CHM2045 Exam 3
1/183
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
184 Terms
How are atoms arranged in Lewis Structures (EN)
Least EN in the middle (never Hydrogen)
Hydrogen Octet Rule Exception
Okay with 2 electrons
Be Exception to octet rule
Only needs 4 electrons, but can have more
B exception to octet rule
Only needs 6 electrons, but can have more
Oxygen and Nitrogen exception to octet rule
Like to have 8 electrons, but can have 7
Period 3 and beyond exception to octet rule
Nonmetals in period 3 and beyond can have more than 8 electrons in the octent because they can use their d orbitals to make bonds
Formal charge formula
# of VE - # of electrons around the atom - # of bonds (count each line)
Where should the negative charge be in a lewis structure
On the most EN atom
What is resonance
All the ways a molecule can lok
What does A stand for in AXmEn
Central Atom
What does X stand for in AXmEn
How many atoms are bonded to the central atom (A)
What does E stand for in AXmEn
Number of lone pairs around the central atom
Shortcut to finding the shapes
Count VE
Subtract by highest multiple of 8 (divide by 8 to get the X)
Take the leftover electrons and divide by 2 (to get the E)
AX2 Shape
Linear
Linear (AX2) Ideal Bond Angle
180
Linear hybridization
sp
AX3 shape
Trigonal Planar
Trigonal Planar (AX3) ideal bond angle
120
Trigonal planar hybridization
sp2
AX2E1 shape
Bent
Bent (AX2E1) bond angle
<120 (117)
Bent (AX2E1) hybridization
sp2
AX4 Shape
Tetrahedral
Tetrahedral (AX4) Bond Angle
109.5
Tetrahedral (AX4) hybridization
sp3
AX3E1 Shape
Trigonal Pyramidal
Trigonal Pyramidal (AX3E1) Bond Angle
<109.5 (107)
Trigonal Pyramidal (AX3E1 ) hybridization
sp3
AX2E2 Shape
Bent
Bent (AX2E2 ) Bond Angle
<109.5 (105)
Bent (AX2E2 ) Hybridization
sp3
Electron-pair delocalization
Double bonds are spread over the entire molecule
AX5 Shape
Trigonal Bipyramidal
Trigonal Bipyramidal (AX5 )Bond Angle
90, 120, and 180 bond angles
Trigonal Bipyramidal (AX5) Hybridization
sp3d
AX4E1 Shape
See-Saw
See-Saw (AX4E1 ) Bond Angle
90, 117, and 180
See-Saw (AX4E1 )Hybridization
sp3d
T-Shaped Planar (AX3E2 ) Shape
T-Shaped Planar
T-Shaped Planar (AX3E2 ) Bond Angle
90, 180
T-Shaped Planar (AX3E2 ) Hybridization
sp3d
AX2E3 Shape
Linear
Linear (AX2E3 ) Bond Angle
180
Linear (AX2E3 ) Hybridization
sp3d
AX6 Shape
Octahedral
Octahedral (AX6) Bond Angle
90
Octahedral (AX6) Hybridization
sp3d2
AX6E1 Shape
Square Pyramidal
Square Pyramidal (AX6E1 ) Bond Angle
90
Square Pyramidal (AX6E1 ) Hybridization
sp3d2
AX4E2 Shape
Square Planar
Square Planar (AX4E2 ) Bond Angle
90
Square Planar (AX4E2 ) Hybridization
sp3d2
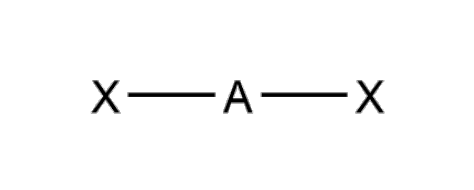
What is the molecular geometry and formula of this molecule?
Linear: AX2
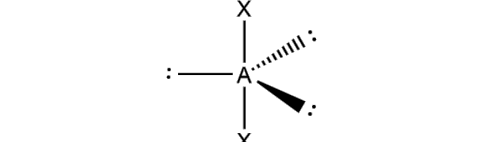
What is the molecular geometry and formula of this molecule?
Linear: AX2E3
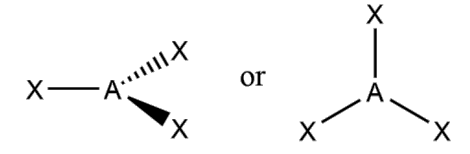
What is the molecular geometry and formula of this molecule?
Trigonal Planar: AX3
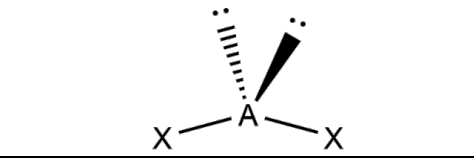
What is the molecular geometry and formula of this molecule?
Bent: AX2E1
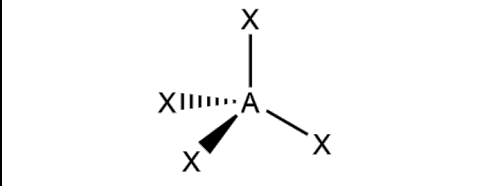
What is the molecular geometry and formula of this molecule?
AX4
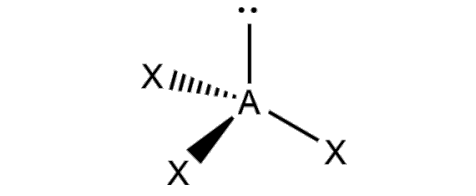
What is the molecular geometry and formula of this molecule?
Trigonal Pyramidal: AX3E1
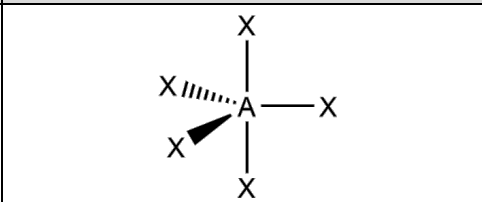
What is the molecular geometry and formula of this molecule?
Trigonal Bipyramidal: AX5
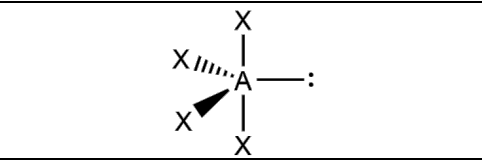
What is the molecular geometry and formula of this molecule?
See-Saw: AX4E1
What is the molecular geometry and formula of this molecule?
T-Shaped: AX3E2
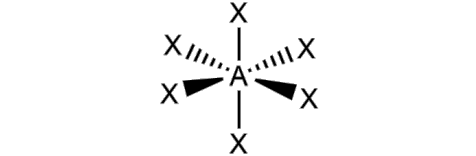
What is the molecular geometry and formula of this molecule?
Octahedral: AX6
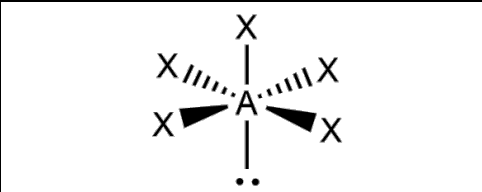
What is the molecular geometry and formula of this molecule?
Square Pyramidal: AX5E1
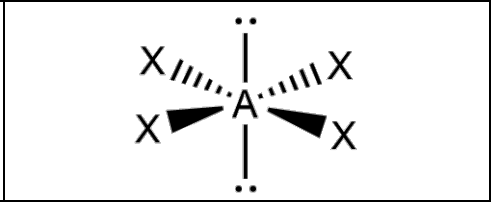
What is the molecular geometry and formula of this molecule?
Square Planar: AX4E2
What is the electron geometry of: Linear
Linear
What is the electron geometry of: Trigonal Planar
Trigonal Planar
What is the electron geometry of: Bent
Trigonal Planar
What is the electron geometry of: Tetrahedral
Tetrahedral
What is the electron geometry of: Trigonal Pyramidal
Tetrahedral
What is the electron geometry of: Bent (AX2E2)
Tetrahedral
What is the electron geometry of: Trigonal Bipyramidal
Trigonal Bipyramidal
What is the electron geometry of: See-Saw
Trigonal Bipyramidal
What is the electron geometry of: T-Shaped Planar
Trigonal Bipyramidal
What is the electron geometry of: Linear (AX2E3)
Trigonal Bipyramidal
What is the electron geometry of: Octahedral
Octahedral
What is the electron geometry of: Square Pyramidal
Octahedral
What is polarity?
The uneven pulling/sharing of electrons around the central atom
What 2 dipole moment’s can polar molecules have?
Net dipole or non-zero dipole moment
What dipole moment can a nonpolar molecule have?
Zero dipole moment
Fir single center compounds, what can you use to determine if the molecule is polar?
Is the molecular geometry symmetrical
Are the outer atoms the same
Symmetrical Molecular Shapes
AX2‒ Linear, AX3‒ Trigonal Planar, AX4‒ Tetrahedral, AX5‒ Trigonal Bipyramidal ,
AX2E3‒ Linear, AX6‒ Octahedral, AX4E2‒ Square Planar
Asymmetrical Molecular Shapes
AX2E1‒ Bent (V-shaped), AX3E1‒ Trigonal Pyramidal, AX2E2‒ Bent (Water Bent), AX4E1‒ See-Saw, AX3E2‒ T-shaped, AX5E1‒ Square Pyramidal
T/F: When any central atom has one lone pair, it is automatically polar.
True
How to determine the hybridization of a molecule?
Count the number of electron groups around the atom (count double and triple bonds as one)
What is a sigma bond?
Single localized bond in a compound (direct overlap)
For what 3 overlaps can a sigma bond be created?
Overlap of 2 s orbitals, 2 sp orbitals, or an sp orbital with an s orbital.
When is an sp orbital created?
When an s and a p orbital come together to delocalize the electron density around the central atom
Sigma v. Pi bonding in bond rotation
Sigma bond: end-to-end bonding which allows bond rotation about the bond axis
Pi bond: do not allow bond rotation
In a pi bonds, where do the 2 electrons move?
One e moves from the top lobe to the bottom lobe, and the other e moves from the bottom lobe to the top lobe continuously
Pi bonding gives rise to what kinds of isomers
Cis (same side) and trans (opposite side)
What is a cis isomer
When 2 of the same atoms are on the same side of the molecule
What is a trans isomer
When 2 atoms on the same side of the molecule are different. So the 2 atoms that are the same are on opposite sides of the molecule.
T/F: For a pi bond to occur, a sigma bond must occur first
True
Are sigma or pi bonds weaker? Why?
Pi bonds are weaker
Because the orbitals overlap less side to side (pi) than end to end (sigma)
What kinds of bonds does a single bond have?
1 sigma bond
What kinds of bonds does a double bond have?
1 sigma + 1 pi bonds
What kinds of bonds does a triple bond have?
1 sigma + 2 pi bonds
What is paramagnetic
There is at least one unpaired electron in the molecule
What is diamagnetic
All the electrons in the molecular are paired, there are no radicals