Presentations day 1
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
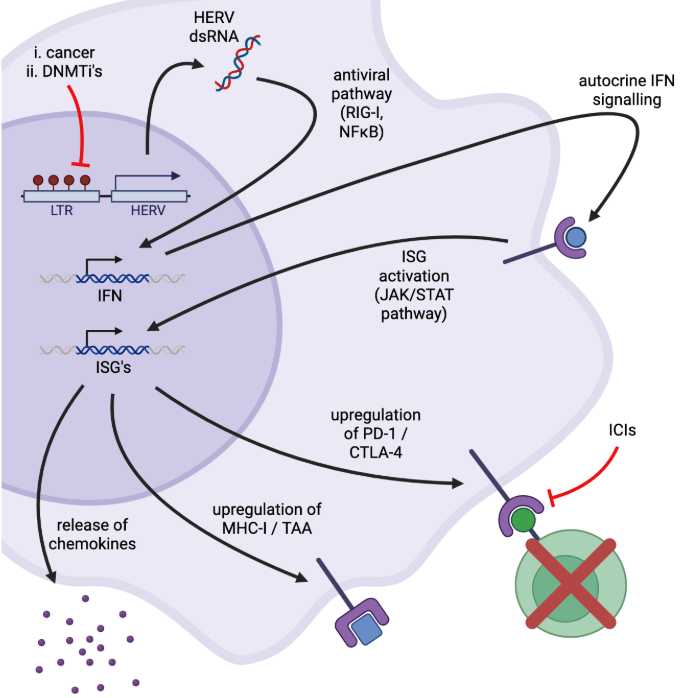
what are HERVs?
human endogenous retroviruses → make up 8% of human genome
mutations/recomb b/w LTRs → loss of replicability → 10% due to buildup of mutations, 90% LTR leaving (middle lost)
why aren’t HERVs constitutively expressed?
silencing via epigenetic modifications → to prevent expression of viral promoters
methylation of CpG (cysteine, guaning) islands → histone recruitment
works with histone deacetylase to clump DNA to prevent expression
when many methyl groups added to CpG repeats, steric hinderance blocks gene replication
how do HERV’s act as a tumor marker and what does that have to do with ‘viral mimicry’?
certain cancers (eg. seminoma) interrupt CpG methylation processes → relief of HERV suppression in cancer cells = increased HERV expression → decreased methylation = increased HERV exp
HERV expression leads to antiviral response
dsRNA → IFN → ISGs
shown by RNAseq
what are the effects of increased ISG expression with HERVs?
upregulation of MHC I /TAA → good
release of chemokines → good
upregulation of PD-L1/CTLA-4 → not good, decreases T cell activity
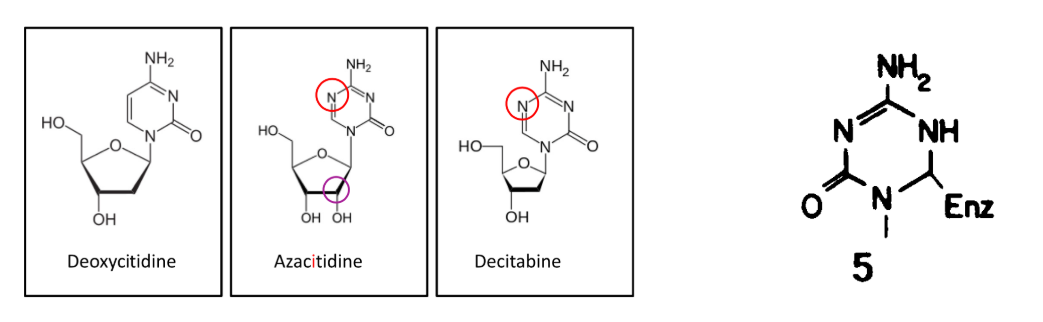
what are DNMTi drugs?
DNA methyltransferase inhibitors
cytosine analogues
covalently ‘trap’ DNMT at CpG dinucleotides at the replication fork → trap enzyme, blocking further activity → effective at low doses
enzyme normally allows switch from proton to electron → nucleophilic attack, methyl attaches
drug has N where C would usually be that would be methylated → when enzyme attacks N gets covalently trapped
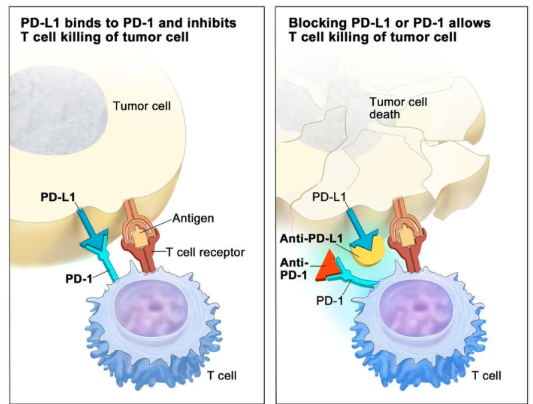
describe ICIs as cancer therapies
‘immune checkpoint inhibitors’ relieve inhibition of T cells by tumor cells
target inhibitory immune checkpoint receptors on tumor cells → PD-L1, CTLA-4)
can also target ligands on T cells → PD-1, CD80/86
small antibody mol → bind to receptors, block signalling
how could HERVs and the ICIs be combined?
use DNMTis alongside ICIs → inhibiting methylation would theorhetically increase HERV expression → increase HERV get increase PD-L1 → more targets for ICI → ICI more efficient
saw decreased tumor burden when combined, but doesn’t directly show due to HERVs specifically
what are the promising features and challenges with exploiting HERVs to improve immunotherapy efficacy
promising features:
reversal of T cell exclusionary cancerous phenotypes (‘cold’ tumors → ‘hot tumors’)
DNMTi ‘priming’ restores responsiveness to aPD-1 therapy → allow histones chromatin to break apart & activate path?
many clinical trials underway
challenges:
variable results between cancer types and patients → blocked IFN prod = blocked DNMTi action. immunosuppression = impaired action
neutralization of ‘good’ viral elements → cleared by IS
chronic antiviral signalling = immunosuppressive
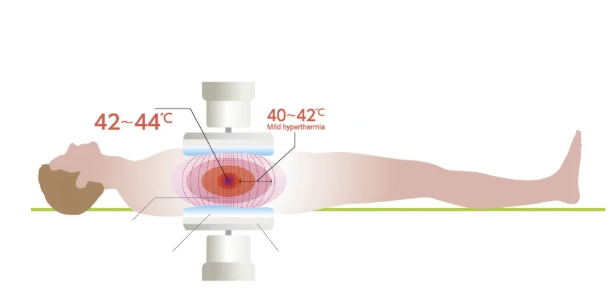
what is hyperthermia cancer therapy?
hyperthermia = increasing temp → 39-44C
increases tumour susceptibility → cooks the cell
more targeted chemotherapy → thermoresponsive liposomes
specificity can be improved
what virus is used as part of the hyperthermia presentation?
macrobrachium rosenbergii nodavirus → using VLP
freshwater prawn virus
capsid protein can form VLPs
capsid stability forms in association with RNA → like HBV, need stable assoc.
many chemo drugs bind RNA → can bind chemo drug to RNA then form capsid around it
how did they target cancer cells in the heat-seeking VLP treatment
folate receptor is overexpressed in ~40% of cancers, as folate is required for dNTP (purine) synthesis
add folic acid to the surface of VLP to act as receptor-binding protein
describe how the heat-seaking VLP treatment works
add folic acid to surface of MrNVLP → package Dox, a nucleic acid chelating agent, inside by binding incorporated E. coli RNA → destabilize Dox from virus only when heat is added
showed temp sensitive
showed VLPs are specific for folate receptors → VLPs more specific for cells expressing high levels of folate receptors
what are the promising features of MrNVLP-Dox?
no seroprevalence → freshwater prawn virus
no/limited Ab neutralization → folate (aka vitamin B12) covers whole VLP so can’t access capsid
many chemotherapies can bind RNA stably
can easily add diff receptors for diff cancers
lowers risk of off-target effects
potential Ag presentation on tumors of VLP
immune activation from RNA → E. coli RNA interact w/ PRRs
what are the challenges associated with MrNVLP-Dox
no in vivo experiments shown
could make it easier for Dox to enter healthy cells → female reproductive tissues increase expression during shedding cycle, macrophages express it too
Dox can slowly release from VLP at 37C → likely require intratumoral administration
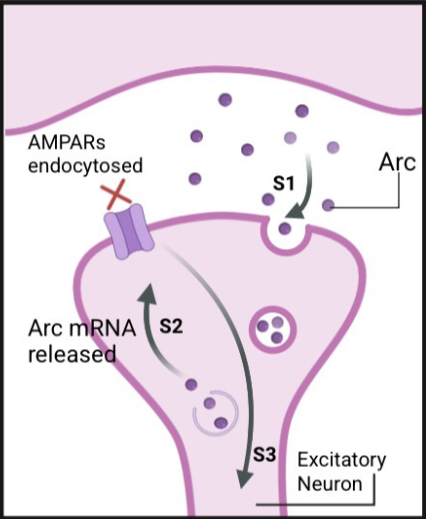
what does Arc do?
downregulates synaptic signalling
neuronal protein expressed in response to synaptic activity
enriched at excitatory synapses
Arc capsids may be required to eliminate synaptic material
required for learning and memory processes
Arc capsids endocytosed, Arc mRNA released, binds AMPARs, which get endocytosed and dampens activity of neuron
describe Arc’s N lobe
Arc’s N-terminal lobe has a:
hydrophobic pocket
Beta1 (B1) strand
B1 mediates binding to synaptic proteins
Stargazin → AMPA receptor trafficking, synapitc regulation
where does Arc originate from?
Ty3/gypsy retrotransposon
encodes protein similar to gag’s capsid (CA) protein → related to retroviruses
domestication of retrotransposon-derived gene → lost ability to form infectious particle (lost zinc knuckles), was repurposed
how does Arc compare to HIV protein?
Arc superimposes on Gag’s CA domain → HIV C-lobe
function: self-assembly into capsid, RNA packaging, intracellular delivery
but sequences aren’t super similar → but residues forming hydrophobic pockekt are conserved
why is there a need for studying Arc?
implicated in cognitive diseases → schizophrenia, autism, alzheimers
understanding its action
binding properties
therapeutic potential to modulate: synaptic plasticity, memory pathways
novel mechanism of intracellular communication → sharing of genetic material
generally describe how Arc is hypothesized to function
Arc self-assembles into viral-like capsids in the donor neuron
Arc mRNA packed into capsid
which are released from donor cells in extracellular vesicles dubbed ACBARs
mRNA released into recipient neuron to be translated and continue down path to inhibit neuronal activity
what/how did they show Arc’s functions?
showed Arc self-assembles into virus-like capsids using its full gag-related genome → CA domain not sufficient for assembly, self-assemble spontaneously
Arc capsid can transfer Arc mRNA between neurons → incubated hippocampal neurons from Arc KO mice with purified rat Arc → measured Arc mRNA levels in Arc KO mice using Arc FISH, then incubated with prArc
what makes Arc work/knowledge promising?
a tool to map memory engrams (in use)
tightly linked to neuronal activity during learning
marks neurons involved in specific experiences
a neuron-specific RNA delivery system → endogenous
targeting Arc to treat cognitive disorders
Arc dysregulation → synaptic dysfunction?
pharmacological modulation
what makes Arc work challenging?
risk of triggering immune response in brain
oligomeric particles, highly repetitive & symmetric
package RNA → TLR → inflammation in brain → neural degeneration"?
delivery beyond neural circuits
cargo specificity
targeting
high scale production?
what is the current problem with how we trace neural circuits? what do we need?
brain has 100 billion neurons and 1 quadrillion synapses → very complex system
need a tool that can:
trace connections across synapses
identify directionality
label specific neurons
link structure to function
compare non-viral vs viral tracers
non-viral: chemical, eg. silver stain → stains well but can’t distingush individual synapses
rely on injection
label non-specifically
label “axons of passage” → false positives, intermingled rather than connections
no directional control
viral tracers:
cell type specificity
can separate from intermingled neurons
trans-synaptic labelling
directional control
why is rabies a good candidate for neural tracing?
enveloped & bullet shaped
easy to genetically engineer envelope proteins
bullet structure → efficient travel across long distances
retrograde & neurotropic
moves backwards from synapse towards cell body → exclusive directional control
naturally infects neurons
trans-synaptic spread
specific spread across junctions, rather than non-specific diffusion
immune evasion
can persist in body without being cleared
describe the first gen rabies neural tracers
had deletions in the envelope glycoprotein → G protein is key to entering cell, without it won’t spread well
problems:
cytotoxic
kill neurons within 2 weeks
impacts endogenous gene expression
short-term, static visualization
describe the second gen rabies neural tracers
deletion of the glycoprotein and large polymerase gene → don’t spread, don’t replicate
features:
90% neuron survival
preserved physiological integrity
long term imagine
regulatable viral replication
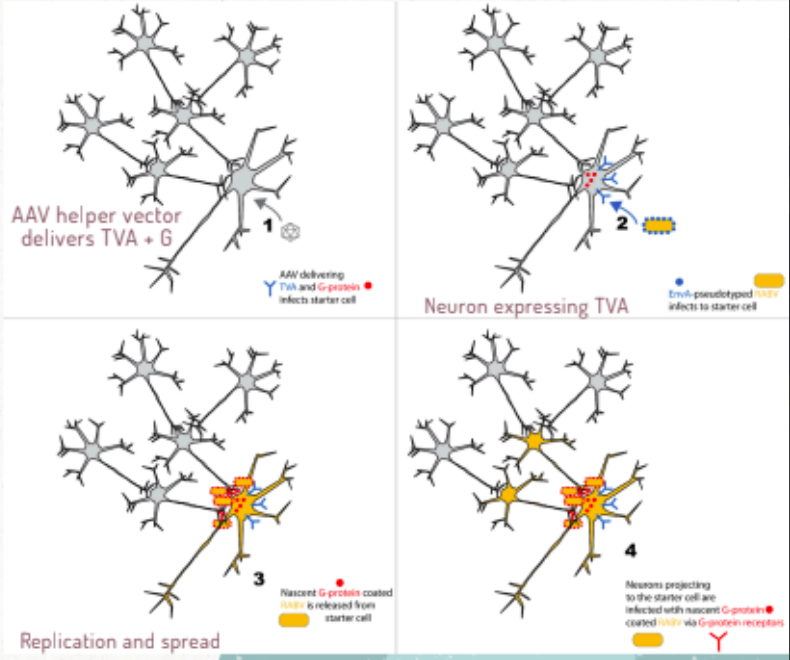
describe the machanism behind the rabies neural tracing
can precisely target cells using the EnvA and TVA system:
pseudotype rabies with EnvA
mammalian neurons lack TVA recepto
starter cells genetically engineered to express TVA + G protein via AAV vector → prgeny rabies goated with G protein and can move to neighbouring cells
infection becomes cell-type specific
what results were shown for the rabies neural tracing?
first gen is fast but toxic, no source cells left
very few labelled, initially, after 5 weks label appears in thalamus, secondary motor cortex, somatosensory, doxycycline suppresses G and L → turns off viral replication and spread
showed that corticostratial neurons are integrating inputs from opposite cortex an thalamus → evidence of cross-tall
what are the applications of the rabies neural circuit tracing
identified the presynaptic inputs to dopamine neurons in the ventral tegmental area in mice → region involved in reward and motivation
found that VTA dopamine neurons receive input from over 20 brain regions
map spacial organizations
what are the challenges with rabies neural circuit tracing?
neurotoxicity → limits range of species that can be tested
possible synapse bias → may preferentially infect inhibitory vs excitatory neurons, study using first gen showed no bias but no studies done on second gen
possible transport limitations → distant neurons may not be getting labeled, slow or incomplete viral transport across long distances
what are the promising features of the rabies neural circuit tracing
3rd gen tracer → some studies being done with dL version, even better labelling
gene manipulation → design inhibitors/agonists that may manipulate the behaviour of infected neurons
neuron studies → could monitor change in neural connections over time, what areas degrade faster, brain injury studies, smell/scent studies…