Chapter 10 - equilibrium 1
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is dynamic equilibrium?
In a dynamic equilibrium, the reactants and products are dynamic (they are constantly moving)
In a dynamic equilibrium, the rate of the forward reaction is the same as the rate of the backward reaction in a closed system, and the concentrations of the reactants and products are constant (the concs. don’t have to be same with all the particles they just have to be constant)
What is a closed system?
A closed system is one in which none of the reactants or products escape from the reaction mixture
In an open system, matter and energy can be lost to the surroundings
When a reaction takes place entirely in solution, equilibrium can be reached in open flasks as a negligible amount of material is lost through evaporation
If the reaction involves gases, equilibrium can only be reached in a closed system
What is Kc?
The equilibrium constant which gives you an idea of how far to the left or right the equilibrium is
What is the equation for a general reversible reaction?
aA + bB ⇌ cC + dD
The corresponding equilibrium constant expression is written like this:
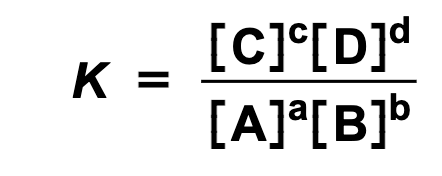
Where:
[A] and [B] = equilibrium reactant concentrations (mol dm-3)
[C] and [D] = equilibrium product concentrations (mol dm-3)
a, b, c and d = number of moles of corresponding reactants and products
The Kc of a reaction is specific and only changes if the temperature of the reaction changes (not if theres a ctalyst, a change in pressure or a change in concentration)
What is a homogenous system and how can you work out Kc for it?
It is a system where everything is in the same physical state
For this, all the producst and reactants are included in the Kc expression

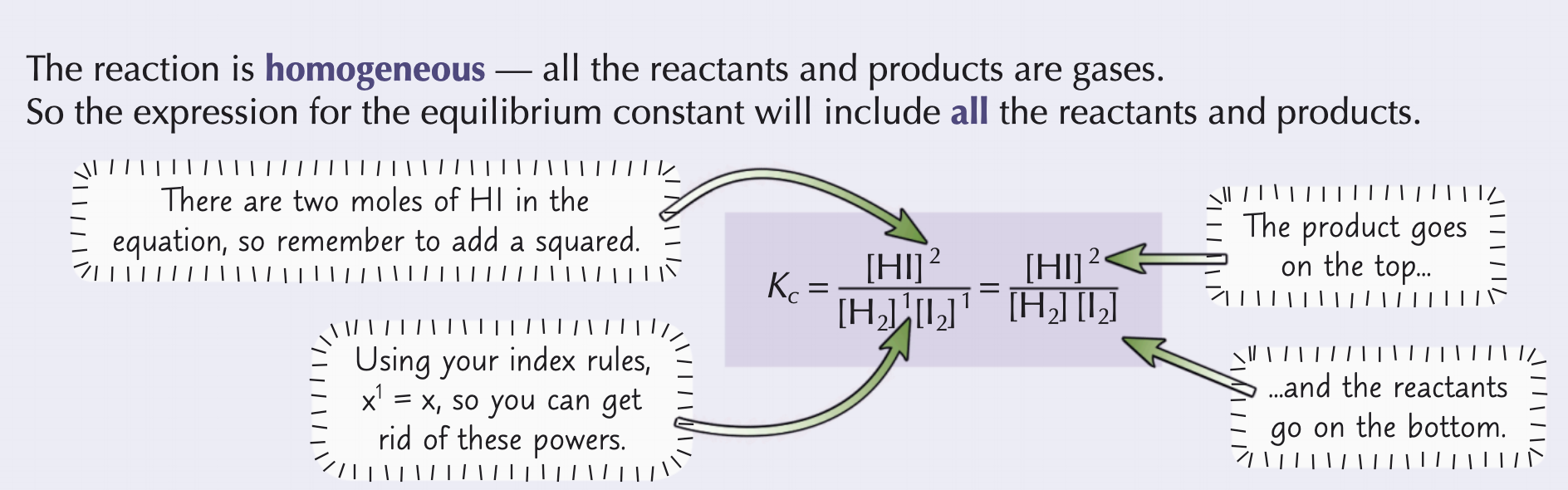
What is a heterogenous system and how can you work out Kc for it?
In a heterogeneous system, not everything's in the same physical state.
Unlike with homogeneous equilibria, not everything is included in the expression for K
You don't include solids or pure liquids in the expression for K, when you're dealing with heterogeneous equilibria. This is because their concentrations stay constant throughout the reaction.
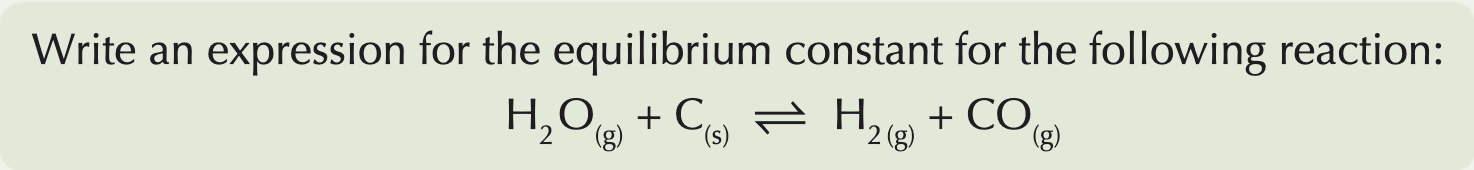
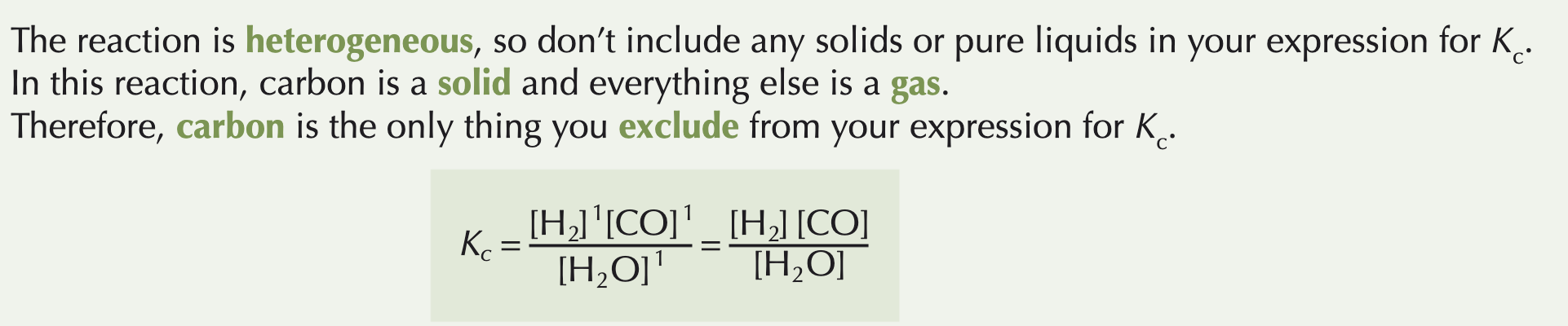
Do you include catalysts in Kc equations?
You don't include catalysts in expressions for the equilibrium constant. Catalysts don't affect the equilibrium concentrations of the products or reactants they just speed up the rate at which dynamic equilibrium is reached.
What does the value of Kc say about the equilibrium position?
Kc = 1 - the position of equilibrium is halfway between the reactants and products
Kc < 1 - the position of equilibrium lies towards the left-hand side, i.e. the reactants
Kc > 1 - the position of equilibrium lies towards the right-hand side, i.e. the products
What is Le Chatelier’s principle?
Le Chatelier’s principle says that if a change is made to a system in dynamic equilibrium, the position of the equilibrium moves to counteract this change
What affect does changing the concentration have on the position of equilibrium?
If you increase the concentration of a reactant, the equilibrium tries to get rid of the extra reactant. It does this by making more product. So the equilibrium's shifted to the right.
If you increase the concentration of the product, the equilibrium tries to remove the extra product. This makes the reverse reaction go faster. So the equilibrium shifts to the left.
Decreasing the concentrations has the opposite effect.
What affect does changing the pressure have on the position of equilibrium? (only for gases)

What affect does changing the temperature have on the position of equilibrium?

Compromise of conditions in industry
Conditions are balanced to ensure high rate and high yield.
What conditions should be used for the reaction of ethene and steam?
