Module 3 (pharm)
1/157
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
158 Terms
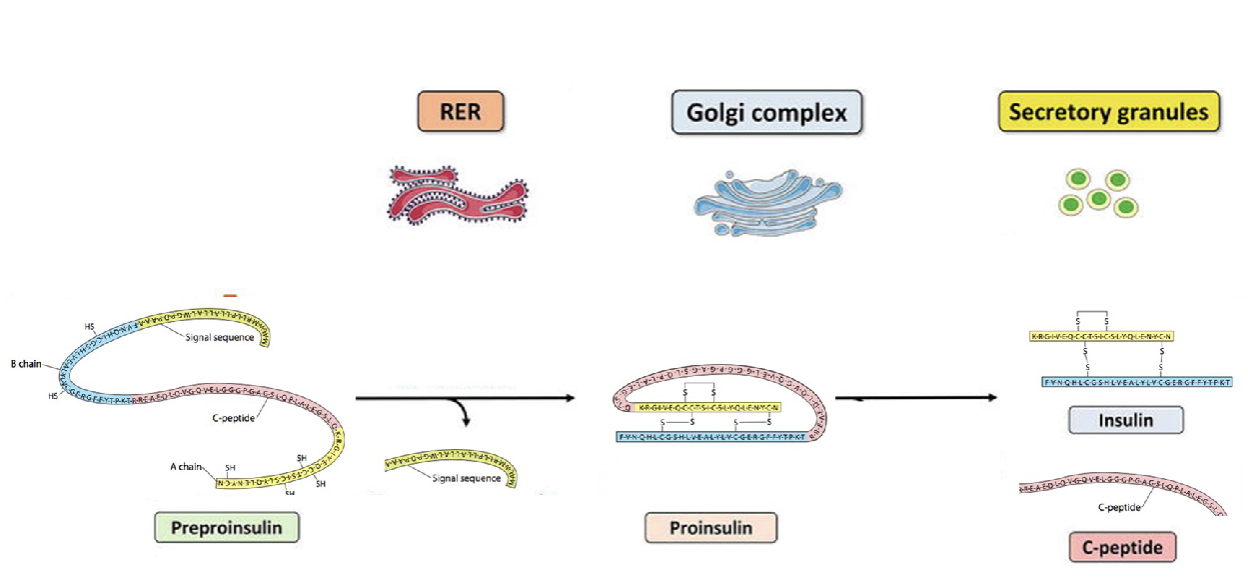
Describe the synthesis of insulin (location, intermediate)
Preproinsulin is translated into proinsulin in the rough ER
A-chain ad B- chain fused together with 3 disulfide bond
Folded proinsulin goes to the golgi where its enzymatically cut into mature insulin
Gets packaged into secretory vesicles
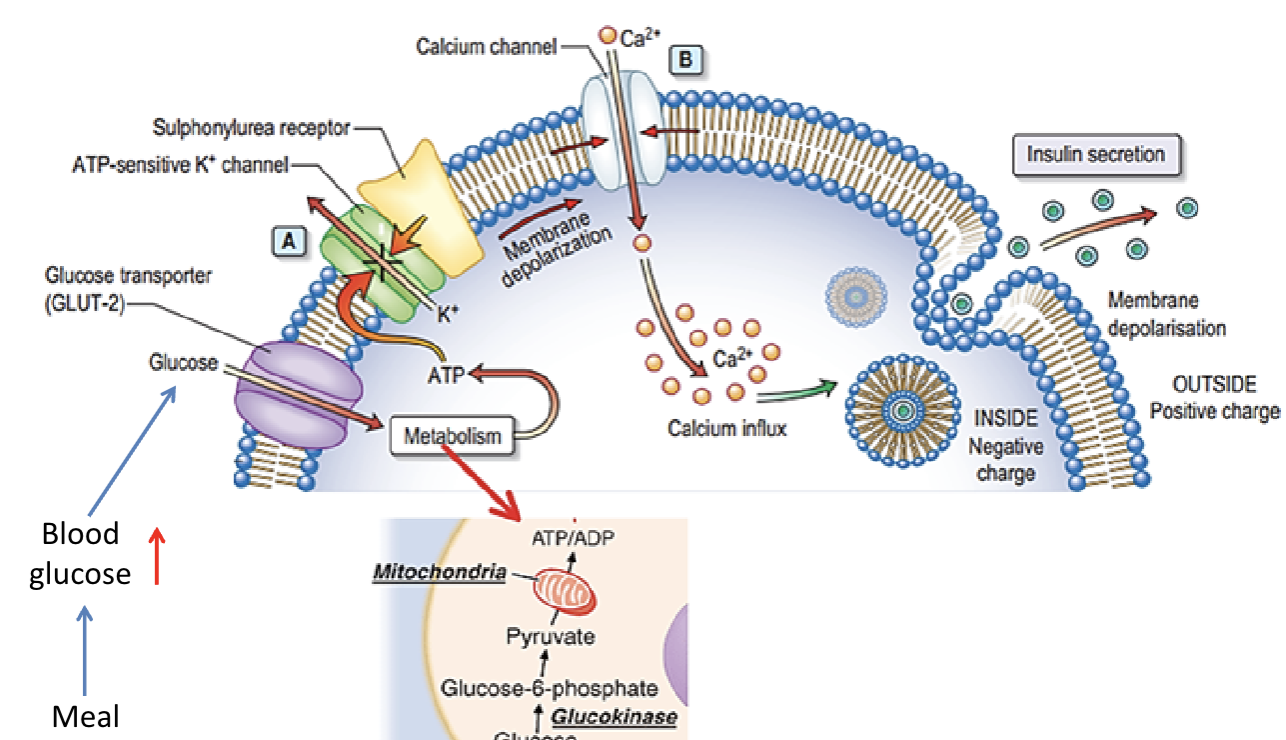
Describe the mechanism through which insulin is secreted:
Glucose comes into through glut 2 (B cells)
ATP/ADP ratio increases causing K+ channel to shut (so K+ can’t go out)
Depolarizes the cell
Ca+ channel open and Ca2+ comes into the cell
Insulin-containing granule fuses with the membrane and insulin is released
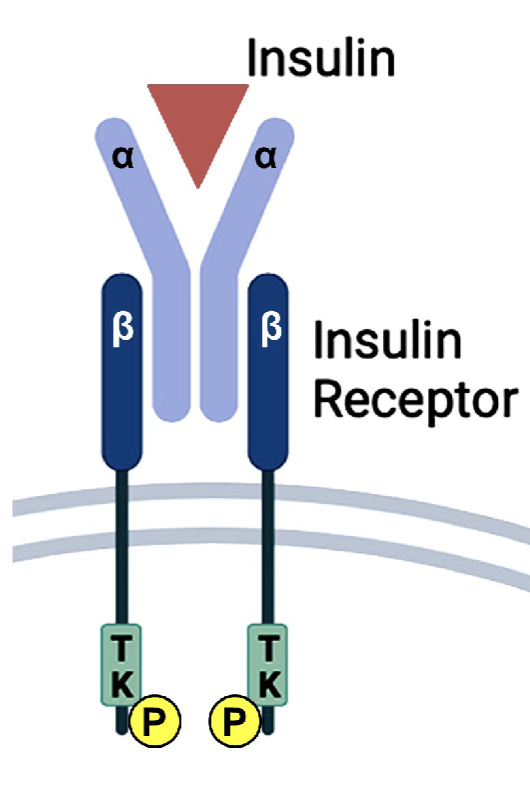
Insulin receptor is what kind of receptor? Binding of insulin to this receptor does what?
kinase-linked receptor:
causes intracellular enzymatic activity of intracellular receptor domain and recruit other molecules intracellular to cause a action
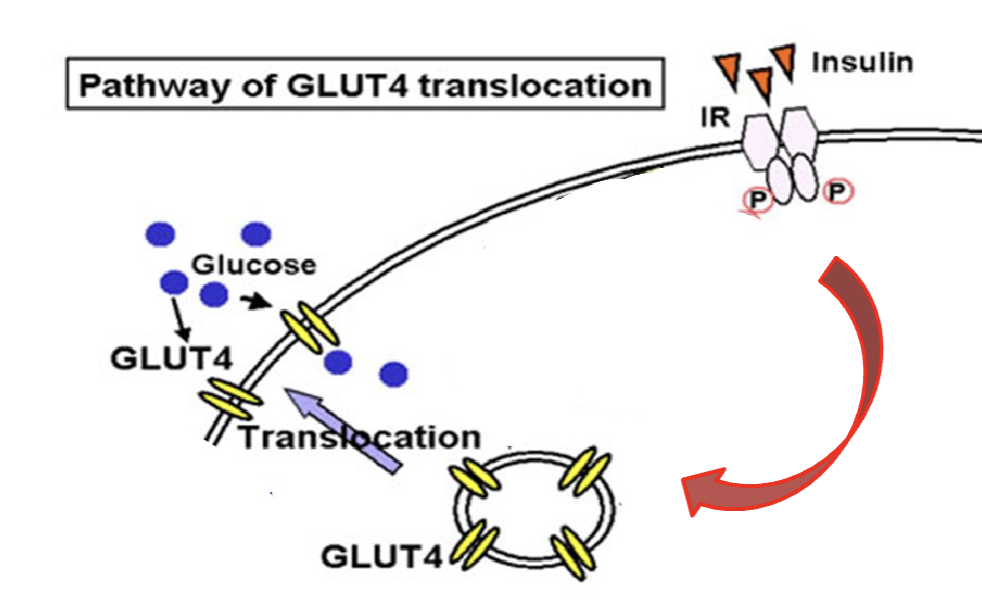
How does insulin induce translocation of GLUT4 to plasma membrane in skeletal muscle and adipocyte?
Insulin bind to insulin receptor and becomes activated
Translocation of glucose transporter (GLUT4) occur to the plasma membrane
Glucose come in to cell via GLUT4
True or false: insulin-IRs binding does induce translocation of GLUT-4 in hepatocytes
False: insulin-IRs binding does not induce translocation of GLUT-4 in hepatocytes
How does insulin effect glucose uptake and glycogen production in skeletal muscle:
Skeletal muscle:
↑ glucose uptake
↑ glycogen production
How does insulin effect glucose uptake and fat storage production in adipose tissue:
Adipose tissue:
↑ glucose uptake
↑ fat storage
How does insulin effect glucose production and glycogen production in liver:
Liver
↓ glucose production (gluconeogenesis)
↑ glycogen production
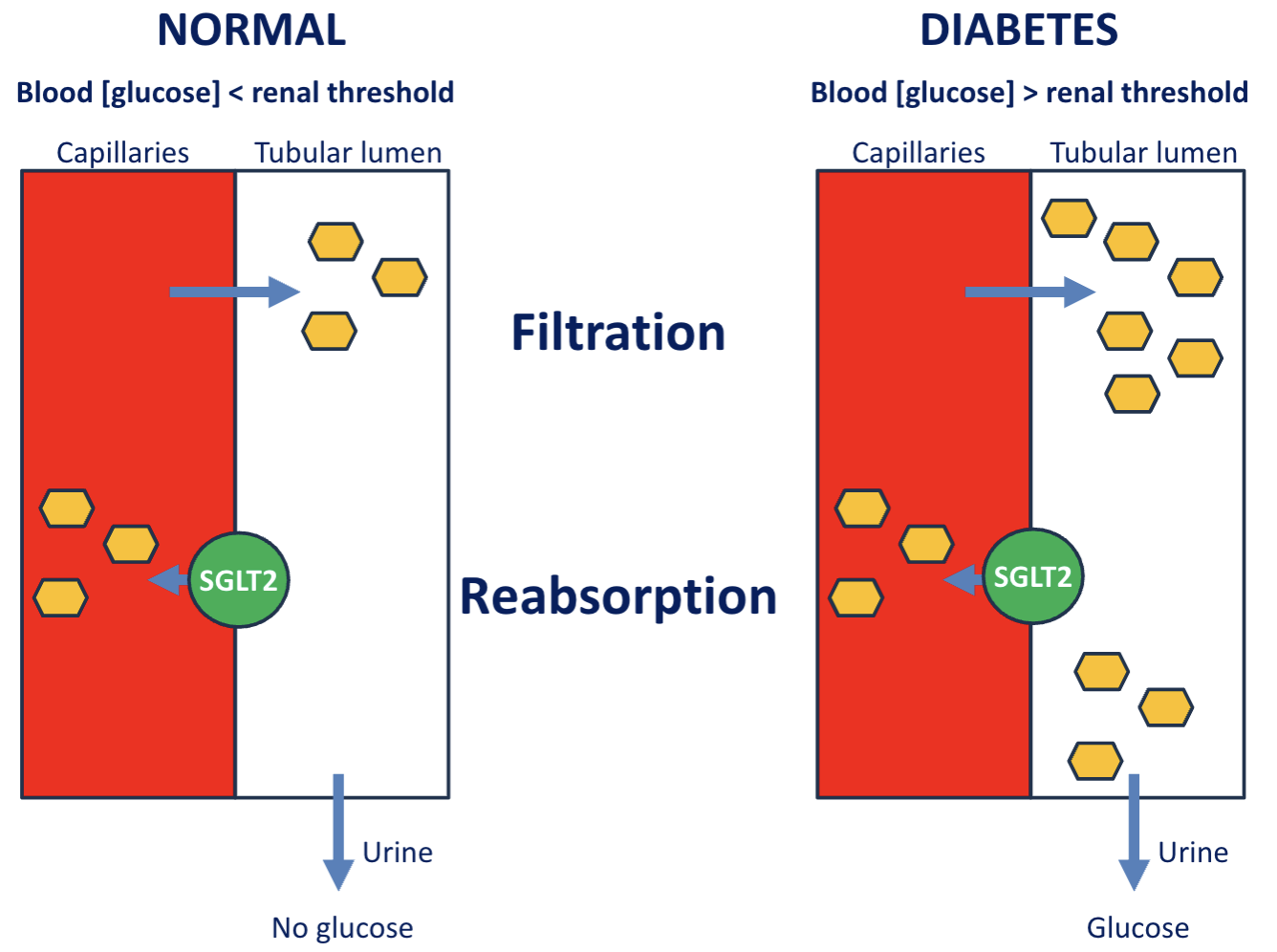
Why does patients with diabetes experience frequent urination?
Not all glucose are reabsorbed by the SGLT in kidney
Glucose in lumen increases drawing water out
High volume of urine
Describe how A1C is used as glycemic test; does it require fasting?
Measure a Hb that is linked to a glucose (glycated Hb), measures average blood sugar level over the past 2 or 3 month
*Does not require to fast
Performed after 8-12 hours of fasting.
Single time-point measurement assessing glucose at that specific moment or condition
Fasting Plasma Glucose (FPG) Test
2 Test that measure Single time-point measurement assessing glucose at that specific moment or condition
Fasting Plasma Glucose (FPG) Test
Oral glucose tolerance test (OGTT)
How does insulin resistance effect circulating glucose, FFA, and glucose uptake in target organ:
Increased circulating glucose and FFA
Decreased glucose uptake in target organs
What 2 things causes low grade inflammation:
adipocytes undergoing
1.hypertrophy (adipocyte enlargement)
hyperplasia (proliferation of new adipocytes)
What’s low-grade inflammation?
It’s chronic low-grade production of pro-inflammatory
factors (adipokines, chemokines and free fatty acids) into the circulation
What’s Lipotoxicity?
accumulation of lipids (e.g. free fatty acids) in non-
adipose tissue, such as liver, muscles, pancreas
What happens when lipid accumulate in non-adipose tissue?
Insulin resistance
B-cell dysfunction
What are adverse effect of insulin treatment: (2)
Hypoglycemia
Weight gain
When do you use insulin as a treatment:
when B cell fails to produce insulin
What are insulin sensitizers used for?
lower glycemia by ↑ insulin sensitivity in target organs
What are insulin secretagogues used for?
To enhance insulin secretion
Insulin sensitizers effect on hyperinsulinemia and hypoglycemia
Insulin Sensitizers do not release insulin therefore they do not cause
hyperinsulinemia and hypoglycemia (unless given in combination with
secretagogues)
initial drug of choice for type 2 diabetes
Biguanides (metformin)- sensitizer
Mechanism of action for Biguanides (metformin)
↓ postprandial and fasting glucose levels (does not promote insulin secretion)
↑ glucose uptake (GLUT4-mediated)
↑ glycogen production
↓ hepatic gluconeogenesis
Adverse effect of Biguanides (metformin)
Largely gastrointestinal.
Loss of appetite and weight loss
what happens if Biguanides (metformin) is taken with insulin?
Hypoglycemia
Thiazolidinediones is what type of anti-diabetic drug? and its mechanism of action?
It’s a senitizers, mechanism of action:
Activates PPARγ
PPARγ receptor regulates transcription of genes resulting in increased insulin sensitivity in adipose tissue, liver, and skeletal muscle
↓ postprandial and fasting glucose levels (does not promote insulin secretion)
↑ glucose uptake (GLUT4-mediated)
↑ glycogen production
↓ hepatic gluconeogenesis
Adverse effect of Thiazolidinediones
Weight gain
Fluid retention
Sulfonylureas is what type of anti-diabetic drug? and its mechanism of action?
It’s a secretagogue (GLUCOSE-INDEPENDENT)
Sulfonylureas block ATP-sensitive K+ channels,
resulting in depolarization, Ca2+ influx
insulin exocytosis
Adverse effect of Sulfonylureas
Hypoglycemia
Weight gain
Glinide is what type of anti-diabetic drug? and its mechanism of action?
It’s a secretagogue (GLUCOSE-INDEPENDENT)
block ATP-sensitive K+ channels
resulting in depolarization
Ca2+ influx
insulin exocytosis
Adverse effect of glinide:
1.Hypoglycemia
Weight gain
What are the 2 incretin secreted by GI tract?
Glucagon-like peptide 1 (GLP-1)
Glucose-dependent insulinotropic polypeptide (GIP)
GLP-1/GIPR agonist is what type of anti-diabetic drug? and its mechanism of action?
It’s a secretagogue (GLUCOSE-DEPENDENT)
incretin bind to GPCR (Gs-coupled GLP-1R/GIPR in the β-cells)
Gctivation of GLP-1R/GIPR is responsible for
GLUCOSE DEPENDENT RELEASE of insulin
What inactives GLP-1/GIP?
Dipeptidyl peptidase 4 (DPP-4)
When can GLP-1/GIP become resistant to DPP-4? And its effect on GLP-1/GIP concentration:
Selective modification of key amino acids renders these peptides resistant to DPP-4 degradation, thereby extending their circulating half-life
Incretin- and incretin related drugs are what kind of molecule and what improves their oral bioavailability?
—> they are polypeptides
—> Salcaprozate sodium and related compound improve oral bioavailability of peptides
Adverse effect of Incretins and Incretin-related drugs:
Weight loss
GI issue: nausea, vomiting
Low incidence of hypoglycemia
Why does Incretin-related drugs cause nausea and vomiting:
it acts on the GLP-1R → Nausea- promoting in the Chemoreceptor trigger zone (CTZ)
Why is it dangerous to give incretin-related drugs and general anesthesia?
Protective airway reflexes may be lost
risk of regurgitation & aspiration
↑ risk of pneumonia or acute respiratory
distress
DPP-4 inhibitor is what type of anti-diabetic drug? and its mechanism of action?
It’s a secretagogue (GLUCOSE-DEPENDENT)
Prevent GLP-1/GIP inactivation
produce 2- to 3-fold levels of active GLP-1/GIP
GLP-1/GIP binds to GLP-1R/GIPR
Allows insulin to be secreted
DPP-4 inhibitors adverse effect:
doesn’t cause satiety, or fullness
Upper respiratory infections
α-Glucosidase Inhibitors mechanism of action:
α-glucosidase is an intestine enzyme which breaks down carbohydrates into glucose and other simple sugars that can be absorbed
α-glucosidase inhibitors block the intestinal breakdown of complex carbohydrates and interferes with their absorption, resulting in lower postprandial glucose levels
Adverse effect of α-Glucosidase Inhibitors
GI effects, e.g. flatulence, diarrhea, and abdominal cramping.
Doesn’t cause hypoglycemia when used as monotherapy.
When used with insulin secretagogues or insulin, hypoglycemia may
develop
Mechanism of action of SGLT-2 Inhibitors:
decrease reabsorption of glucose,
increase urinary glucose excretion
lower blood glucose
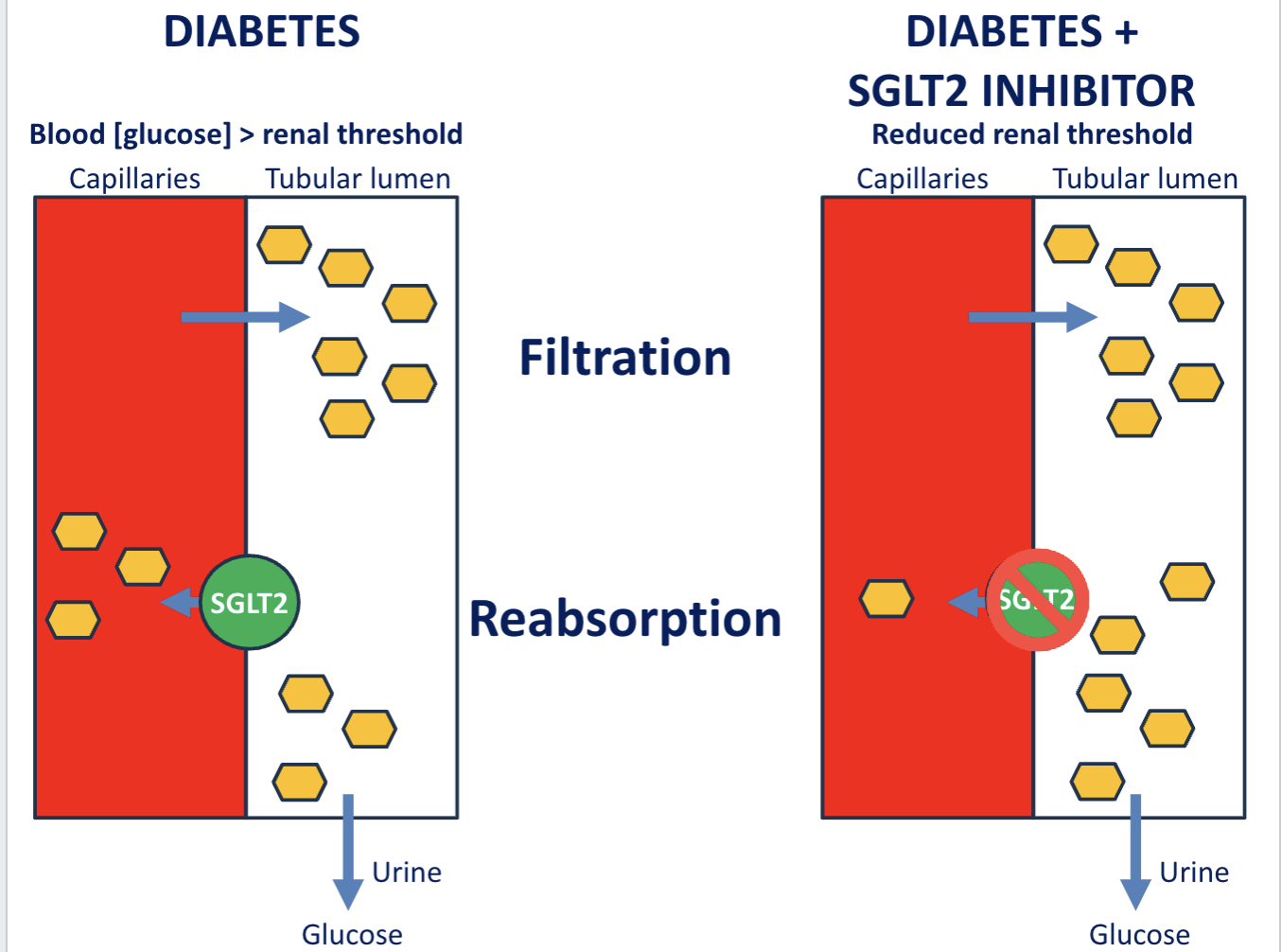
Adverse effect of SGLT-2 Inhibitors
weight loss
Urinary tract infections (especially in women)
Lack of drug efficacy for organism/ neoplastic cell
natural resistance
Loss of drug efficacy over course of treatment in a patient or in a population
Acquired resistance
Acquired resistance develop 3 ways, what are they?
Gene amplification
Gene mutation
Gene transfer in bacteria
What are the 3 form of gene transfer? describe each:
Conjugation: transfer of R plasmid between bacterial cells
Transposons: transfer of resistance gene between plasmid and plasmid, chromosome
Transduction: transfer of resistance gene from bacteriophage and bacteria
What are 4 drug target sites in bacteria?
Folate synthesis in cytoplasm
Protein synthesis by ribosome
Nucleic acid synthesis
Cell wall synthesis
Funny People Need Coffee
what does B lactams do
kill bacteria by inhibiting cell wall synthesis
Function of B-lactamases
enzyme that inactive B-lactams ring (penicilin) by cutting opening the ring —> drug can no longer bind to the active site
Transpeptidases that cross-link peptidoglycan polymer in final step of cell formation—> giving cell wall rigidity, what drug messes with this function?
Penicillin binding protein —> penicilin
2 class of drug that bind to transpeptidases and block cross-linking of peptidoglycan polymers in cell wall synthesis
penicilins
Cephalosporins
penicilins and Cephalosporins which are both used to block cell wall synthesis in bacteria, how do they differ? (3)
Spectrum
Susceptibility to B-lactamases
Oral bioavailability
Who has more peptidoglycan layer and what does this mean?
Gram + and it provides high structural integrity against osmotic pressure
True or false: both gram (+) and (-) have penicillin binding protein (PBP)
True
required cofactor in the synthesis of pyrimindine dTMP for DNA replication
THF
In bacteria THF is formed from
PABA
In human THF formed FROM
DHF
What enzyme converts DHF to THF?
DHF reductase
What is function of sulfonamides?
Analogues of substrate PABA which inhibit DHF synthetase in bacteria
Bacterial Protein synthesis inhibitors interact with what
30 S or 50 S ribosomal subunit
Treatment of infection, documented by culture of microbes and determination of susceptibility to drugs is what type of treatment
Definitive
Treatment of infection based on most likely microbes and their probable susceptibility/ you don’t know what’s causing the problem but making a best guess in terms whats likely to help is what type of treatment
Empiric
Therapy to prevent infection
Prophylactic
What are some mechanism of resistance? (4)
Reduce intercellular level of the drug by decreasing uptake and increasing reflux
Inactivation of the drug
Alter drug target by increasing amount and decreasing affinity to drug
Increase repair of the drug effect
True or false ; overuse of anti-bacterial medication contributes to resistance
True, estimated 30% inappropriate antibacterial prescribing, especially anti-vectorial agents for viral infection
What is the function of reverse transcript test in HIV?
A retrovirus with two identical, single-stranded RNA replicated by reverse transcriptase into DNA to be incorporated into the host DNA
What are some site of actions of anti-HIV agent (4)
Entry inhibitor
reverse transcription inhibitor
integrase inhibitor
protease inhibitor
What’s zidovudine (AZT) and what is it used for?
Reverse transcriptase inhibitor and it is used as an antiviral agent drugs for HIV
Courteous inhibitors are cleared by
CYP3 A4 which is a enzyme in the liver and intestinal track that metabolizes drug
What is the benefit of giving prose inhibitors along with CYP3 A4 inhibitors as well
It increases the efficacy of protein, inhibitors by slower, elimination, causing boosting of the drug effect
Highly active oral antiretroviral therapy (HAART) is used for
long term regimen treatment for HIV and it requires different drugs from different classes
Non-B hepatitis affecting blood transfusion, recipients and IV drug users and it is a single stranded RNA virus
HCV infection
Cause of liver failure, liver, cancer, and need for transplant
HCV infection
Name three treatment for HCV
Protease inhibitor
NS 5A replication complex in inhibitor
NS 5B polymerase inhibitor
How does protease inhibitor in HCV treatment work?
It binds and inhibits the viral NS3/4A protease blocking the release of viral protein required for replication from large precursor
Mechanism of NS 5B polymerase inhibitor in HCV treatment
Block RNA dependent RNA polymers, preventing the synthesis of new viral RNA
-chain Terminator
Mechanism of NS 5A replication complex in inhibitor in HCV treatment
Block viral RNA replication and assembly
What are three antimalarial agent?
chloroquine e type (chloroquine)
Artemisinins
Primaquine-type (primaquine)
What is a possible treatment of respiratory illness COVID-19 due to SARS-CoV-2 infection
Protein inhibitor with CYP3 A4 inhibitor
What are 2 treatment targets for malaria?
Target schizonts in erythrocytes
Target hypnozoites in liver
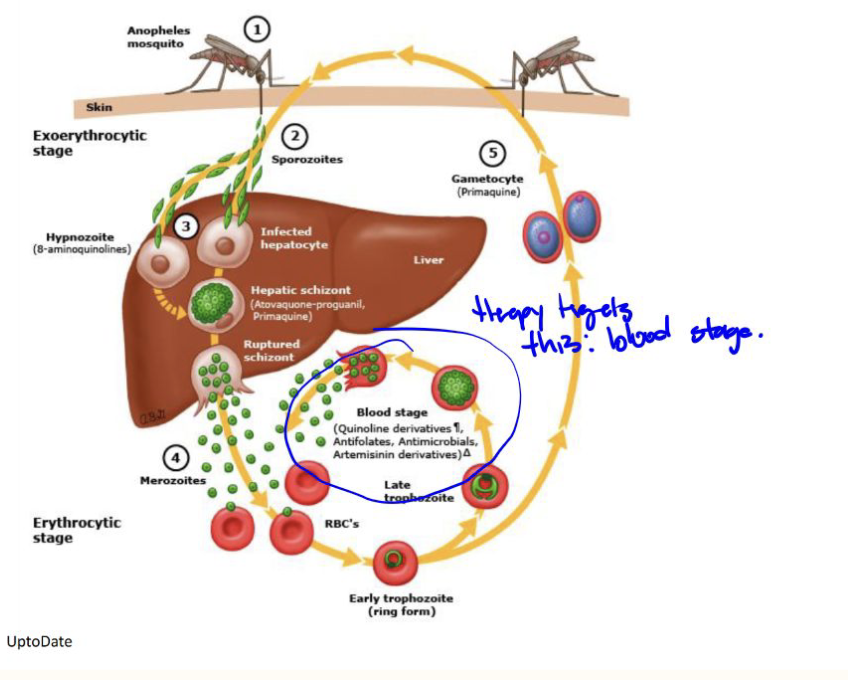
Chloroquine e type (chloroquine) used as anti-parasitic drug. How does it work?
Parasite digests hemoglobin
Inside RBCs, Plasmodium breaks down hemoglobin for nutrients.
This releases free heme (Fe²⁺-protoporphyrin IX) → toxic to the parasite.
Parasite detoxifies heme
It normally converts toxic heme → hemozoin (non-toxic crystal).
Chloroquine blocks this detox step
Chloroquine accumulates in the parasite’s acidic food vacuole.
It inhibits conversion of heme → hemozoin.
Toxic buildup → parasite death
Free heme + chloroquine–heme complexes damage membranes and proteins.
This leads to oxidative damage and parasite death
Artemisinins is antimalarial agent, describe its mechanism of action:
Heme induced endoperoxide opening generates toxic free radicals
Toxic for radicals are lethal for parasites
Targets liver stages (hypnozoites) of:
Plasmodium vivax
Plasmodium ovale
Prevents relapse
primaquine
Efficacy of anti-malarial drug—>primaquine is due to
CYP2D6 hydroxylation and CYP-reductase generation of H2O2
poisons made by plants and animals
Toxins
any toxic substance, including man-made poisons
Toxicants
Regulatory agency responsible for overseeing pesticides, industrial chemicals, hazardous waste, pollutants in air, water, and soil from manufacturing waste.
Environmental Protection Agency (EPA)
Regulatory agency responsible for overseeing Safety Data Sheets
for each hazardous chemical in standard format to communicate information on hazards in workplace
Occupational Safety and Health Administration (OSHA)
Provides EPA with authority to require reporting, record-keeping and testing requirements, and restrictions relating to chemical substances and/or mixtures
Toxic Substance Control Act
Importance in setting allowable levels of chemicals in air, water, workplace by EPA and OSHA
Regulatory Risk Assessment
Challenge of extrapolation from Regulatory Risk Assessment:
Animals to human
Experimental dose
High acute toxicity does not necessarily equal
high chronic toxicity
Low acute toxicity does not necessarily equal
low chronic toxicity
Define LD50
Amount of a toxic substance required to kill an individual
How does electrophilic alkylating agents cause toxicity? (mechanism)
They alkylate DNA, which then does not uncoil properly.
• Results in cytotoxicity, inhibition of cell growth, initiation of apoptosis, and risk of carcinogenic mutations.
How does oxidative stress cause toxicity:
Xenobiotic foreign to body
Goes through bioactivation —> generate free radical—> Lipid radical—> damages cell membrane