CH 450 Final Exam
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
Difference between cell surface receptors and nuclear receptors?
Cell surface: plasma membrane, bind hydrophilic (insulin, adrenaline) ligands, trigger signaling cascades (cAMP or Ca2+), fast, modifies existing proteins
Nuclear: cytosol or nucleus, bind hydrophobic (steroids) ligands, directly regular gene expression (transcription factor), slow, synthesizes new proteins
What ligand activates the β-adrenergic receptor?
Epinephrine (adrenaline) and glucagon (more relevant)
What type of receptor is the β-adrenergic receptor?
G-protein coupled receptor (GPCR)
What does a G protein do? Key properties?
Transduces extracellular signal from receptor to effector (like adenylyl cyclase)
- Bind GTP when active, GDP when inactive
What is cAMP's role? What does it activate?
Is a second messenger in intracellular signaling transferring external signals (ex. hormones) from surface to the interior
Mainly activates protein kinase A (PKA): phosphorylates target proteins in response to cAMP signals (gluconeogenesis)
How is the PKA signal terminated?
When cAMP is degraded, or G protein inactivated via GTPase activity (GTP + H2O --> GDP + Pi)
What is the primary mechanism for terminating a cAMP signal?
Degradation by Phosphodiesterases (PDEs). These enzymes break the 3',5'-phosphodiester bond in cAMP, converting it into inactive 5'-AMP.
What type of receptor is the insulin receptor and what does it do?
Receptor tyrosine kinase, autophosphorylation in response to binding of insulin
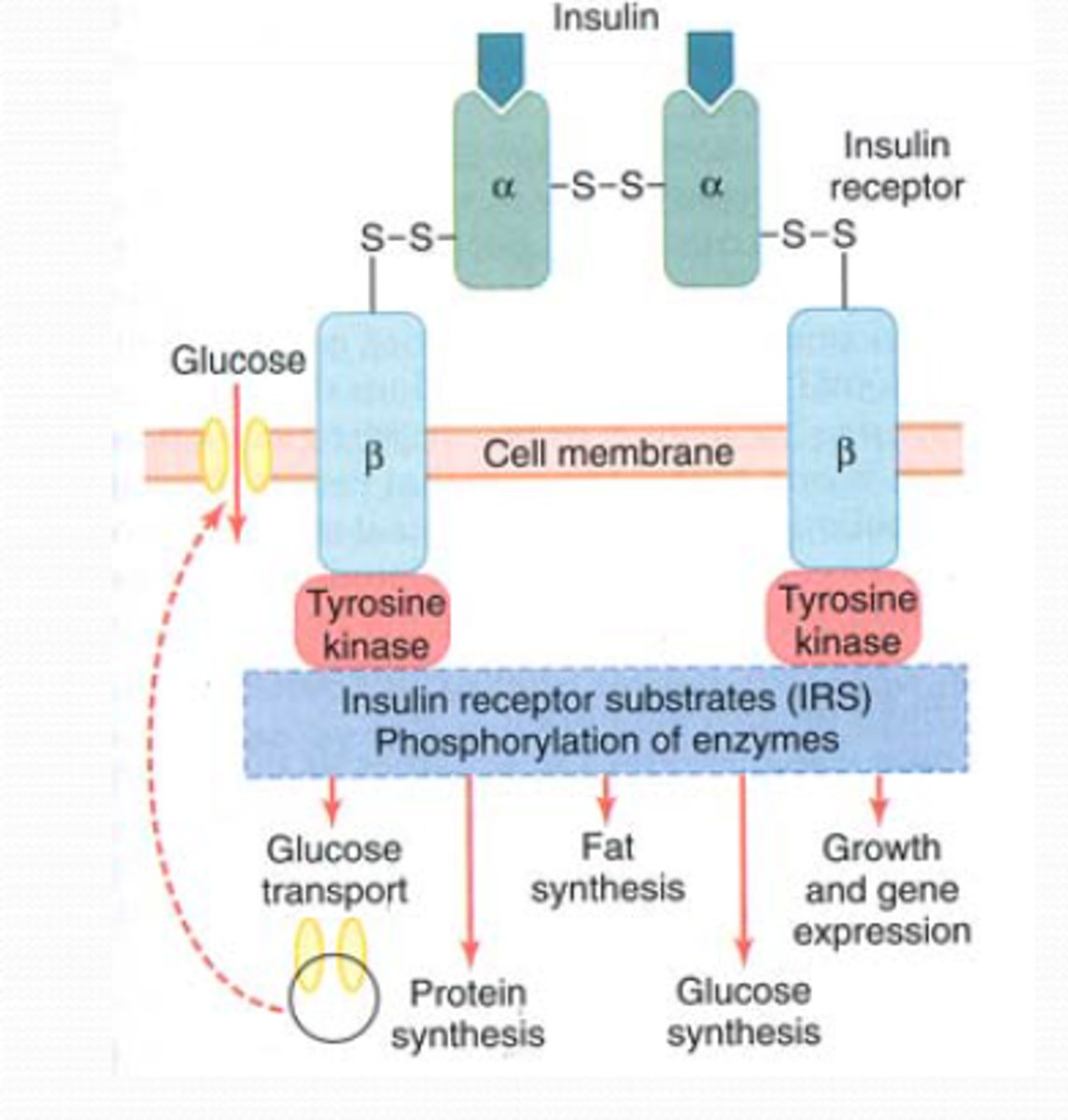
Downstream effects of insulin signaling?
- Increased glucose uptake
- Enzyme regulation
- Gene expression changes
- Can phosphorylate various proteins to affect activities
In principle, the physiological effects of epinephrine should be mimicked by addition of cAMP to the target cells. In practice, addition of cAMP to intact target cells elicits only a minimal physiological response. Why?
The added cAMP does not cross the plasma membrane
Dibutyryl cAMP is widely used in studies of cAMP function, and has two butyryl (-CO(CH2)2CH3) added to the cAMP molecule. What is the basis for the difference in cellular response to these two substances?
Dibutyryl cAMP crosses the plasma membrane after being added to intact cells.
The respiratory symptoms of asthma result from constriction of the bronchi and bronchioles of the lungs, caused by contraction of the smooth muscle of their walls. Raising cAMP in the smooth muscle reverses the constriction of the bronchi and bronchioles.
What is the mechanism of action for the medication albuterol, an inhaled beta-adrenergic agonist, in treating asthma? Would you expect this drug to have any side effects? If so, what design change could you make to the drug to minimize side effects?
1. Albuterol binds to G protein-coupled receptors, mimicking the actions of epinephrine
2. Yes, because beta-adrenergic receptors control many other physiological processes
3. Increase the specificity of the drug for the specific subtype of B-adrenergic receptor
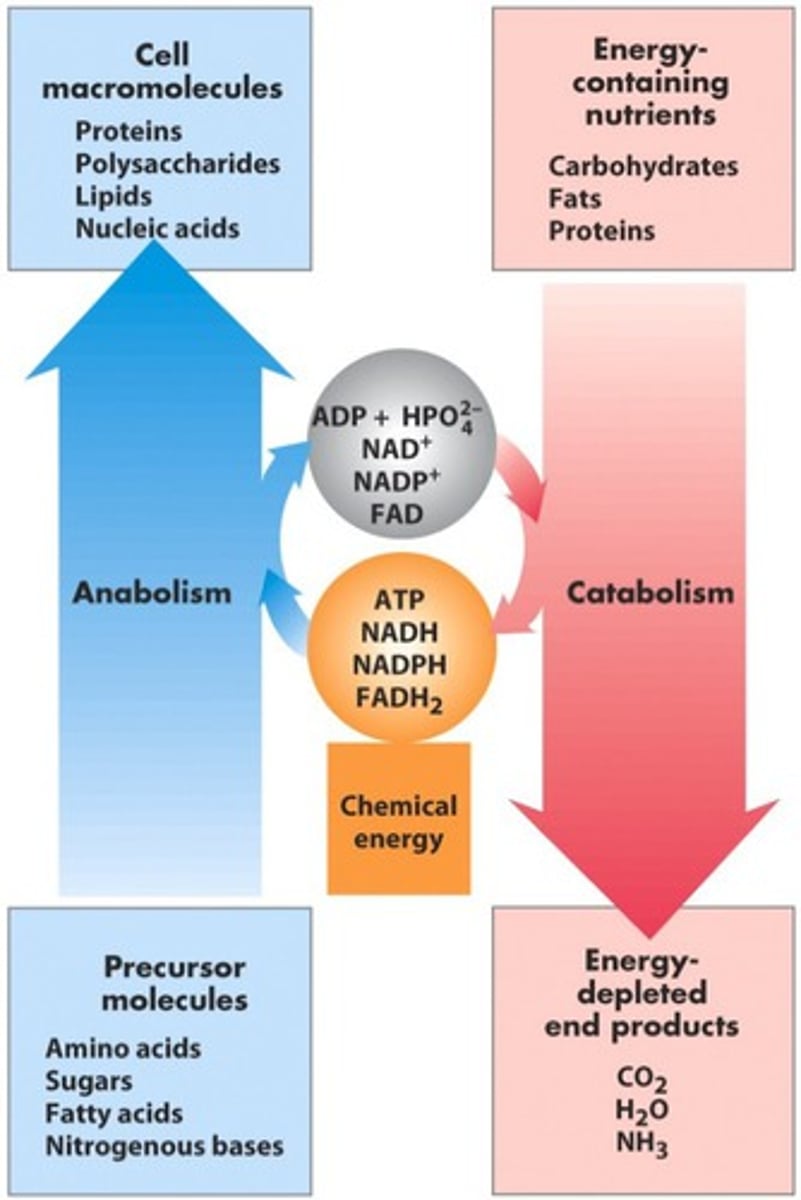
Catabolism vs anabolism?
Catabolism: breakdown, releases energy
Anabolism: synthesis, requires energy
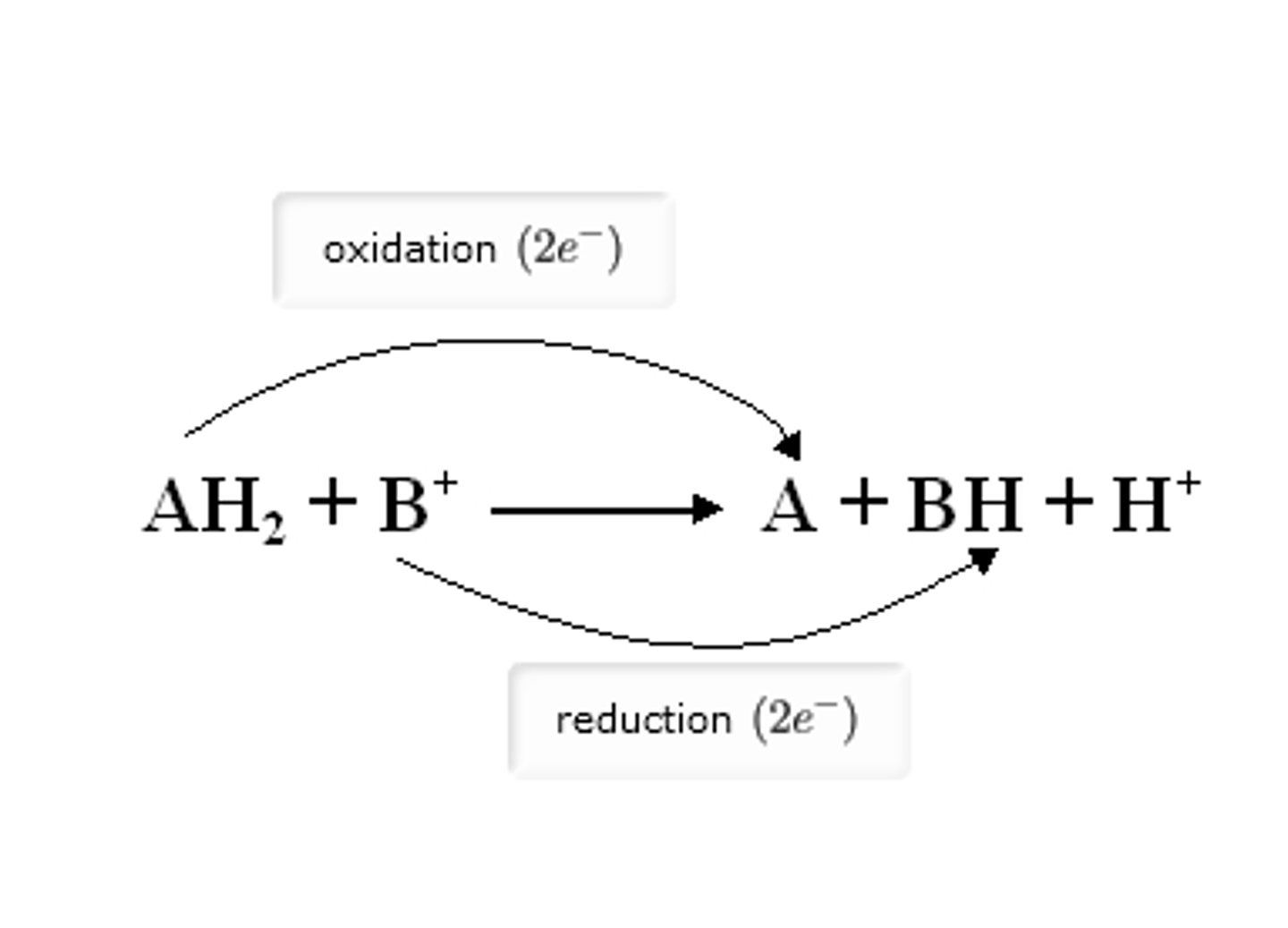
How to identify oxidation-reduction (redox) reactions
These reactions involve the transfer of electrons and are the primary way cells extract energy from fuel molecules.
Key Indicator: Look for the movement of hydrogen atoms (H) or changes in oxygen content
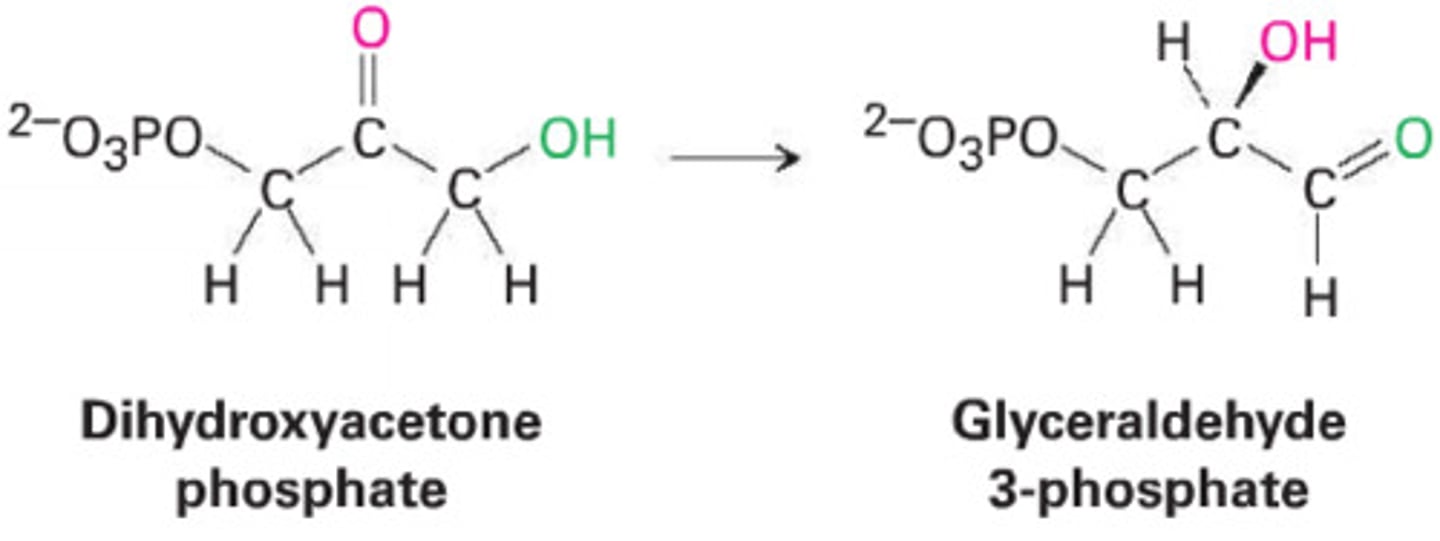
How to identify internal rearrangement reactions
These involve the redistribution of electrons within a single molecule to change its structure without changing its formula (isomerization) or removing atoms to form double bonds (elimination).
Key Indicator: The molecular formula typically stays the same, but functional groups move to different carbons
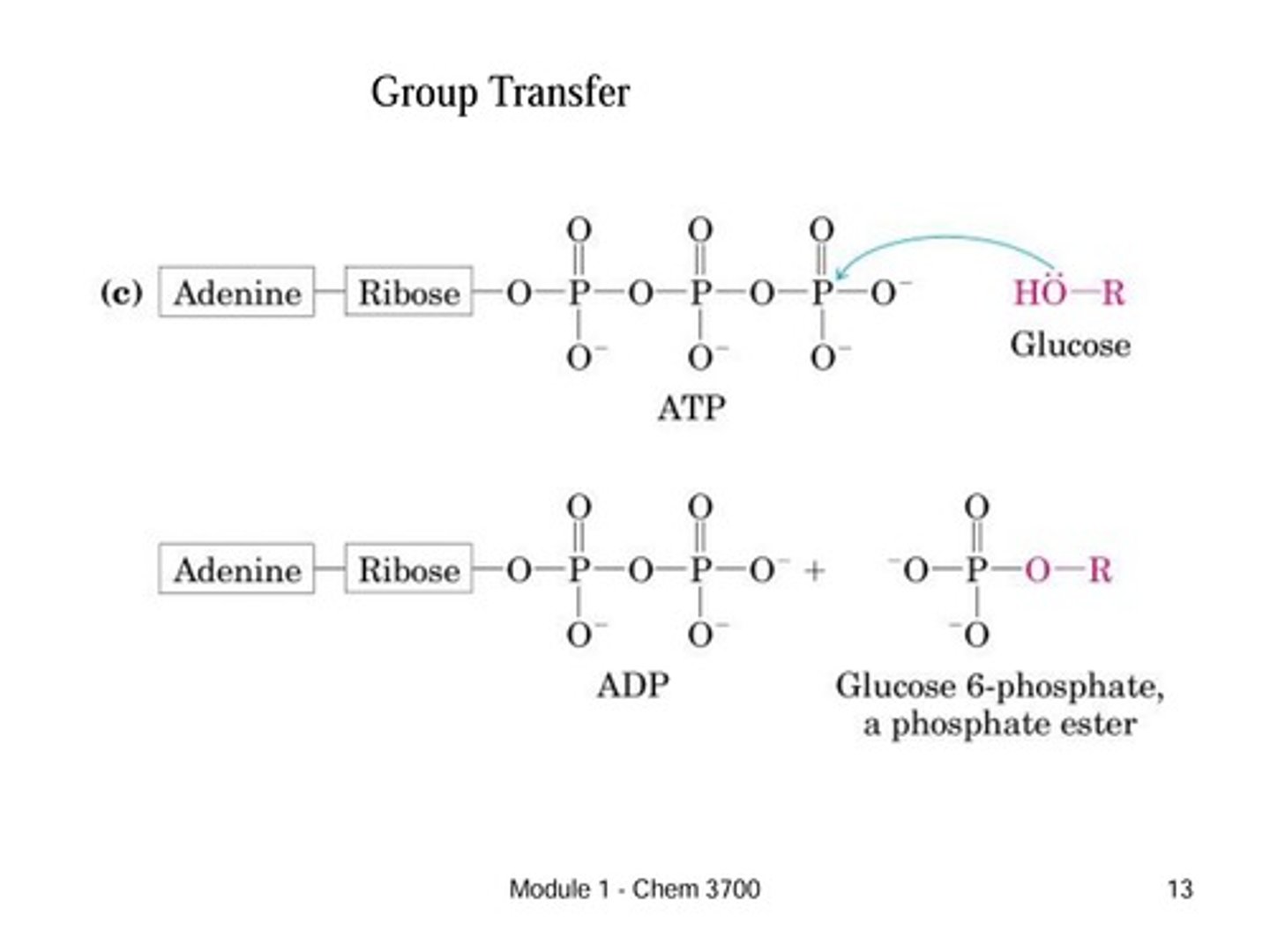
How to identify group transfer reactions
This is the transfer of a functional group (acyl, glycosyl, or phosphoryl) from one molecule to another.
Key Indicator: A specific group (like phosphate) disappears from a donor (like ATP) and appears on an acceptor molecule
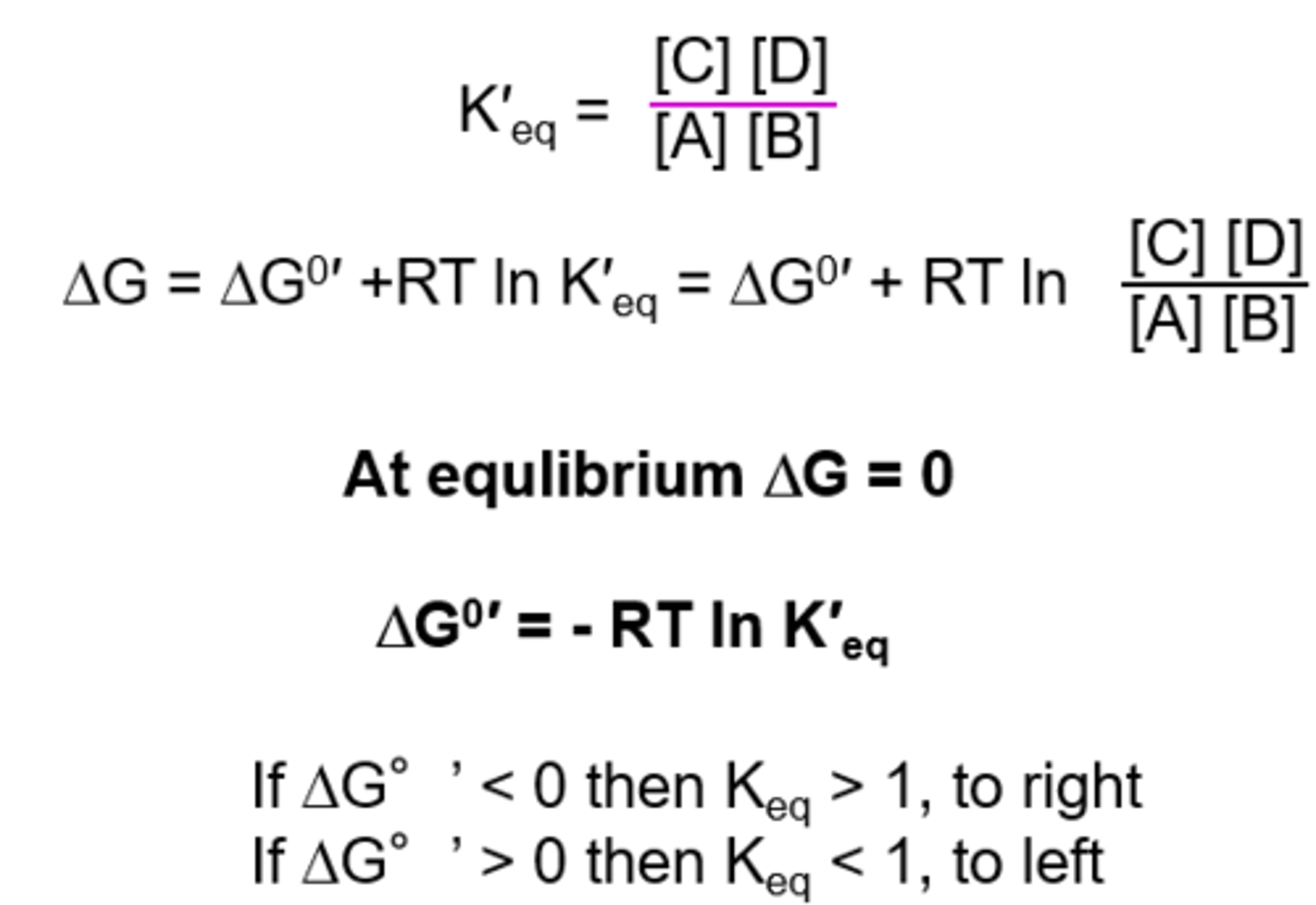
Relationship between ΔG°′ and Keq
ΔG°′ = −RT ln Keq
Difference between ΔG and ΔG°′
ΔG°′: standard conditions
ΔG: actual cellular conditions
Why does ATP hydrolysis have a large negative ΔG°′?
1. Charge Repulsion: ATP has ~4 negative charges clustered together; removing a phosphate group releases this "electrostatic strain."
2. Resonance Stabilization: The free phosphate has more resonance structures than when bound, making it much more stable (lower energy)
3. Ionization: Proton comes off at the end (?)
What affects actual ΔG
Concentrations (Q, mass action ratio)
Examples of high-energy phosphate compounds
- PEP
- 1,3-biphosphoglycerate
Are made in second half of glycolysis, that are used to transfer a phosphate onto ADP to make ATP
What is reaction coupling?
Combining reactions so net ΔG is negative
NOTE: When coupling reactions, the one with the larger negative ΔG will be the one to provide the energy, other one a phosphorylation
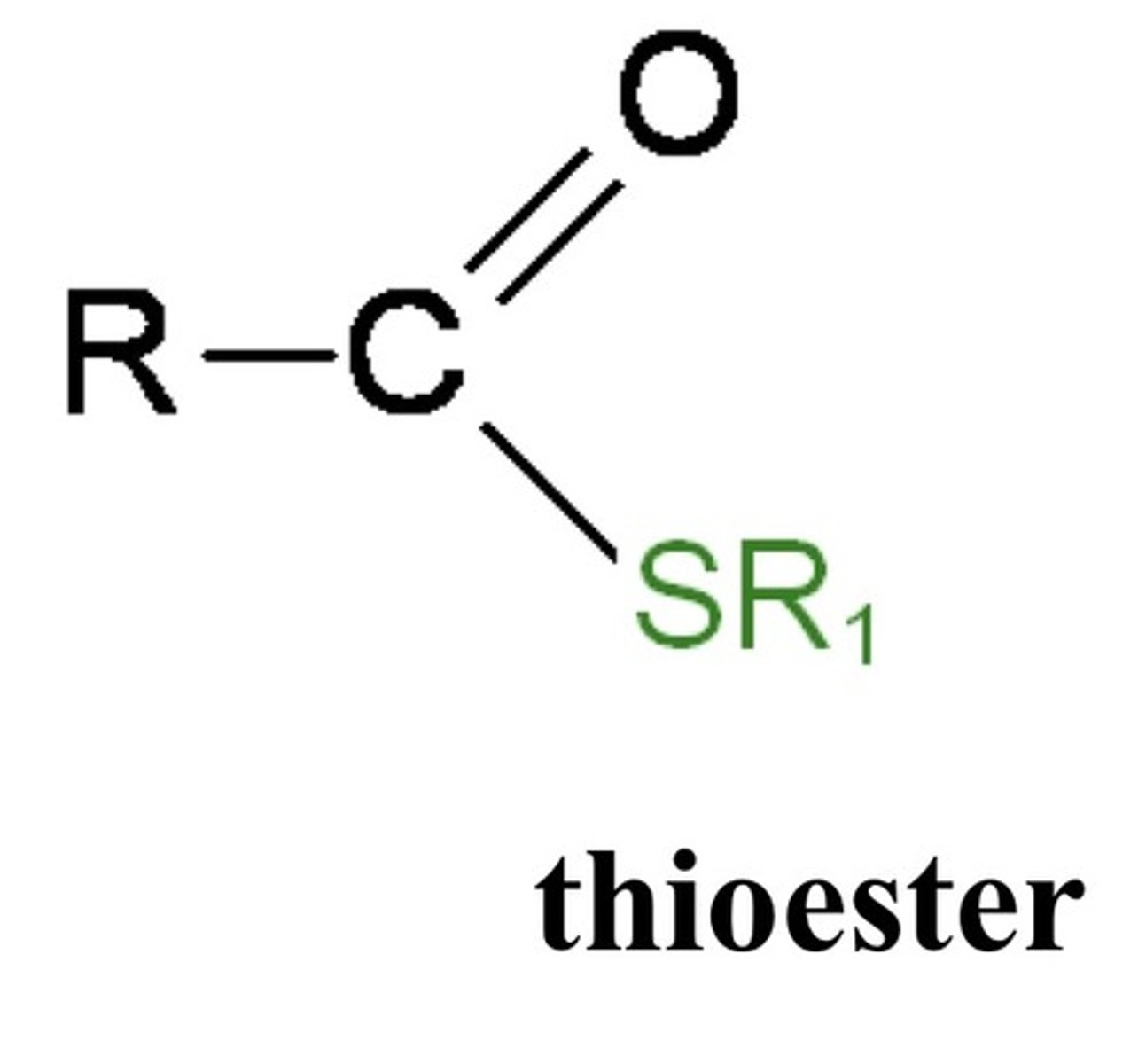
What are thioesters?
High-energy sulfur-containing esters (ex: acetyl-CoA)
- Hydrolyzed with large negative ΔG
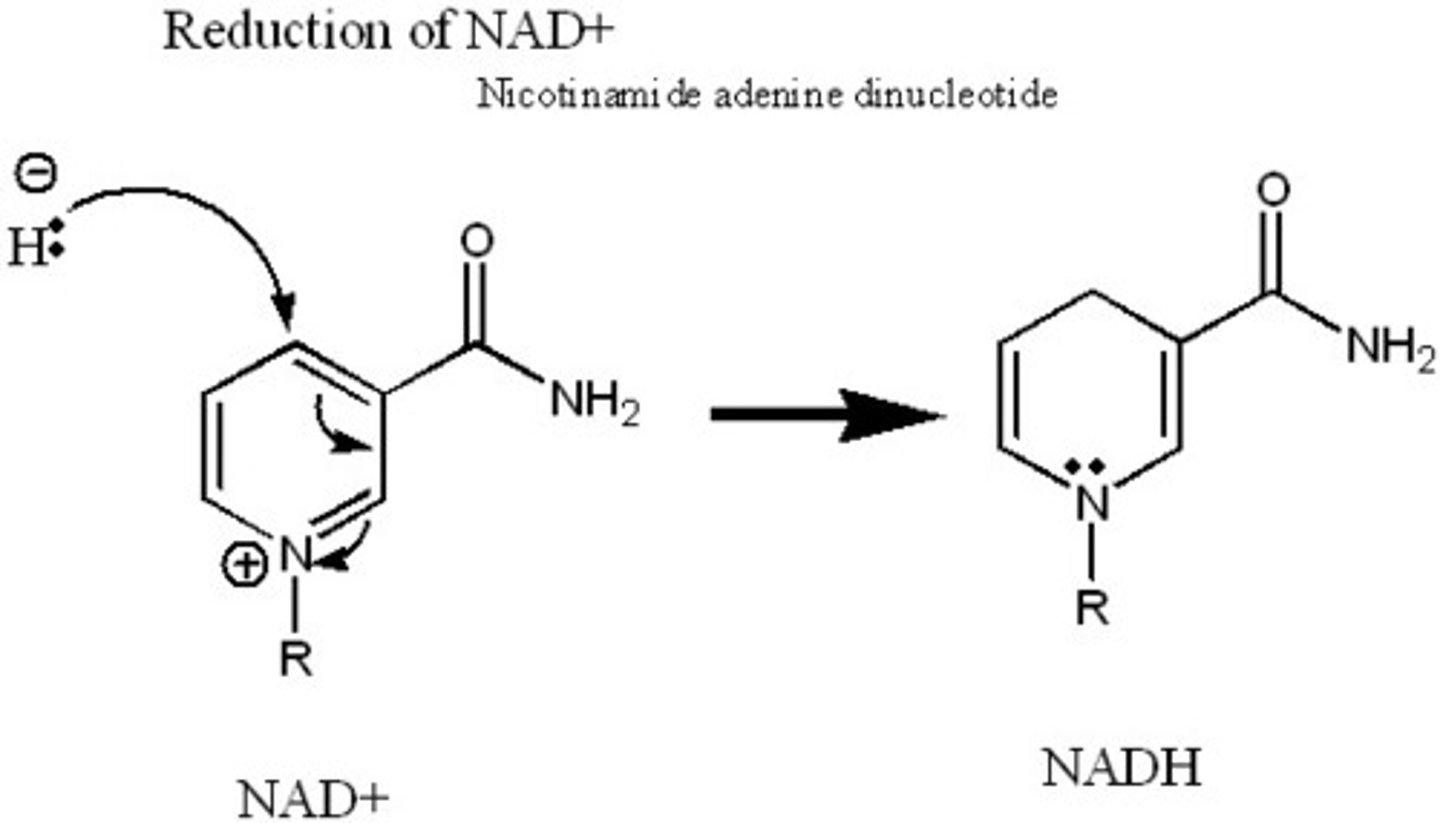
Main election carriers for oxidation reduction reactions
NAD+ and FAD (know how to draw NAD+ and NADH)
Goal of glycolysis?
Break down glucose (6 carbon sugar) --> 2 pyruvate (3-carbon sugars) + energy (2 ATP, 2 NADH?)
Two stages of glycolysis?
1. Energy investment
2. Energy payoff
Key strategy of glycolysis?
Make a pseudo-symmetrical hexose that can be cleaved into two similar trioses. Make high energy phosphate compounds that can transfer a phosphate to ADP to make ATP
Enzymes types to recognizes
Kinase --> adds phosphate
Isomerase --> rearranges
Dehydrogenase --> redox (NADH and NAD+)
Phosphatase --> removes phosphate
Fate of pyruvate
Aerobic conditions: converted to acetyl CoA
Anaerobic or hypoxic (low oxygen): converted to lactate or ethanol (yeast)
Why produce lactate/ethanol?
Regenerate NAD+ to allow glycolysis to continue
Feeder pathways for glycolysis
di- and polysaccharides (complex sugars) are cleaved to monosaccharides and enter the glycolytic pathway at different points.
- Endogenous and dietary glycogen have different entry points
Where does gluconeogenesis occur?
Liver and not in muscle
Substrates for gluconeogenesis?
Lactate, amino acids (NOT fatty acids)
Is gluconeogenesis energy cheap or costly?
Costly
Is glycolysis in the cytosol or mitochondria?
Glucose to pyruvate in cytosol
When we want to reverse from pyruvate to glucose, first reaction occurs in mitochondria (pyruvate can go between cytosol and mitochondria)
Role of liver in metabolism
Maintains blood glucose level
- If low, hormone glucagon is secreted
- If high, hormone insulin is released
Pentose phosphate pathway purpose
Produces 5 carbon sugars
NADPH production
Enzyme for glycogen degradation?
Glycogen phosphorylase
Substrate: glycogen
Product: glucose-1-phosphate
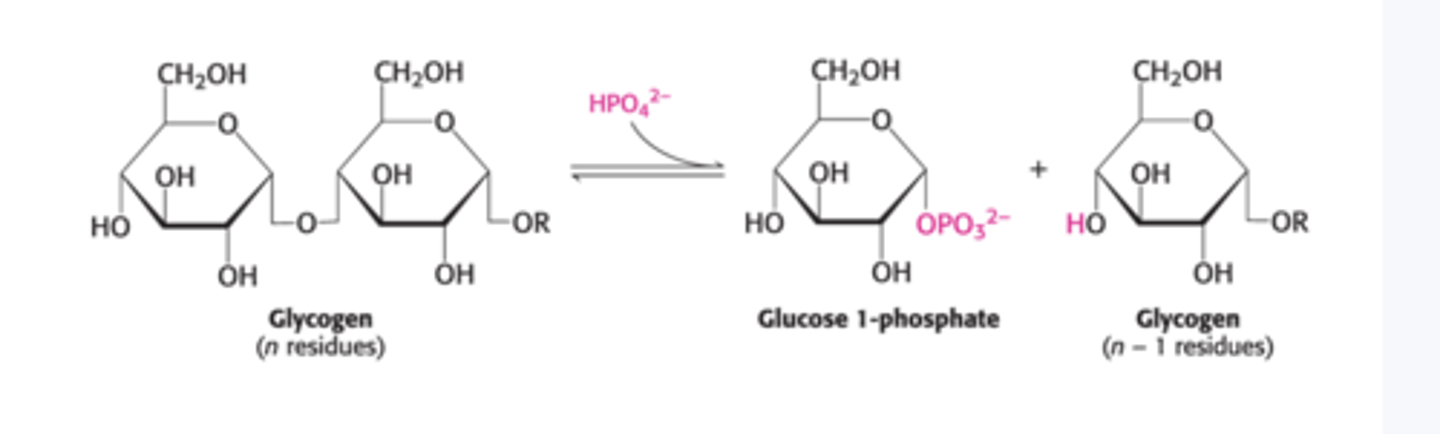
Roles of debranching enzyme?
Removes branches and put it on the main chain (transfers four glucose units); allows glycogen phosphorylase to continue and cleaves off a glucose (comes off as pure glucose)
Enzyme for glycogen synthesis?
Glycogen broke down to glucose-1-phosphate with help of inorganic phosphate
Glycogen synthase
What initiates glycogen synthesis?
Glycogenin
Hormone that promotes glycogen synthesis
Insulin
Effect of phosphorylation on glycogen metabolism?
- Activates breakdown
- Inhibits synthesis
Location of citric acid cycle?
Mitochondrial matrix, and its released into the citric acid cycle with pyruvate dehydrogenase complex
What does pyruvate dehydrogenase produce?
Acetyl-CoA (first reaction)
Products per cycle?
3 NADH
1 FADH2
1 GTP/ATP
2 CO2
Role of citric acid cycle?
Oxidize acetyl-CoA --> energy carriers
Number of carbons entering cycle?
2 (acetyl-CoA) and 3 (pyruvate)
Regulation occurs at which enzymes?
Key irreversible steps
What is standard reduction potential (E°′)?
Tendency to gain electrons
What determines if redox reaction is favorable?
Positive ΔE°′ → negative ΔG°′
Final electron acceptor in ETC?
O₂ → H₂O
Electron donors to ETC
NADH, FADH2
What do Complexes I-IV do?
Transfer electrons + pump protons
What is the proton gradient used for?
ATP synthesis
Enzyme that makes ATP
ATP synthase (F₀F₁)
P/O ratio (ATP per NADH)
~2.5
P/O ratio (ATP per FADH2)
~1.5
Approximate ATP per glucose
~30-32 ATP