Chemistry: Matter, Properties, Changes, and Atomic Structure
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
What is chemistry?
The study of composition, structure, properties, and reactions of matter.
Define matter.
Anything that has mass and occupies space.
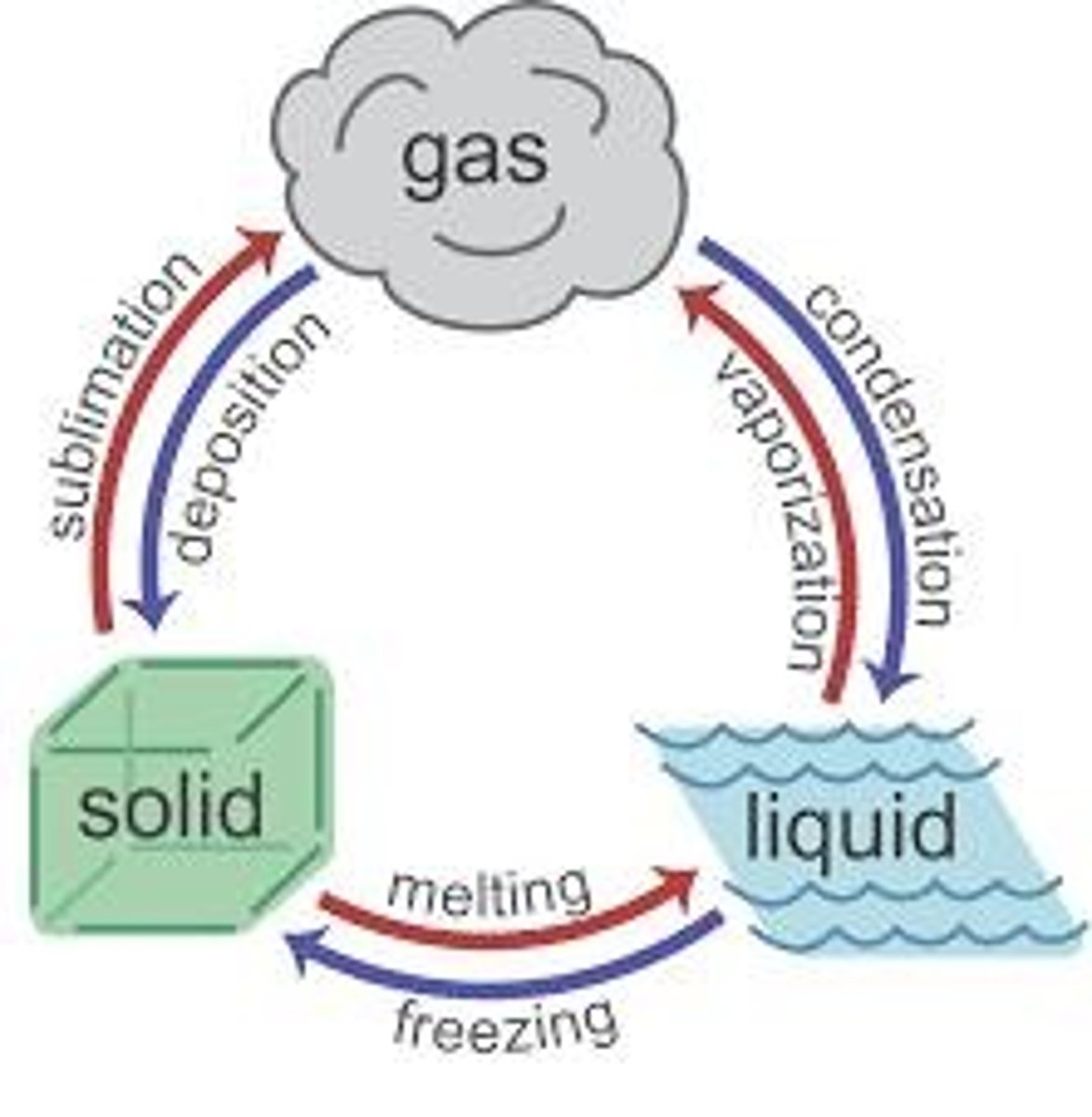
What are the three states of matter?
Solid, liquid, and gas.
Describe the characteristics of solids.
Strong attractive forces keep particles close together, with a definite shape and volume.
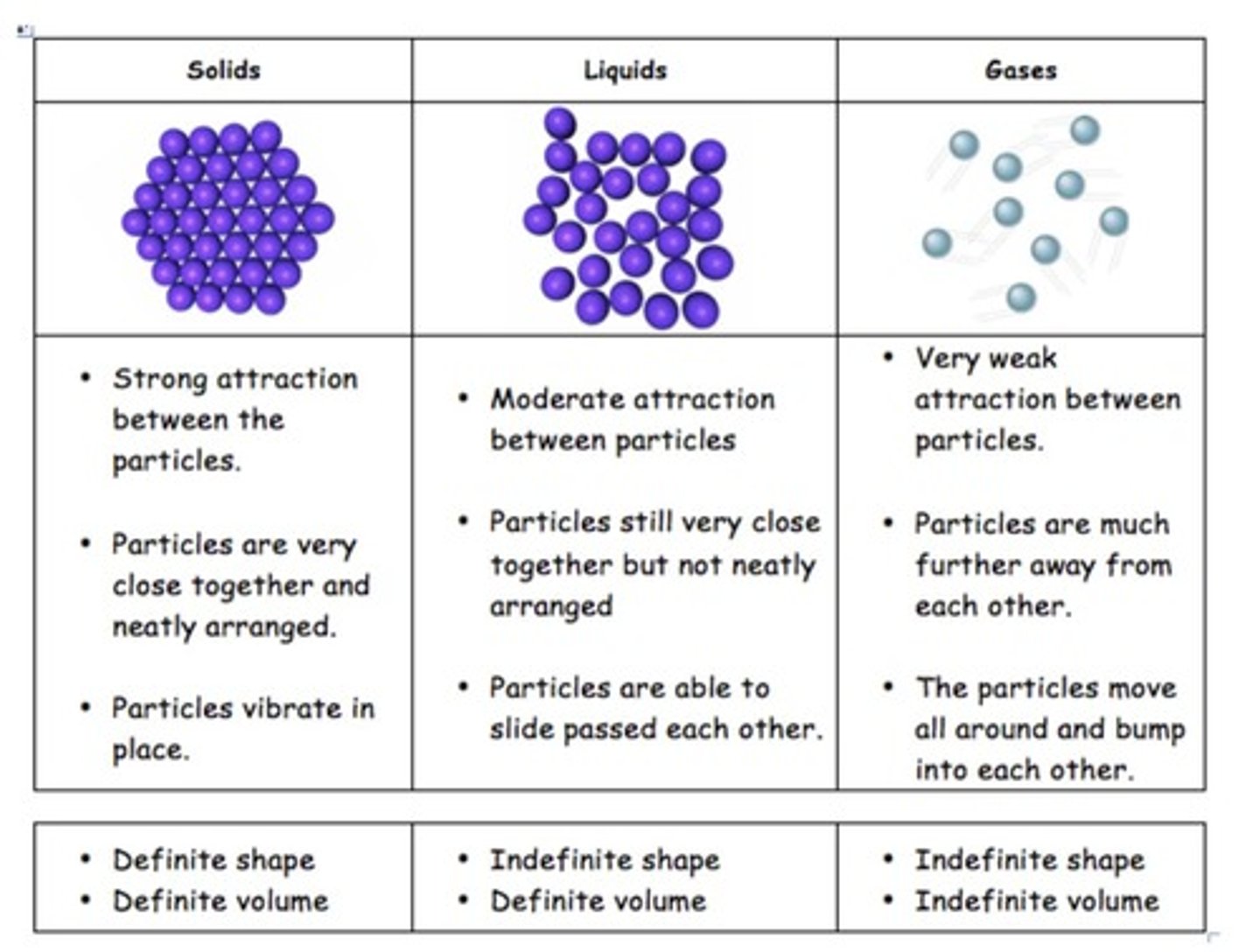
What defines a liquid?
Particles can move freely but stay close together, maintaining a definite volume without a rigid structure.
What are the properties of gases?
Molecules move fast with great distances between them, having no definite shape or volume.
What factors affect the states of matter?
Temperature, pressure, and the strength of forces holding particles together.
What is a distinguishing characteristic of a substance?
A property used in its identification and description.
What are the two types of properties of matter?
Physical properties and chemical properties.
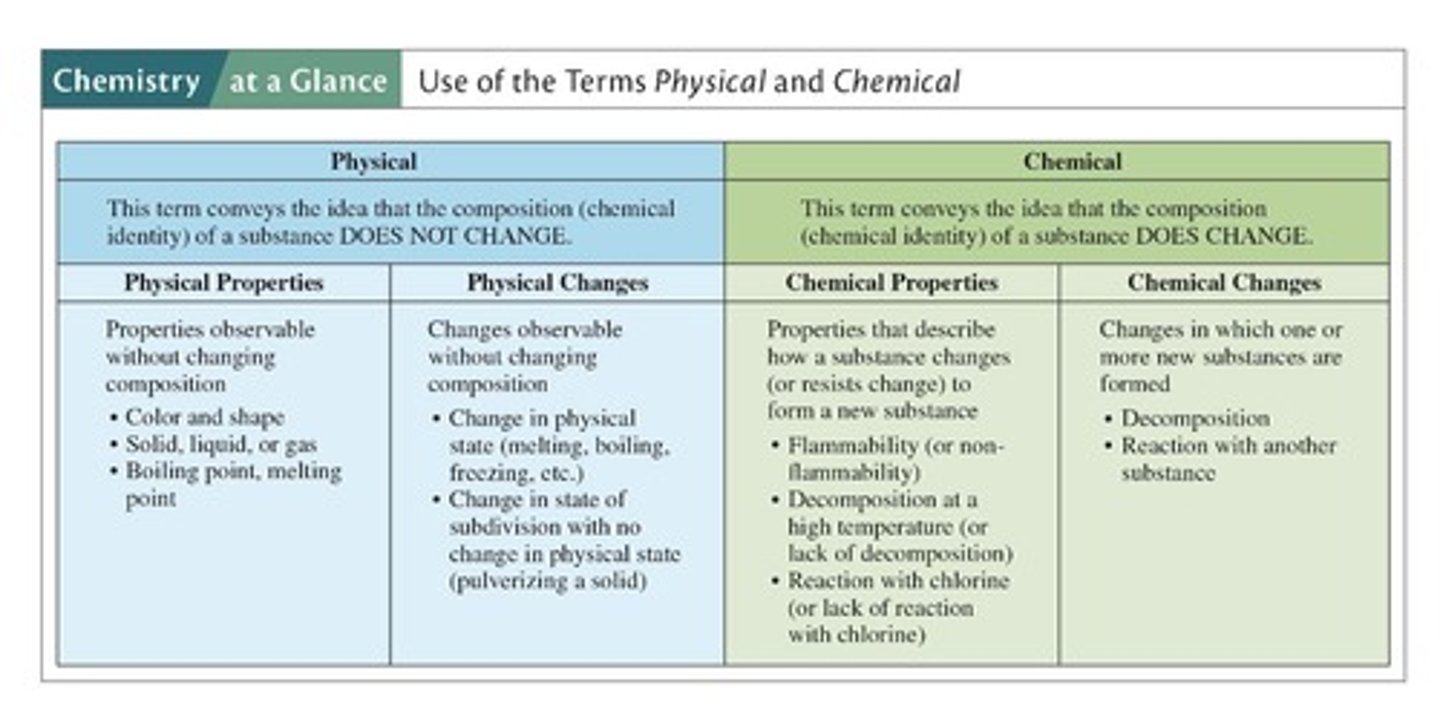
What are physical properties?
Characteristics observed or measured without changing the sample's composition.
Give examples of qualitative physical properties.
Color, odor, conductivity, hardness, solubility.
Give examples of quantitative physical properties.
Density, mass, melting point, boiling point.
What are chemical properties?
Characteristics displayed as a sample undergoes a change in composition.
What is a physical change?
An alteration in appearance without changing the composition of the substance.
What is a chemical change?
A change where reacting substances form new substances with different properties.
What happens to energy during physical or chemical changes?
Matter gains or releases energy.
What is energy?
The ability to do work, classified as kinetic or potential.
What is a pure substance?
Matter consisting of only one type of atom or molecule with distinct physical properties.
What are the classifications of pure substances?
Elements and compounds.
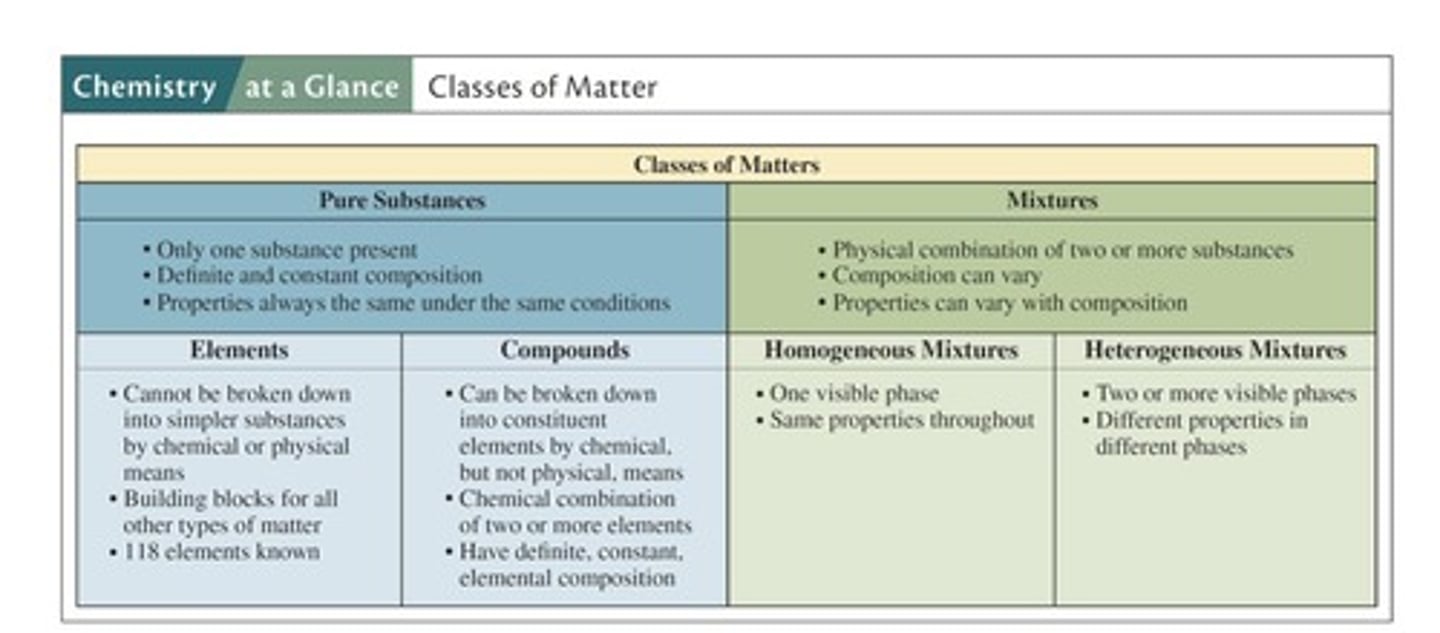
Define an element.
A pure substance composed of only one type of atom that cannot be broken down further.
What is a compound?
A pure substance formed from two or more different types of atoms chemically bonded together.
What is an example of a physical change?
Boiling or freezing water.
What is an example of a chemical change?
Burning a match or candle.
What is the significance of temperature and pressure in states of matter?
They determine the state in which a substance exists at Earth conditions.
What does it mean for a substance to have a definite shape?
It retains its shape regardless of the container it is in.
What does it mean for a substance to have a definite volume?
It occupies a fixed amount of space regardless of the container.
What are the three types of elements?
Metals, nonmetals, and metalloids.
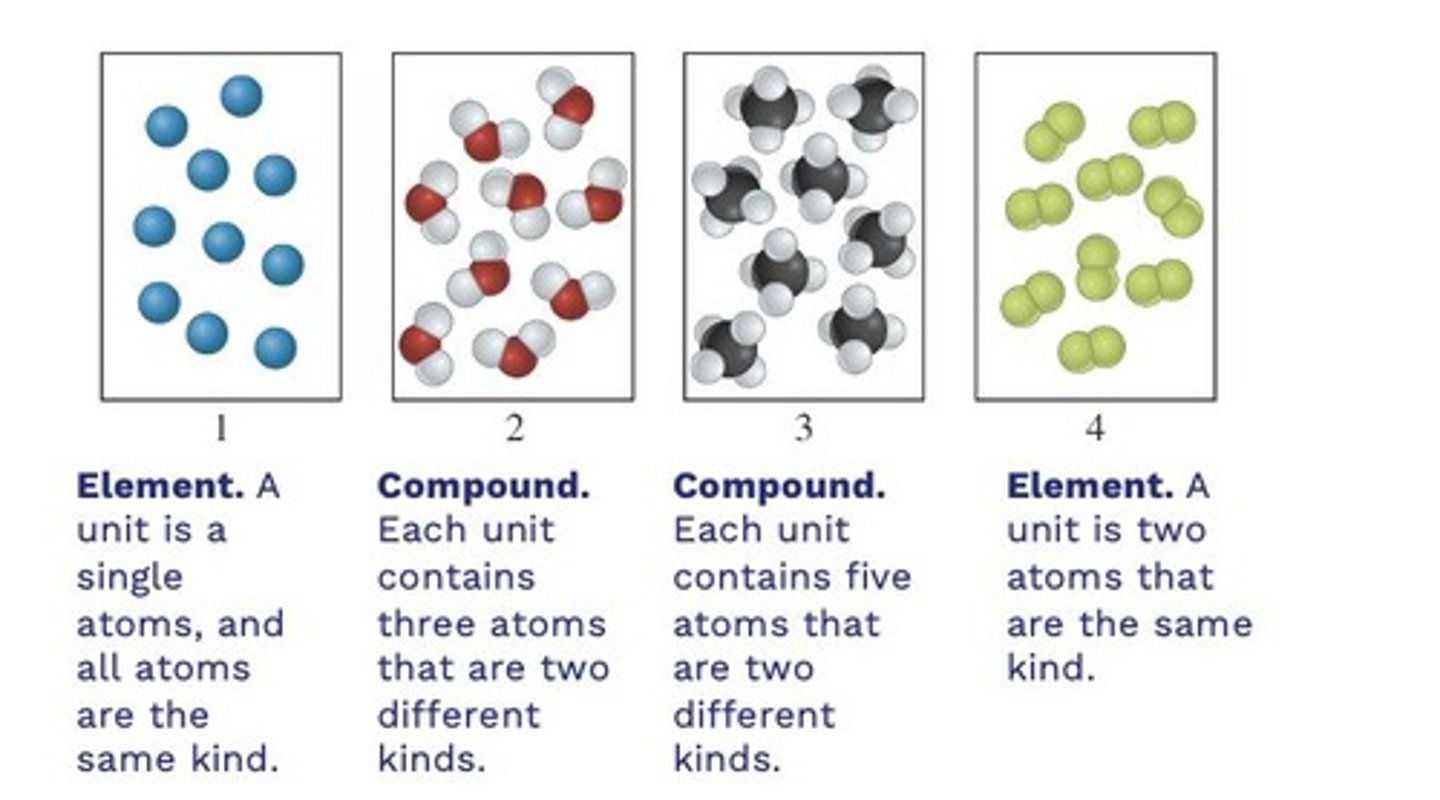
Provide an example of a compound.
Water (H2O), Table salt (NaCl), Sugar (C12H22O12).
What is a mixture?
Two or more substances that are physically mixed but not chemically combined.
What are the two classifications of mixtures?
Homogeneous and heterogeneous mixtures.
Define a homogeneous mixture.
A mixture with the same composition and properties uniformly throughout the sample.
Provide an example of a homogeneous mixture.
Salt water, brass (zinc + copper), air.
Define a heterogeneous mixture.
A mixture where the components do not have a uniform composition and can be visually distinguished.
Provide an example of a heterogeneous mixture.
Pizza, water and sand.
What is the total number of known elements?
118 known elements.
How many naturally occurring elements are there?
92 naturally occurring elements.
What major elements make up 80% of the atoms on Earth?
Oxygen and silicon.
What is the elemental composition of the human body?
60.5% hydrogen, 25.7% oxygen, 10.7% carbon, 2.4% nitrogen, and 0.7% all others.
What are chemical symbols?
One or two letter abbreviations for the names of the elements.
What is the chemical symbol for sodium?
Na.
What is the chemical symbol for chlorine?
Cl.
What is a diatomic molecule?
A molecule composed of two atoms.
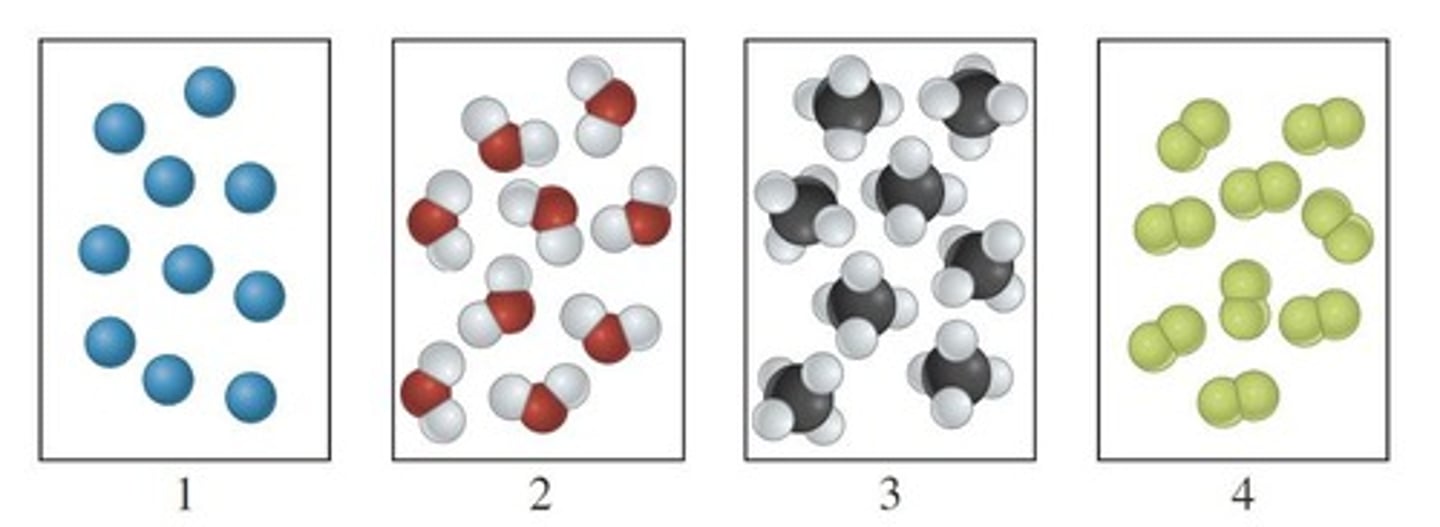
What is a triatomic molecule?
A molecule composed of three atoms.
What is a polyatomic molecule?
A molecule composed of two or more atoms.
What is a chemical formula?
A notation made up of the symbols of the elements present in a compound and numerical subscripts indicating the number of atoms of each element.
What does the chemical formula C6H12O6 represent?
It represents a molecule with 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms.
What is the significance of parentheses in a chemical formula?
Parentheses indicate groups of atoms that need to stay together in polyatomic entities.
What does Ca(NO3)2 represent?
It represents 1 atom of Ca, 2 atoms of N, and 6 atoms of O.