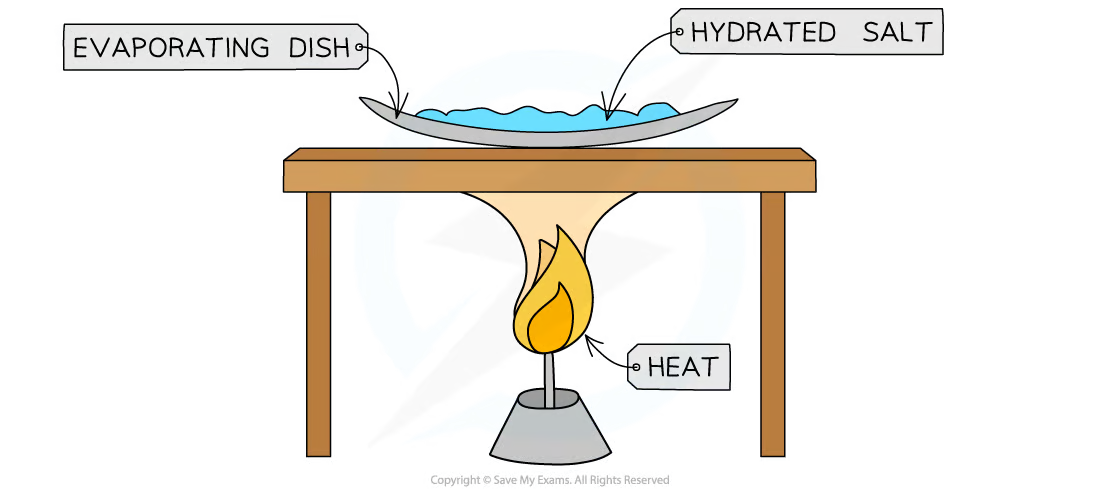
Experiment (Finding Formulae of Simple Compounds): Aim
to determine formula of hydrated copper sulfate: CuSO4.xH2O
Experiment (Finding Formulae of Simple Compounds): Method
measure mass of evaporating dish
add known mass of hydrated salt
heat over bunsen burner, gently stirring
stop when salt turns from blue to white (all water lost)
record mass of dish and contents
Experiment (Finding Formulae of Simple Compounds): Overheating the salt
decomposes and gives a larger change in mass
Experiment (Finding Formulae of Simple Compounds): Results
mass of white anhydrous salt
measure mass of white anhydrous salt (mass of salt remaining)
mass of water
subtract mass of white anhydrous salt from mass of known hydrated salt
divide mass of the salt and water by relative masses (find moles)
simplify the ratio (multiply by 2 if decimal)
find ratio as 1:water
represent ratio as ‘salt.xH2O)
Practical (Determining Formula of Magnesium Oxide): Aim
To determine the empirical formula of magnesium oxide by combustion of magnesium
Practical (Determining Formula of Magnesium Oxide): Diagram
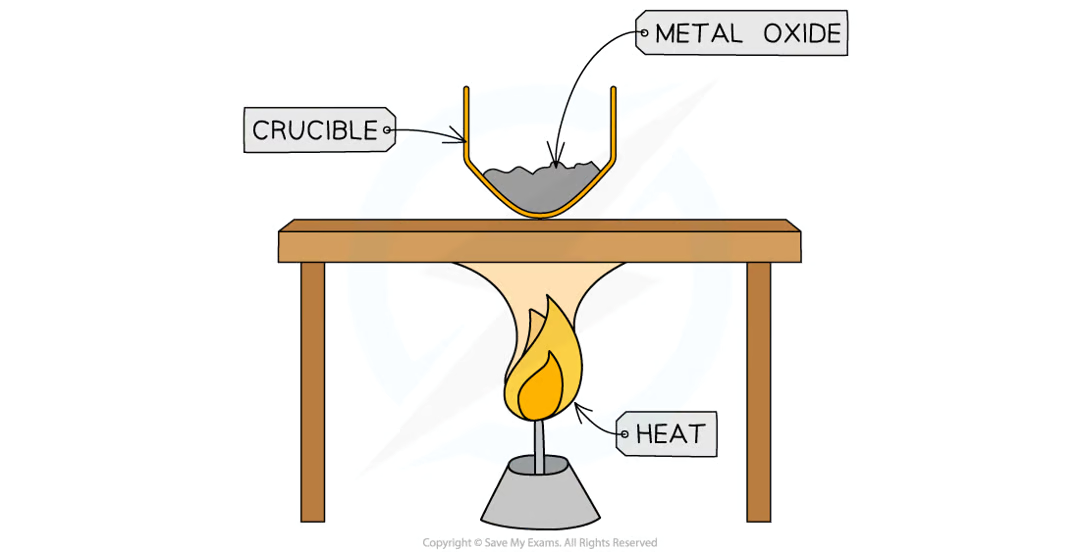
Practical (Determining Formula of Magnesium Oxide): Method
measure mass of crucible with lid
add Mg sample to crucible and measure mass with lid
find mass of Mg
strongly heat crucible over Bunsen burner for several minutes
lift lid frequently to allow sufficient air into crucible for Mg to oxidise without letting MgO smoke escape
continue heating until mass remains constant (max mass) → reaction is complete
measure mass of crucible and contents
calculate mass of crucible and contents by subtracting mass of empty crucible
Practical (Determining Formula of Magnesium Oxide): Results
find mass of metal by subtracting mass of crucible from Mg and mass of empty crucible
subtract mass of Mg used from mass of MgO
divide each mass by Ar,
simplify ratio (multiply by 2 if decimal)
represent as MxOy
Practical (Determining Formula of Copper(II) Oxide): Aim
To determine the formula of copper(II)oxide by reduction with methane
Practical (Determining Formula of Copper(II) Oxide): Diagram

Practical (Determining Formula of Copper(II) Oxide): Method
measure mass of empty boiling tube
place metal oxide into a horizontal boiling tube and measure mass again
support tube in horizontal position by clamp
natural gas(methane) is passed over copper(II) oxide and excess gas is burned off
copper(II) oxide is heated strongly with a Bunsen burner
heat until metal oxide fully changes colour (all oxygen removed)
measure mass of remaining powder in the tube and subtract mass of tube
Practical (Determining Formula of Copper(II) Oxide): Results (Empirical Formula)
measure mass of powder to find mass of metal
divide masses by Ar
simplify ratio
represent ratio as MxOy
Molecular Formula: Definition
Formula showing number and type of each atom in a molecule. Ex: ethanoic acid is C2H4O2
Empirical Formula: Definition
Simplest whole number ratio of atoms of each element present in one molecule or formula unit of the compound. Ex: ethanoic acid is CH2O
Ionic compounds are always _________ formulae
Empirical
Calculating Empirical Formulae
write element
write value given (% or mass)
write Ar
calculate moles by m/Mr
calculate ratio of moles (multiply to make all values whole numbers)
write final empirical formula
Calculating Molecular Formula
find Mr of empirical formula (add masses of all atoms in the empirical formula
divide Mr of molecular formula by Mr of empirical formula
multiply each number in empirical formula by answer to find molecular formula
Calculating Concentration of Solutions in mol/dm3
number of moles (mol) / volume (dm3)
Avogadro’s Law
At the same conditions of temperature and pressure, equal amounts of gases will occupy the same volume of space
Molar Gas Volume at RTP
24dm3 or 24000 cm3
RTP
room temperature and pressure (20oC and 1atm)
Gas Volume Equations
volume = moles x 24 (dm3/mol)
volume = moles x 24000 (cm3/mol)
Metal + Water Reaction Format
metal + water —→ hydroxide + hydrogen gas
Metals and Acids Reaction Rate
only metals above Hydrogen
more reactive metal = more vigourous reaction
K and Na are very dangerous and react explosively
Metal + Acid Reaction Format
Metal + Acid → Salt + Hydrogen
Solid
regular arrangement
low kinetic energy
vibrate in fixed positions
strong forces of attraction
Liquid
arranged close together
more kinetic energy than solid particles
move past each other
weak forces of attraction
Gas
arranged far apart
a lot of kinetic energy
move randomly in all directions
very weak forces of attraction
Evaporation
liquid turns into a gas
occurs at any temperature
faster, because it is a surface level process
Boiling
liquid turns into a gas
only at a certain temperature
slower, because all the particles need to overcome forces of attraction
Diffusion
Spreading out of particles from high concentration to lower concentration until there is equal concentration throughout.
Diffusion: temperature effect
Increases with increase in temperature
Because when particles are heated, they gain kinetic energy and move around more
Bromine Experiment (and Hydrogen variant)
Lower jar: Bromine gas
Top jar: air
When lids are removed, bromine diffuses upwards
Air also diffuses downwards until both jars are uniformly brown
Hydrogen variant
place a lighted splint to check
Expected: sound from top jar because H is less dense
But equal sounds from both jars
Melting
solid to liquid
heat energy absorbed transformed to kinetic energy
happens at a specific temperature or melting point (m.p.)
Freezing
liquid to solid
reverse of melting, happens at (m.p.)
needs a significant decrease in temperature and loss of thermal energy
Sublimation
solid to gas directly
very few solids, eg: iodine, carbon dioxide
reverse reaction: desublimation, deposition
Potassium Manganate (VII) Experiment
Description
when crystals are dissolved, a purple solution is formed
a small number of crystals produce a highly intense colour
Explanation
water and potassium manganate (VII) particles move randomly and slide across each other
therefore mix easily
diffusion in liquids is slower than in gases because particles are closer and move more slowly
NH3 + HCl Reaction
particles diffuse along the tube
white ring of NH4Cl forms where they meet
ring forms closer to HCl end
because Ammonia particles are lighter and move faster
so they travel further in the same amount of time, and further away from the NH3 end
Dilution of Potassium Magnate (VII)
Description
solution can be diluted several times
colour fades but doesn’t disappear until after many dilutions
Explanation
indicates that there a lot of particles in a small amount of potassium magnate (VII) and so the particles are very small
Solvent
liquid in which a solute dissolves
eg: water in salt water
Solute
substance that dissolves in a solvent to become a solution
eg: salt in salt water
Solution
mixture formed when a solute is dissolved in a solvent
eg: sea water
Saturated solution
a solution with the max. conc. of solute dissolved in the solvent
eg: sea water in the Dead Sea
Soluble
describes a substance that will dissolve
eg: salt is soluble in water
Insoluble
describes a substance that will not dissolve
eg: sand is insoluble in water
Solubility
measurement of how much of a substance will dissolve in a given volume of a liquid
Solubility of Solids & Temperature
temperature increase = solubility increases
Solubility of Gases (Temperature and Pressure)
pressure increase = more soluble
temperature increases = less soluble
Solubility Practical
pour tap water into a beaker
heat to a specific temperature
add solute one spatula at a time, with constant stirring until no more dissolves and some remains undissolved in the mixture
monitor temperature and keep it uniform throughout
record mass of empty evaporating basin
filter the mixture of of solution and undissolved crystals into the evaporating basin
evaporate the filtrate until dry crystals are formed
record the mass of the evaporating basin with the dry crystals
Groups
vertical columns
number of electrons in the outermost shell
1-7(0)
Period
horizontal rows
show the number of shells
1-7
Electronic Configuration in Shells
first shell: 2
second shell: 8
third shell: 8
Metals vs Non Metals (Valence Electrons)
Metal: 1-3 valence electrons
Non-metal: 4-7 valence electrons