Naming organic compounds
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
What is the main part of the names of all organic compounds?
The root
What does the root tell us?
The length of the longest unbranched hydrocarbon chain
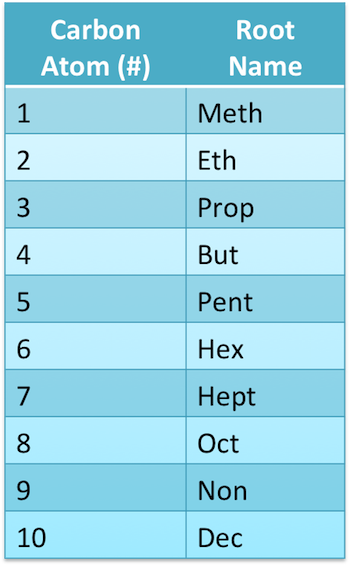
For simple molecules, how is the root significant in the name?
It will be how the name of the molecule begins
What are the different name endings for simple molecules?
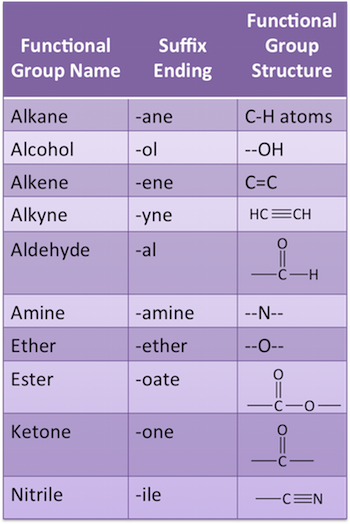
When naming alkenes, what is the rule for the numbers in between the names?
The root and the suffix would be separated by a number to show the position of the double bond.
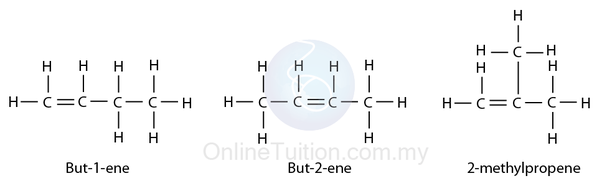
For butane:
Number the carbon atoms in the chain from 1 to n (in this case 1 to 4)
Look at what position the double bond is in:
→ For but-1-ene, the double bond is in between the 1st and 2nd carbons and so you would say it is attached to the 1st carbon, hence the name but-1-ene
→ Same applies to but-2-ene
What do alkyl groups act as?
Branches to the main carbon chain
Alkyl group formula
CnH2n+1
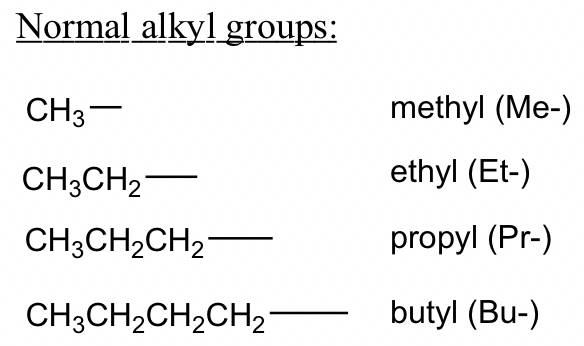
Where are alkyl groups positioned when naming compounds?
As the prefix of the root molecule (the main carbon chain molecule)
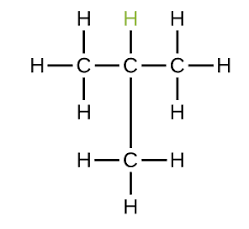
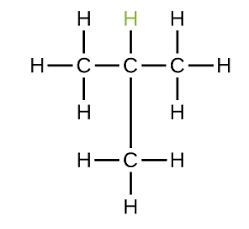
Explain the naming of methylpropane (C4H10)
The main chain would be propane (C3H8) - this becomes the root
The side chain is of methyl (CH3) - this becomes the prefix
The side chain is positioned on the second carbon chain, so the number would come before the prefix to show the position of the alkyl: 2-methylpropane
However, it is not necessary to include the number as if the alkyl were to be positioned anywhere else, the main chain would no longer be propane but would extend to become butane
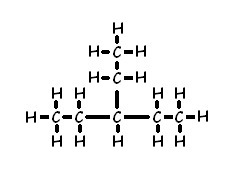
What would be the name of this compound?
3-ethylpentane
The main chain is pentane
The side chain is ethyl, which is attached to the third carbon
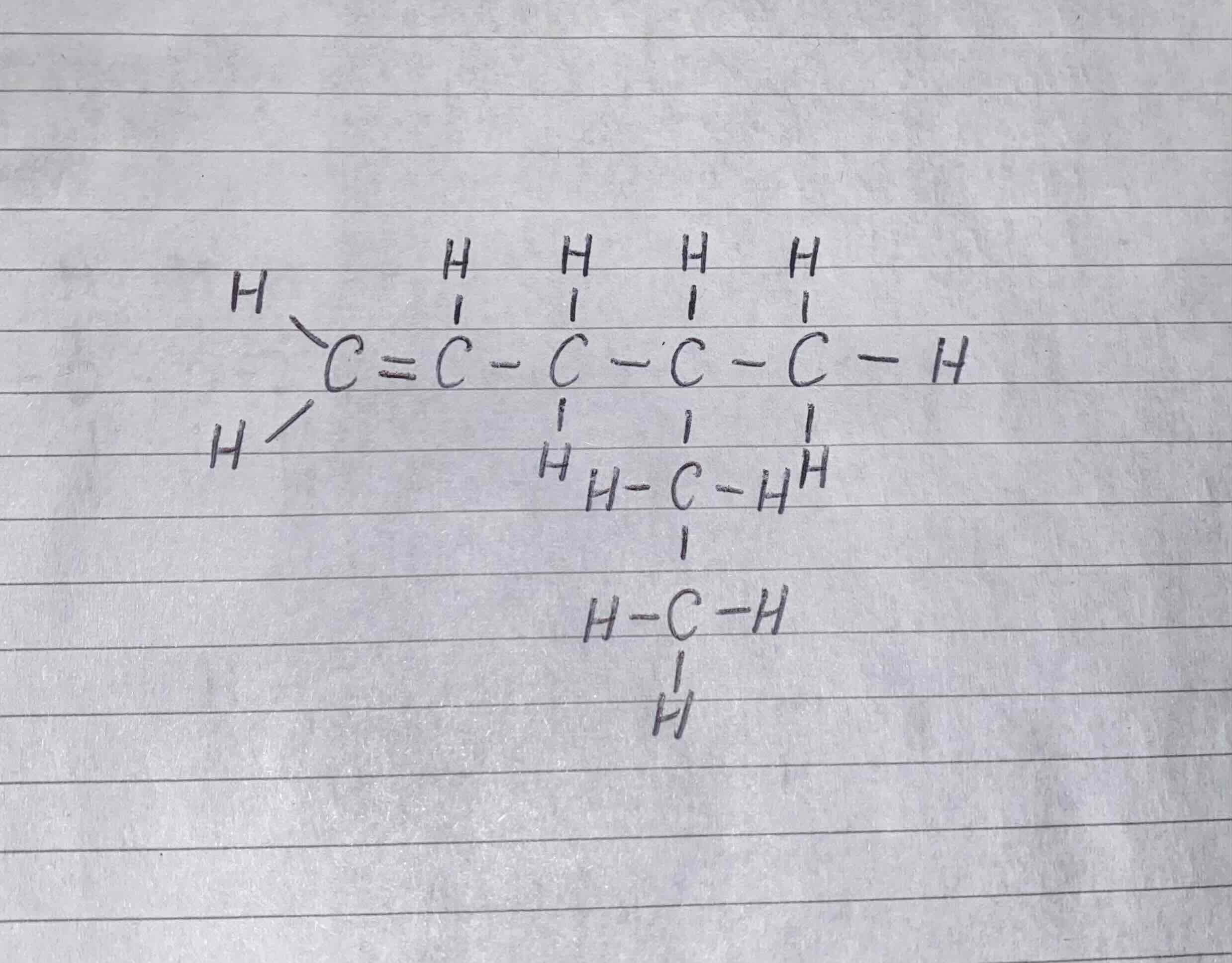
What would be the name of this compound?
4-methylhex-1-ene
The longest chain is hexene, where the double bond is attached to the first carbon so it would be hex-1-ene
The side chain is methyl, which is attached to the fourth carbon, so 4-methyl becomes the prefix
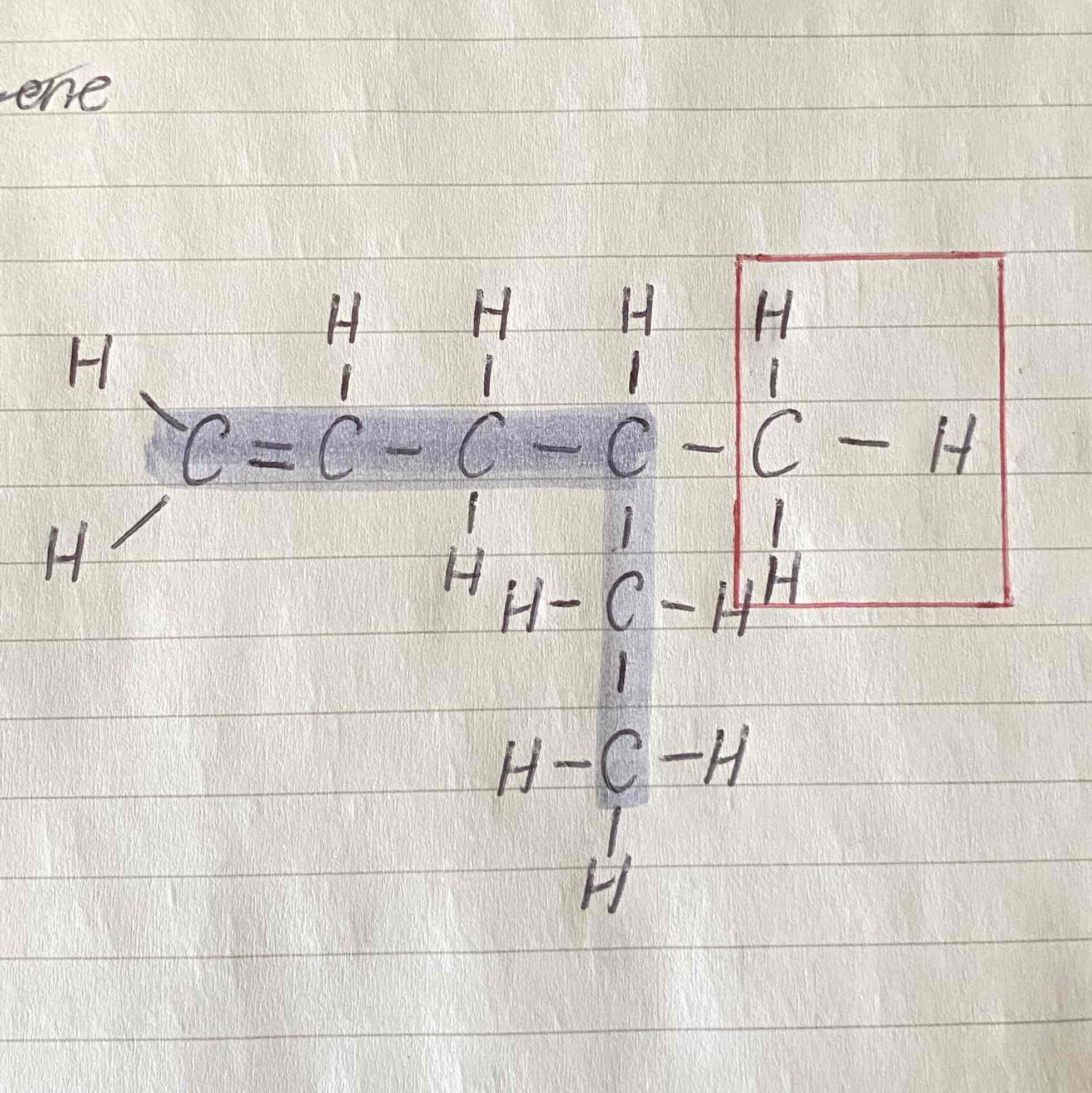
When there are two or more alkyl branches, how do you list the alkyl groups?
In alphabetical order
What is the order of naming compounds?
Position of the side chains
Amount of the same side chains
Name of the side chains
Root molecule
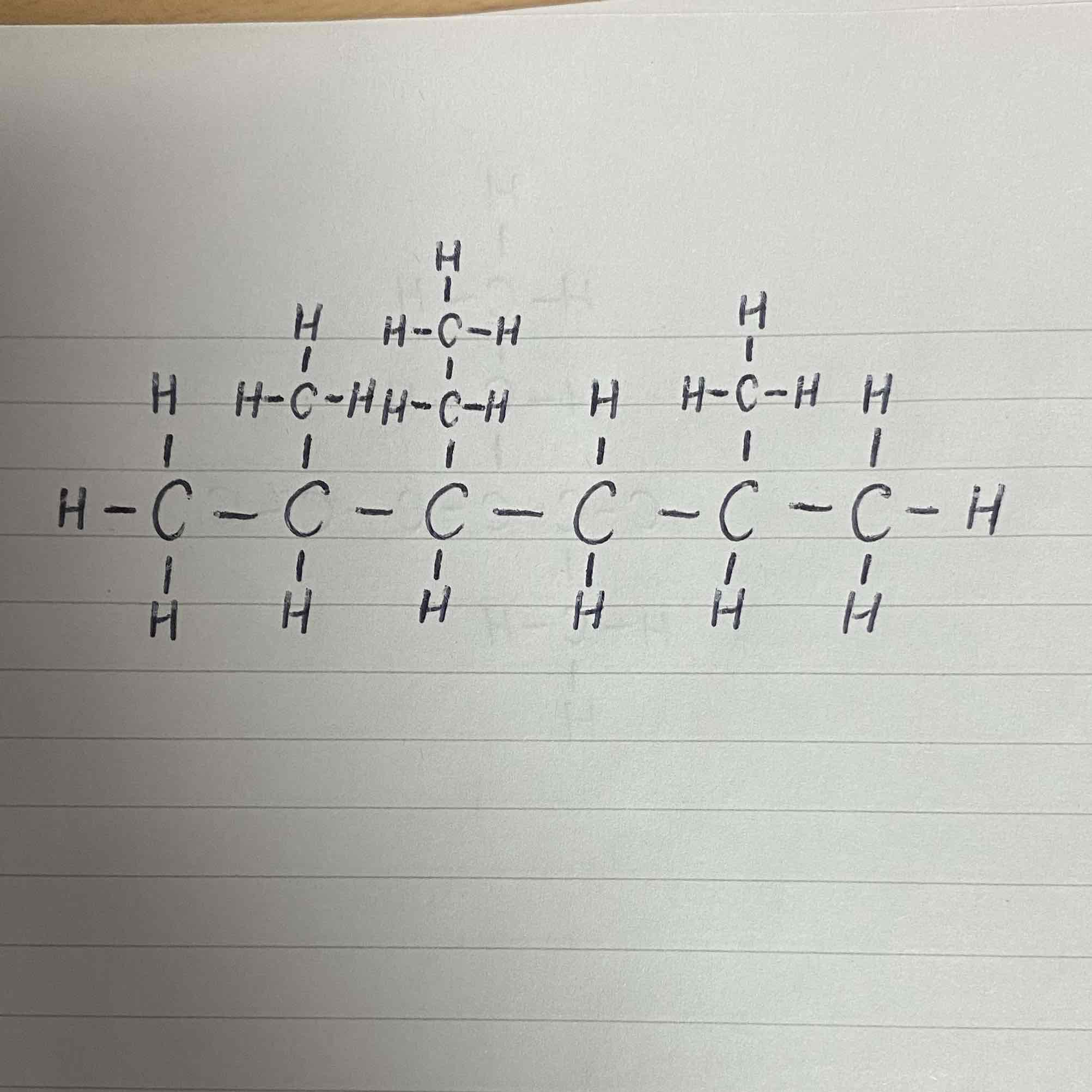
What is the name of this compound?
3-ethyl-2,5-dimethylhexane
What is the general formula for halogenoalkanes?

What happens to the formula if there are more than halogens?
The root molecule still remains as an alkane but for each added halogen there is one less hydrogen
What are the prefixes of the halogens?

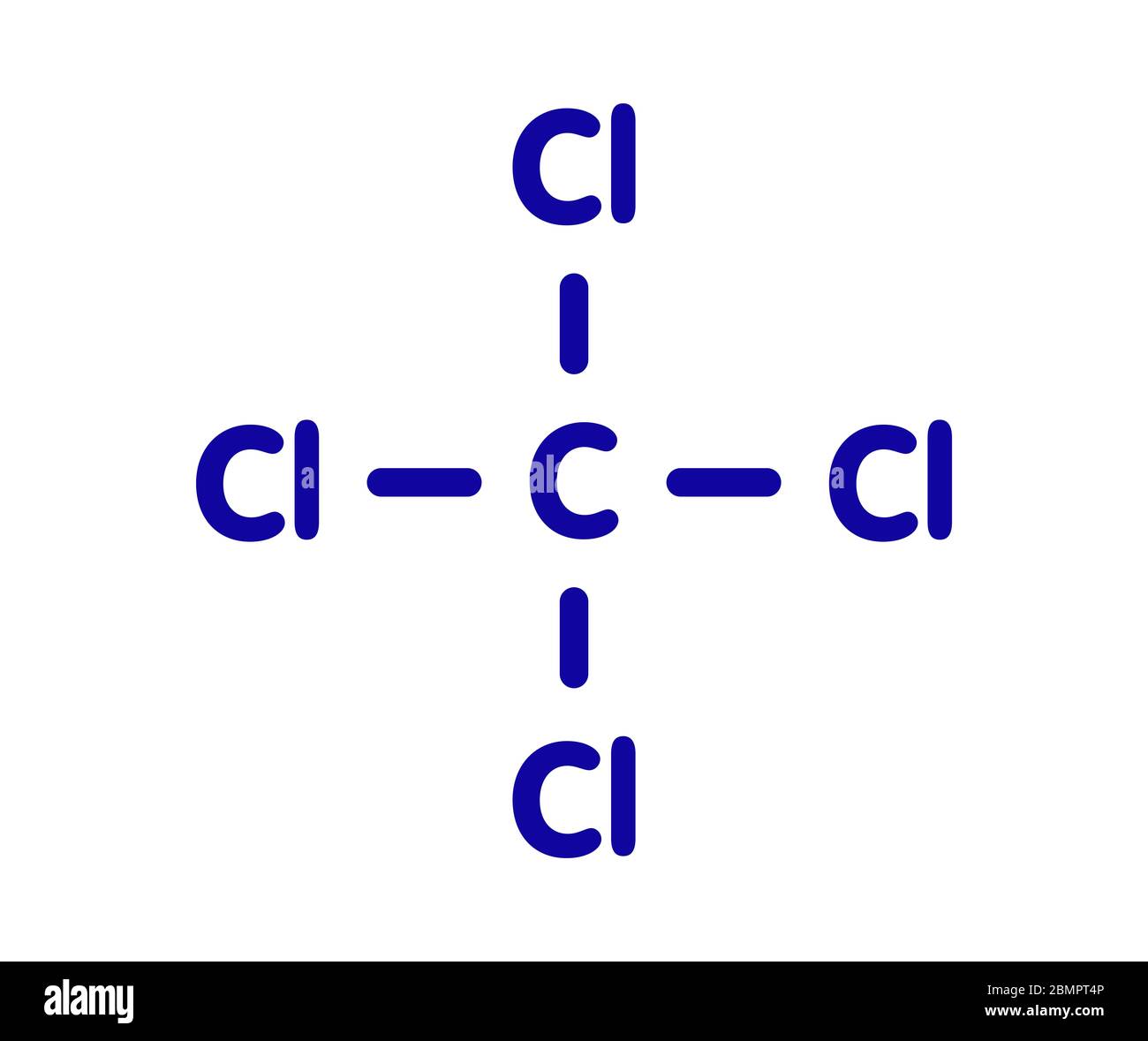
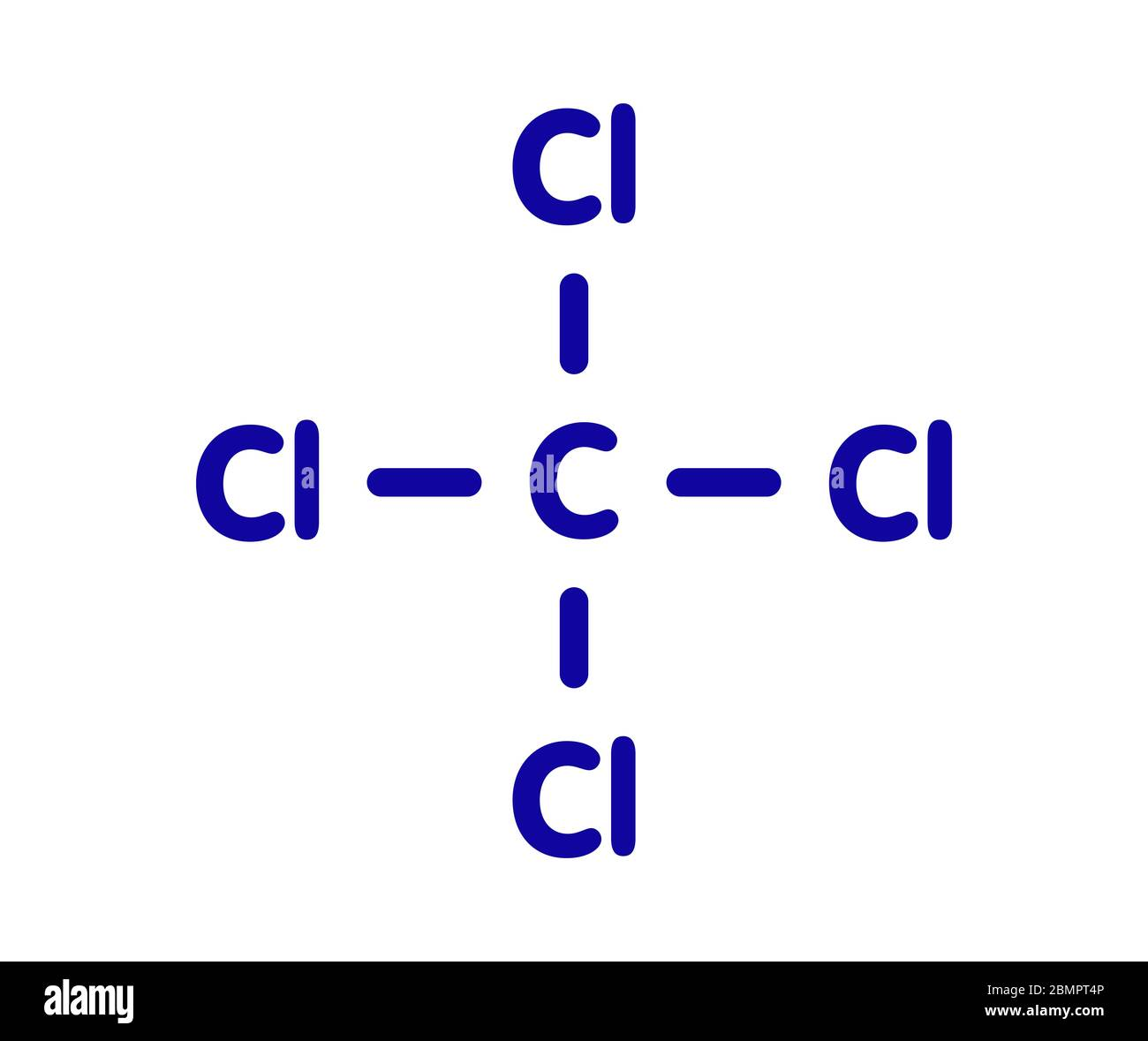
What would be the name of this halogenoalkane?
Tetrachloromethane
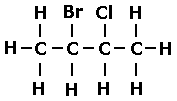
What would be the name of this halogenoalkane?
2-bromo-3-chlorobutane
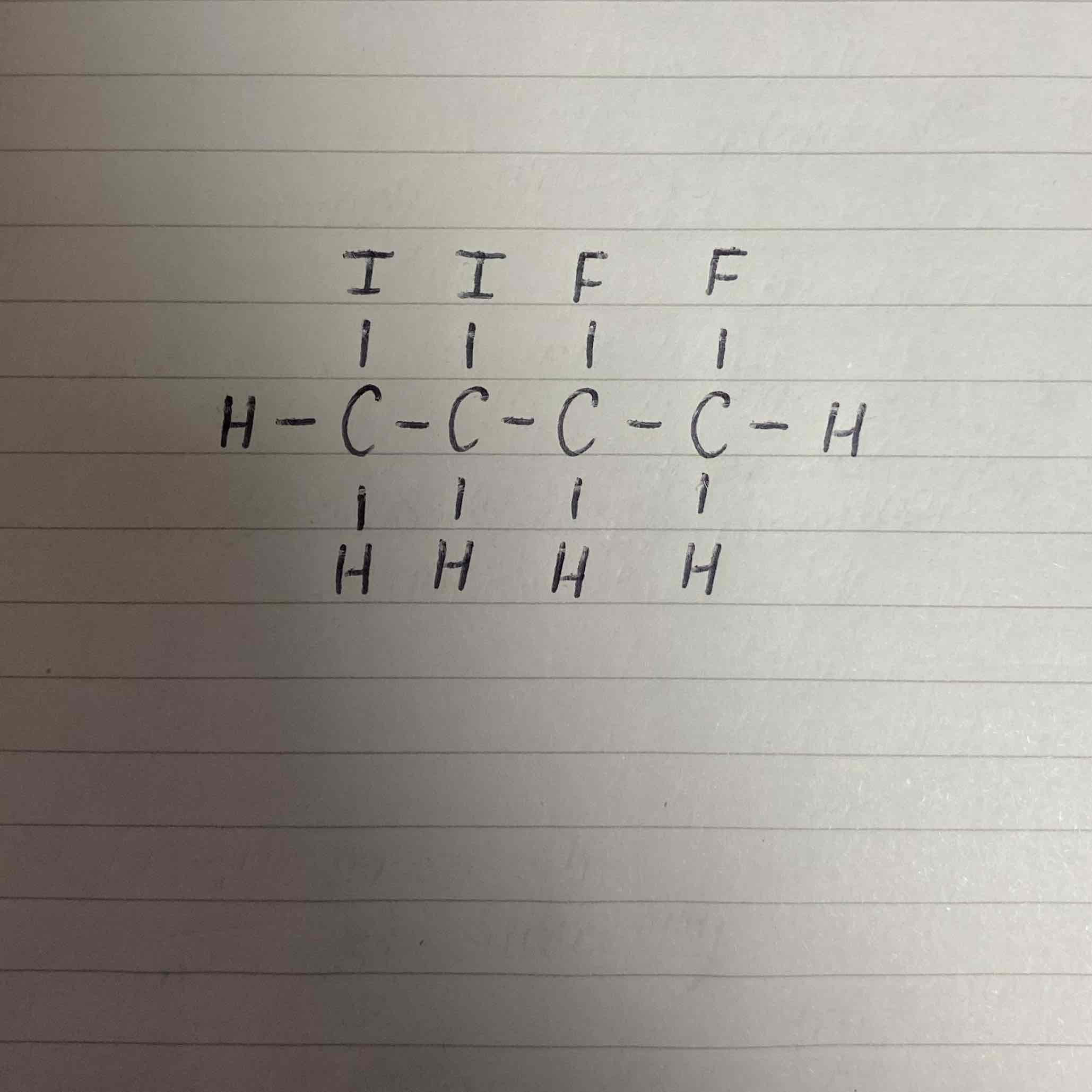
What would be the name of this halogenoalkane?
1,2-difluoro-3,4-diiodobutane
Remember fluorine is before iodine alphabetically so it is mentioned first.
This means the positioning will be dependent on the fluorines
What is the general formula for alcohols?
CnH2n+1OH

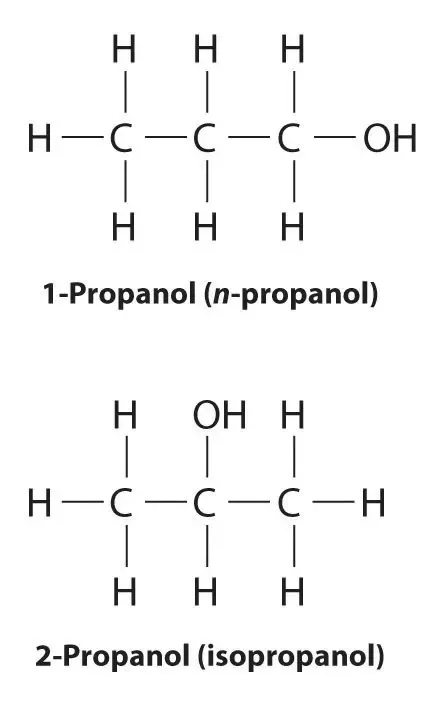
What is the difference between propan-1-ol and propan-2-ol?
The position of the -OH to the carbon
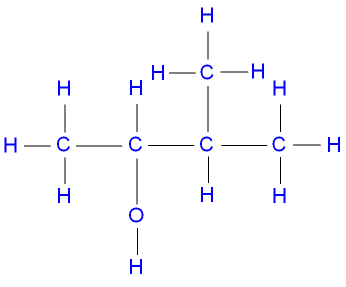
What would be the name of this alcohol?
3-methyl butan-2-ol
The position of the alcohol takes priority, then you identify the position of the alkyl group
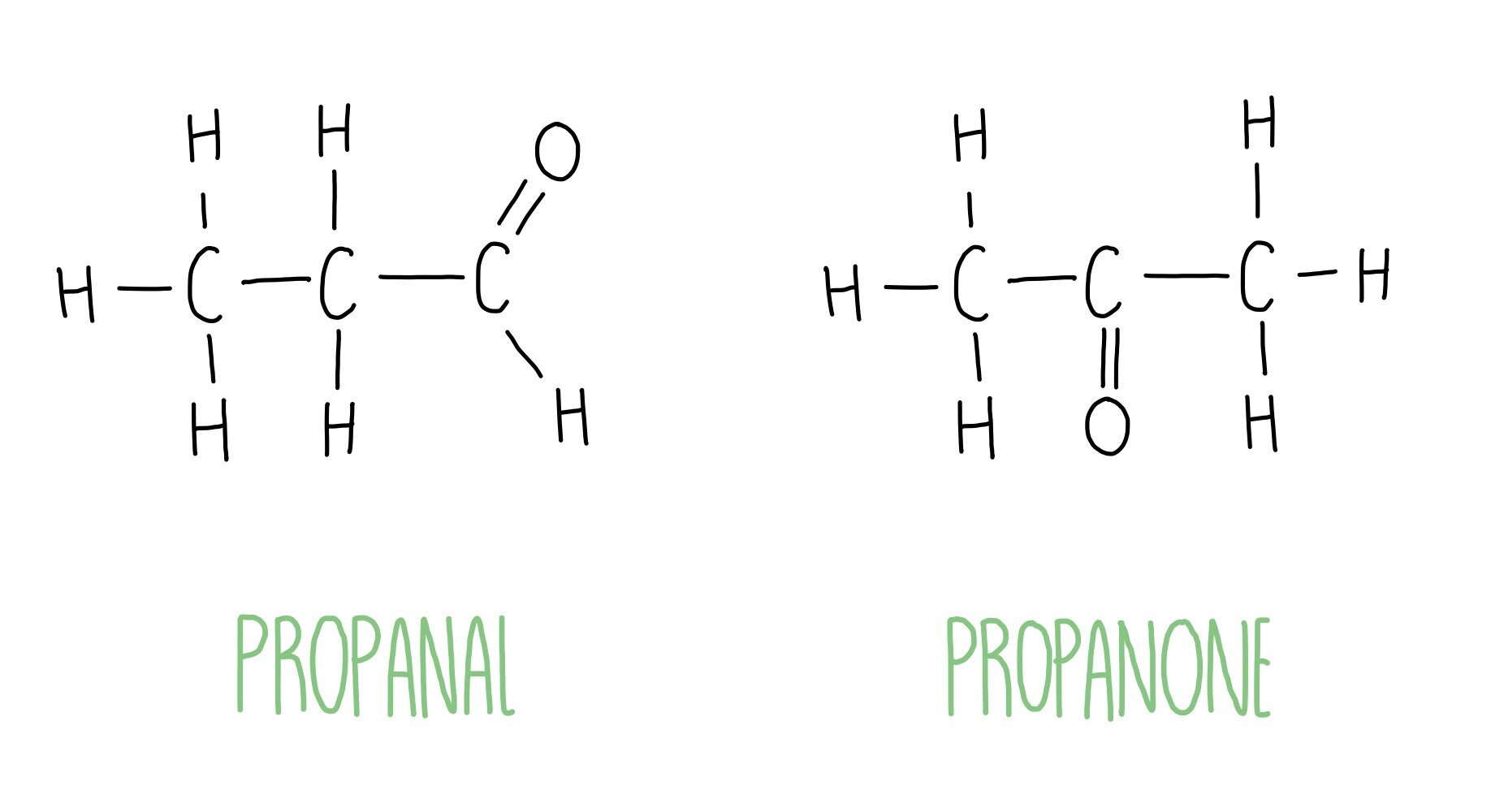
What is the general formula and functional groups for aldehydes and ketones?
Same general formula: CnH2nO
Functional group for ketones is positioned in the middle of the compound

How do you name an aldehyde?
Drop the –e from the alkane name and add the ending –al
How do you name a ketone?
Drop the –e from the alkane name and add the ending –one
Difference in structural formula between aldehydes and ketones
C3H6O
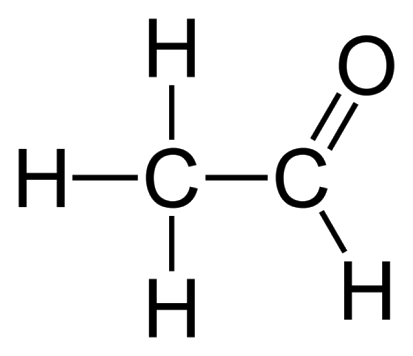
What would be the name of this aldehyde?
Ethanal
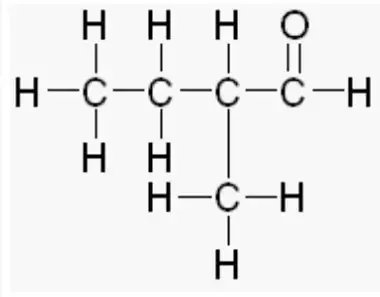
What would be the name of this aldehyde?
2-methylbutanal
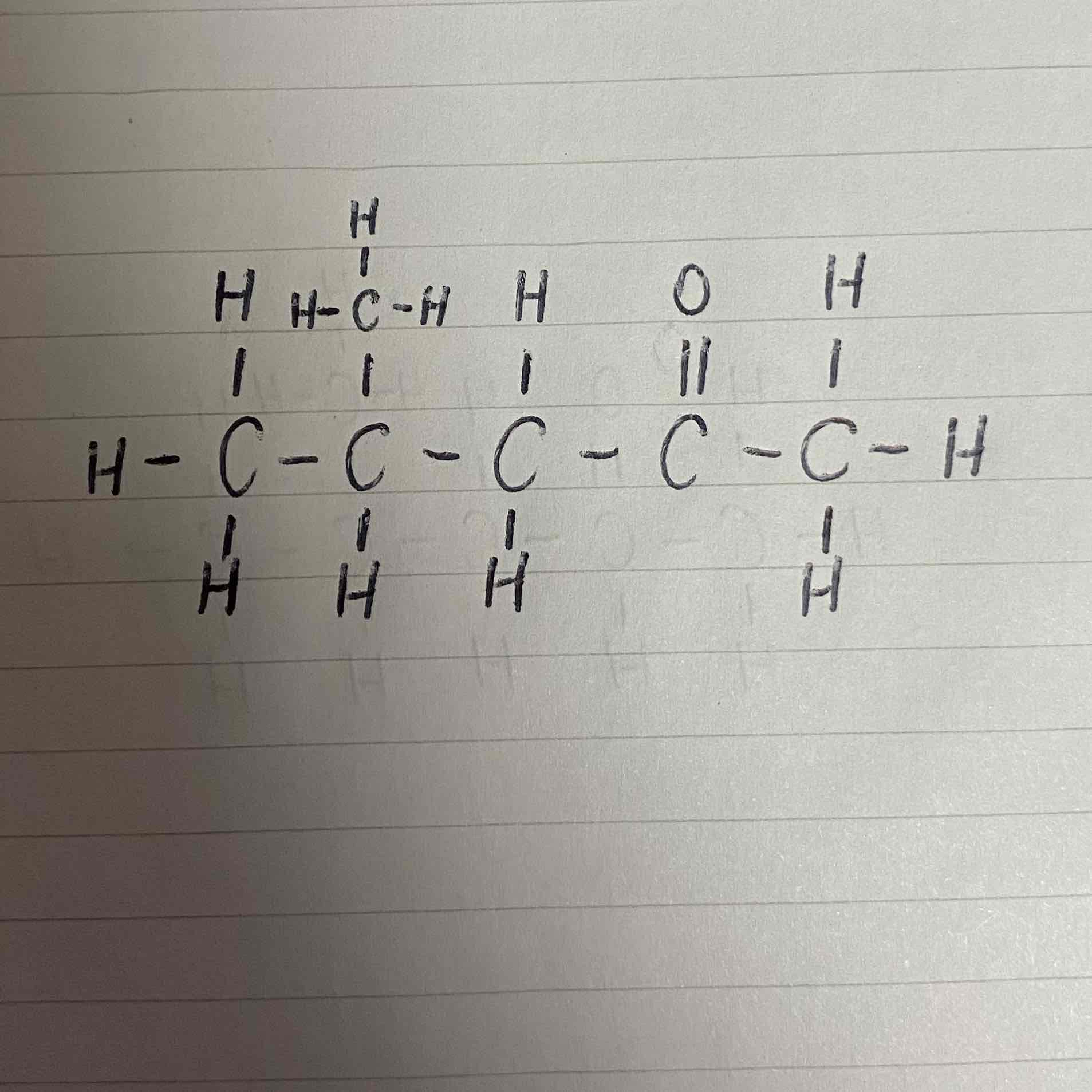
What would be the name of this ketone?
4-methyl pentan-2-one
The position of the functional group takes precedence
Functional group for carboxylic acids
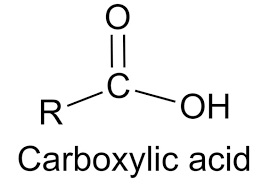
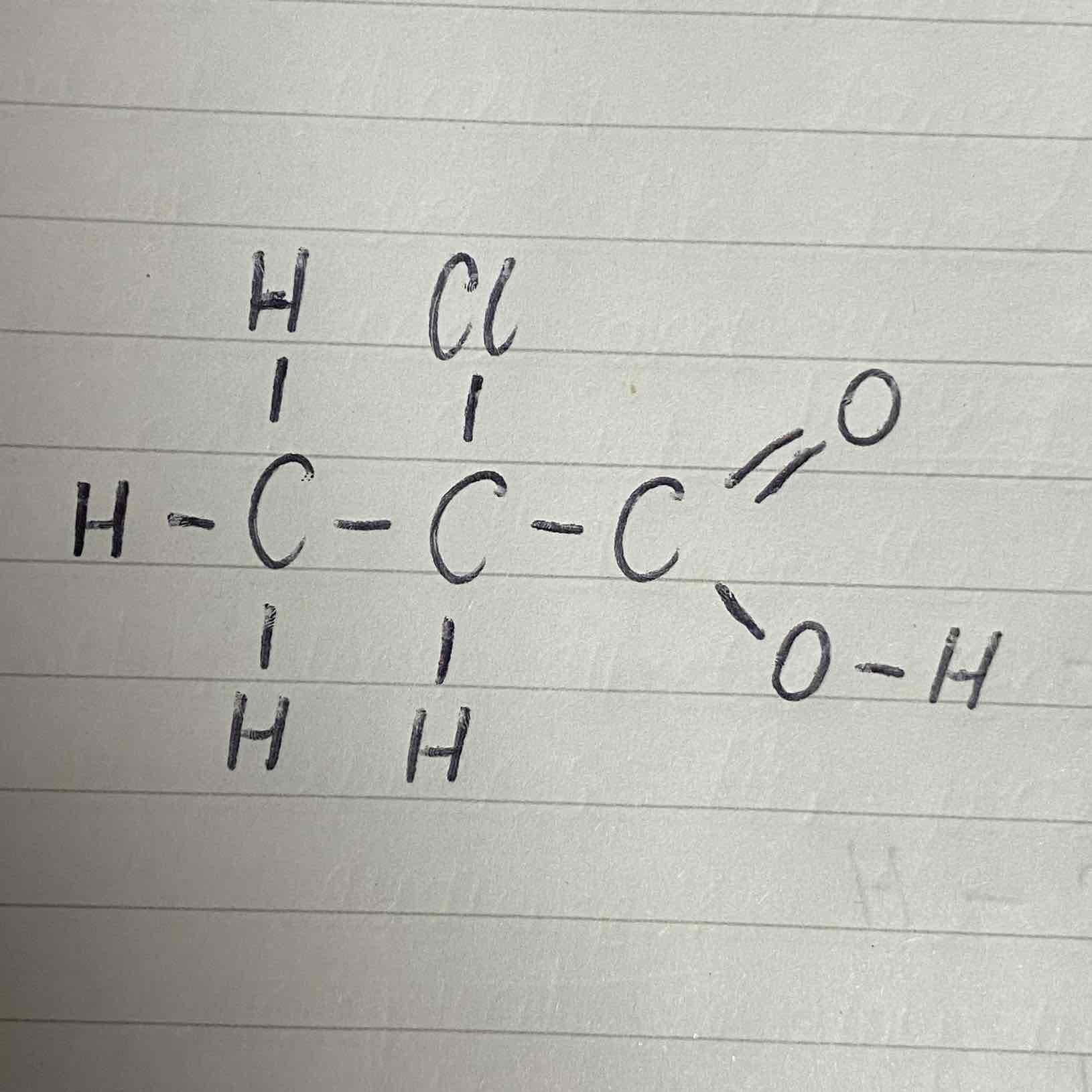
What would be the name of this carboxylic acid?
2-chloropropanoic acid
Functional group for amines
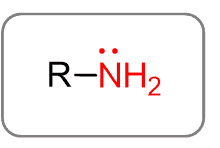
What is particular about naming amines?
The carbon chain is named as an alkyl
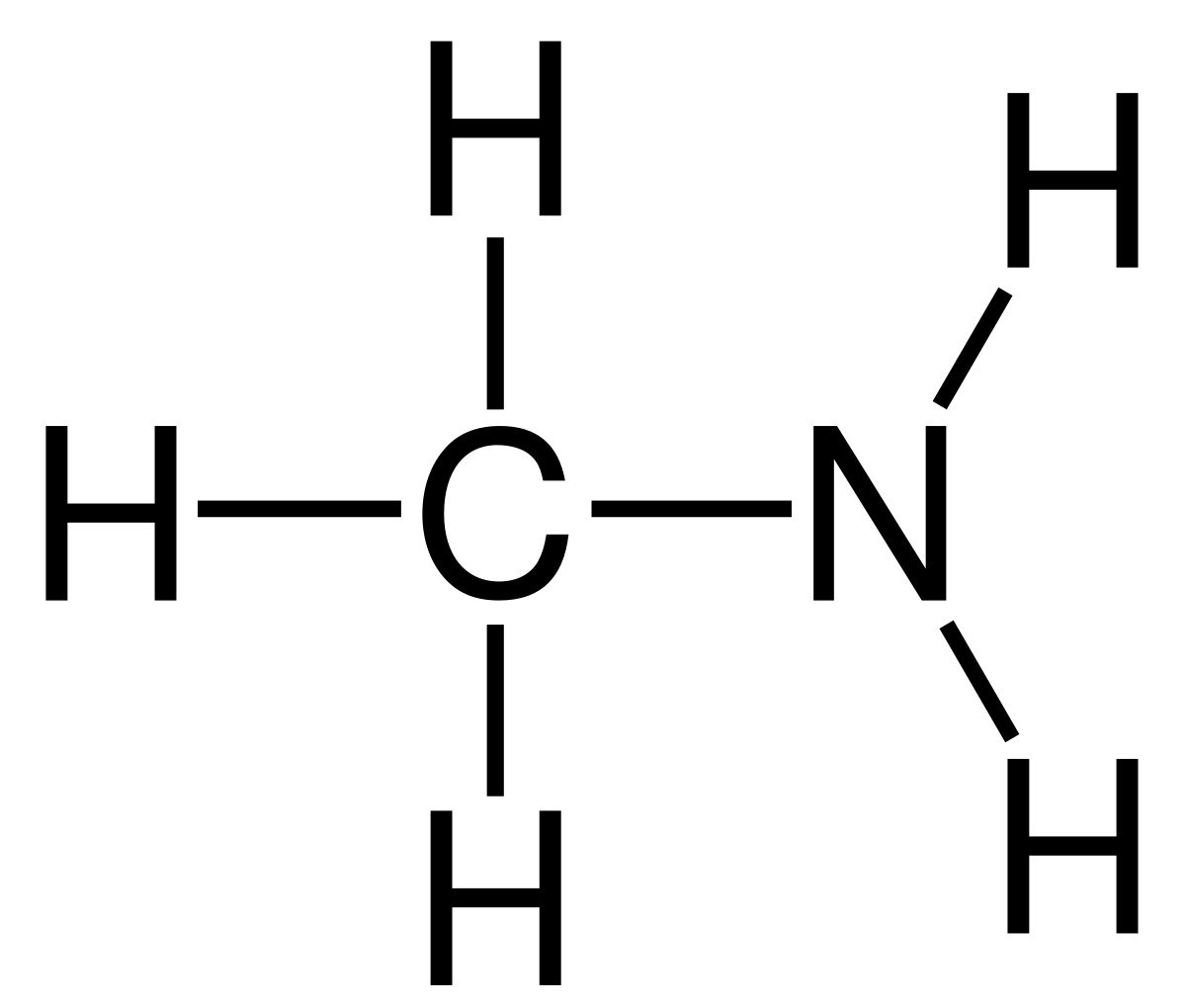
What would be the name of this amine?
Methylamine
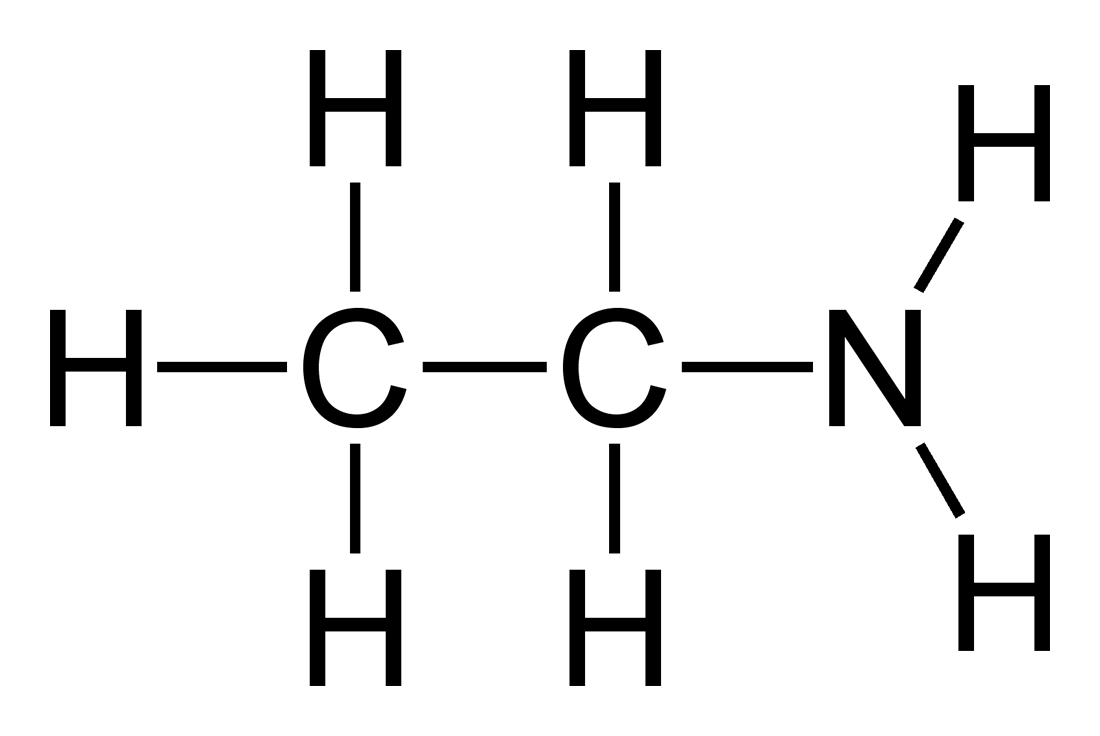
What would be the name of this amine?
Ethylamine
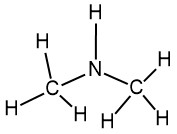
What would be the name of this amine?
Dimethylamine
Functional group for nitriles

How do you name a nitrile?
By adding nitrile at the end of the carbon chain
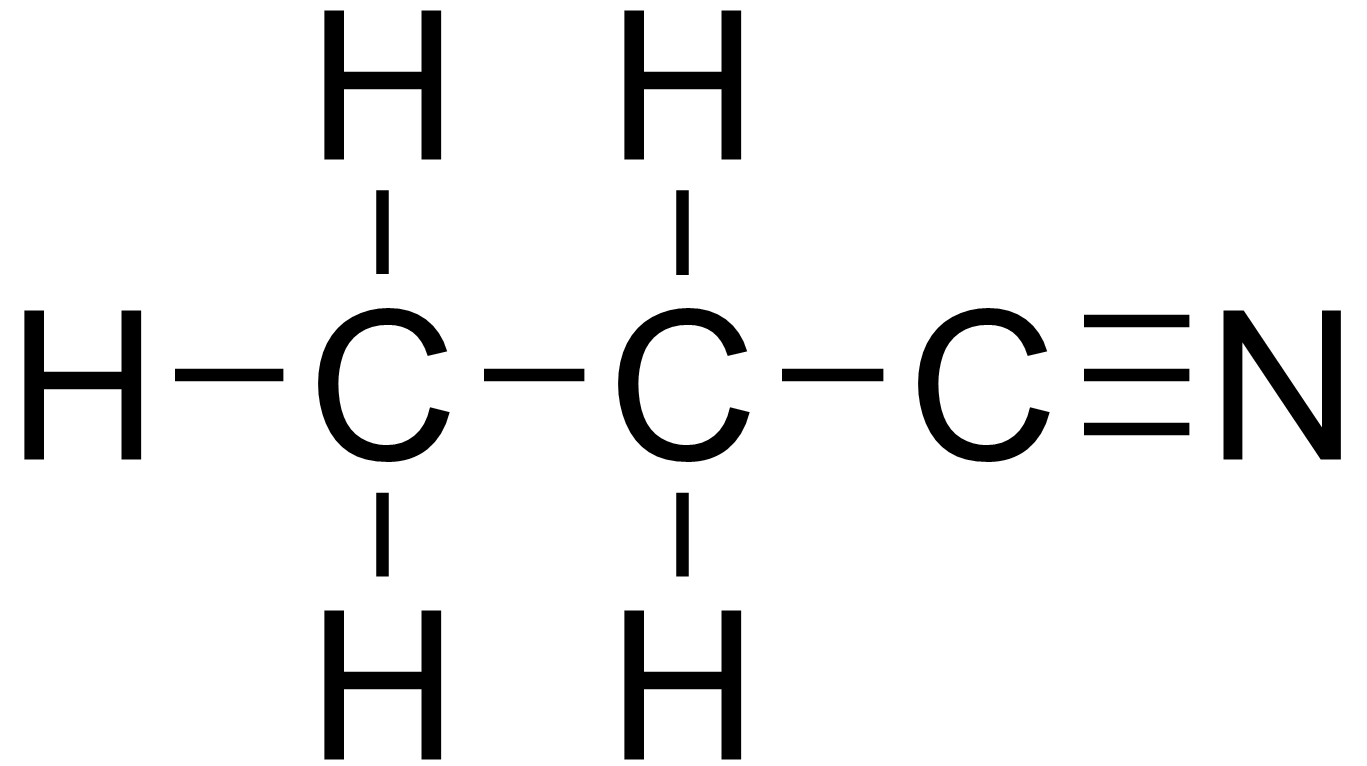
What would be the name of this nitrile?
Propanenitrile
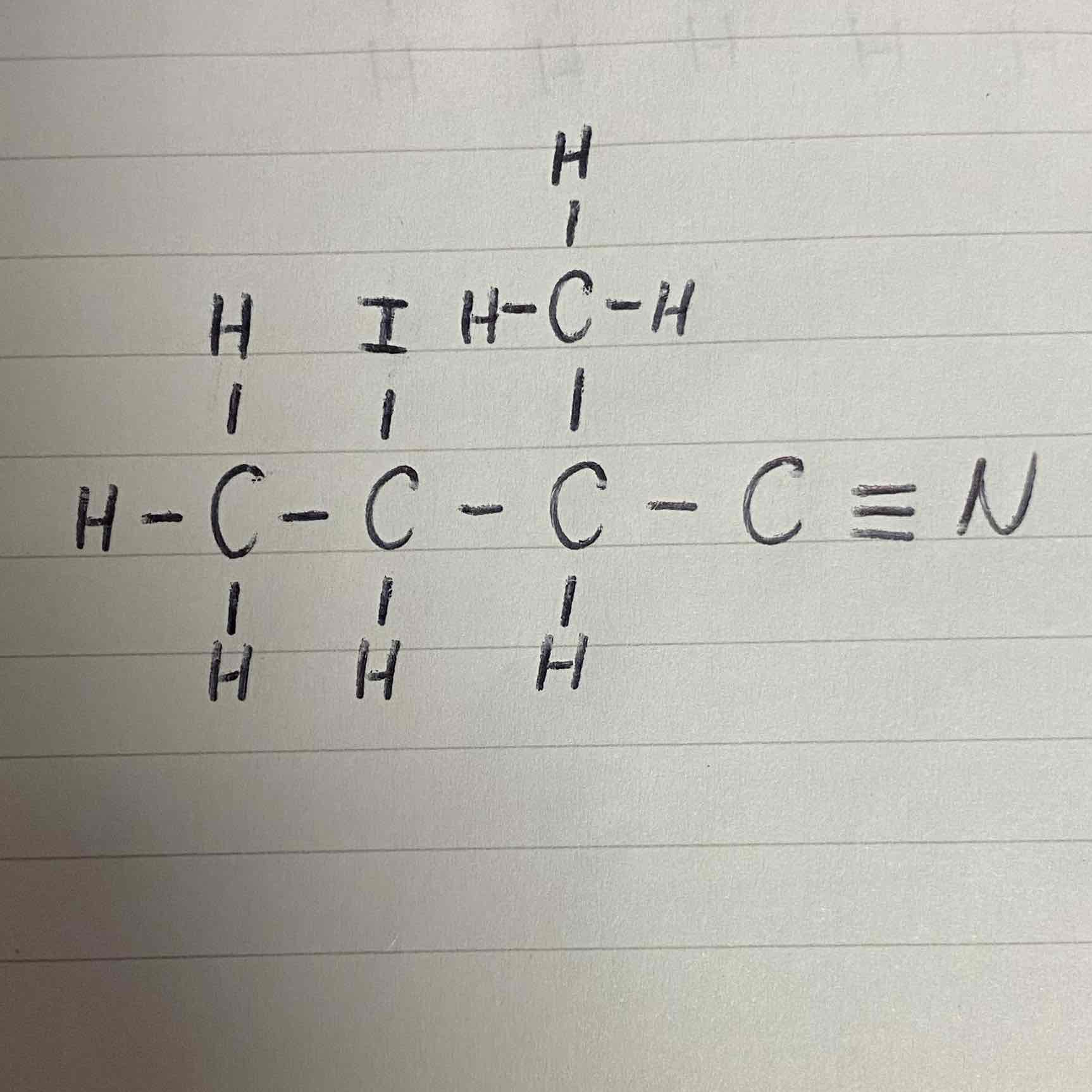
What would be the name of this nitrile?
3-iodo-2-methylbutanenitrile