Bio of Cancer Exam 4 (Ch.11-13,J.C.7/8)
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Glucose
Glycolysis:
-Starts with Glucose (6 carbons) → Glycolysis = 2 ATP produced per glucose
-Ends with 2 x Pyruvate ( 3 carbons)
Energy produced per cycle (two cycles per glucose):
• Production of one ATP per cycle
• Production of three NADH per cycle
• Production of one FADH2 per cycle
* Gluconeogenesis can generate glucose as a precursor for synthesizing other carbohydrates
• Therefore, elevated glucose utilization can be used to drive pathways other than oxidative ATP generation
>Electron transport = ~32 ATP produced per glucose
• GLUT1: A membrane transport protein for glucose
Pyruvate
Glycolysis:
-Starts with Glucose (6 carbons)
-Ends with 2 x Pyruvate ( 3 carbons)→ Pyruvate = Output of glycolysis
Krebs Cycle:
Eight step circular enzymatic cascade
• Initial Substrate = Acetyl-CoA (produced from pyruvate by pyruvate dehydrogenase)
• Final Product = Oxaloacetate (recombined with AcetylCoA to continue cycle
- Pyruvate dehydrogenase (PDH) is the enzyme that catalyzes the conversion of pyruvate to Acetyl-CoA
• The availability of oxygen has a major impact on the fate of pyruvate
-During anaerobic respiration pyruvate is converted into lactate by the enzyme lactate dehydrogenase
• LDHA: The lactose dehydrogenase enzyme that forms lactate from pyruvate
• PDK1: An enzyme that phosphorylates/inactivates PDH to prevent the formation of Acetyl-CoA from pyruvate
Acetyl-CoA
Krebs Cycle:
Eight step circular enzymatic cascade
• Initial Substrate = Acetyl-CoA (produced from pyruvate by pyruvate dehydrogenase)
• Final Product = Oxaloacetate (recombined with AcetylCoA to continue cycle)
* Pyruvate dehydrogenase (PDH) is the enzyme that catalyzes the conversion of pyruvate to Acetyl-CoA
* = Input of Krebs cycle (and many other processes)
Metabolic enzyme regulation: Pyruvate Dehydrogenase
For example,
• When the cells is in need of Acetyl-CoA, the expression of PDKs is reduced and their activity is inhibited
• When the cell does not want to produce Acetyl-CoA, the expression of PDKs is increased, and their activity is increased
-Dietary fats are metabolized by the process of β-oxidation
• Converts lipids into Acetyl-CoA
• Aerobic respiration: In the presence of O2 pyruvate is converted to Acetyl-CoA by pyruvate dehydrogenase
Lipogenesis generates phospholipids from Glyceraldehyde-3-phosphoate (glycolysis) and Acetyl-CoA
• PDK1: An enzyme that phosphorylates/inactivates PDH to prevent the formation of Acetyl-CoA from pyruvate
Lactate
• Anaerobic respiration: In the absence of O2 , pyruvate is converted to lactate by the enzyme lactate dehydrogenase
The importance of lactate production is one of four explanations for the importance of the Warburg Effect
The Warburg Effect: The importance of lactate
-During anaerobic respiration pyruvate is converted into lactate by the enzyme lactate dehydrogenase-
1.) Regeneration of NAD+
• Continual glycolytic flux requires a constant supply of NAD+
2.)Acidification of the extracellular space
• Lactate export also exports H+ ions
• Acidification aids metastasis by facilitating ECM breakdown
3.)Lactate induces angiogenesis
• Similar to VEGF, extracellular lactate can signal to endothelial cells to undergo angiogenesis
• LDHA: The lactose dehydrogenase enzyme that forms lactate from pyruvate
Pyruvate Dehydrogenase
Krebs Cycle:
Eight step circular enzymatic cascade
• Initial Substrate = Acetyl-CoA (produced from pyruvate by pyruvate dehydrogenase)
• Final Product = Oxaloacetate (recombined with AcetylCoA to continue cycle
- Pyruvate dehydrogenase (PDH) is the enzyme that catalyzes the conversion of pyruvate to Acetyl-CoA
Metabolic enzyme regulation: Pyruvate Dehydrogenase
PDH activity is tightly regulated by two types of proteins:
1.) PDH Kinases (PDKs) phosphorylate and inactivate PDH
2.) PDH Phosphatases (PDPs) dephosphorylate and activate PDH
• The expression and activity of PDKs and PDPs are highly regulated
For example,
• When the cells is in need of Acetyl-CoA, the expression of PDKs is reduced and their activity is inhibited
• When the cell does not want to produce Acetyl-CoA, the expression of PDKs is increased, and their activity is increased
• Aerobic respiration: In the presence of O2 pyruvate is converted to Acetyl-CoA by pyruvate dehydrogenase
PDKs/PDPs
• PDH Kinases (PDKs) phosphorylate and inactivate PDH
• PDH Phosphatases (PDPs) dephosphorylate and activate PDH
• The expression and activity of PDKs and PDPs are highly regulated
Aerobic metabolism
Glycolysis→Kerbs→ ETC = ~32 ATP
uses oxygen
Anaerobic metabolism
Which pathways generates ATP faster: aerobic or anaerobic metabolism? Anaerobic (glycolysis)
glycolysis in the cytosol= ~2 ATP
Warburg Effect
• In the 1920s, Otto Warburg discovered the tumor cells utilize anaerobic metabolic pathways even in the presence of ample oxygen availability!
• This phenomenon is now known as the Warburg effect
Four explanations for the importance of the Warburg Effect
1) The rate of ATP production
2) Metabolic intermediates are used for biosynthesis
3) The dangers of oxygen metabolism
4) The importance of lactate production
Four explanations for the importance of the Warburg Effect
1) The rate of ATP production
2) Metabolic intermediates are used for biosynthesis
The Warburg Effect: The rate of ATP production
• Provided ample supply of glucose, glycolysis can generate ATP faster than glycolysis + Krebs + ETC
• Tumor cells still use Krebs/ETC, but it is not as essential as you might expect
Metabolic intermediates are used for biosynthesis
Elevated glycolytic flux drives anabolic pathways
• Most anabolic pathways begin with chemical intermediates from glycolysis/Krebs cycle
Anabolic pathways to know:
- The pentose phosphate pathway (PPP) generates nucleotides for DNA synthesis
-Lipogenesis generates phospholipids from Glyceraldehyde-3-phosphoate (glycolysis) and Acetyl-CoA
-Gluconeogenesis can generate glucose as a precursor for synthesizing other carbohydrates
• Therefore, elevated glucose utilization can be used to drive pathways other than oxidative ATP generation
Four explanations for the importance of the Warburg Effect
3) The dangers of oxygen metabolism
4) The importance of lactate production
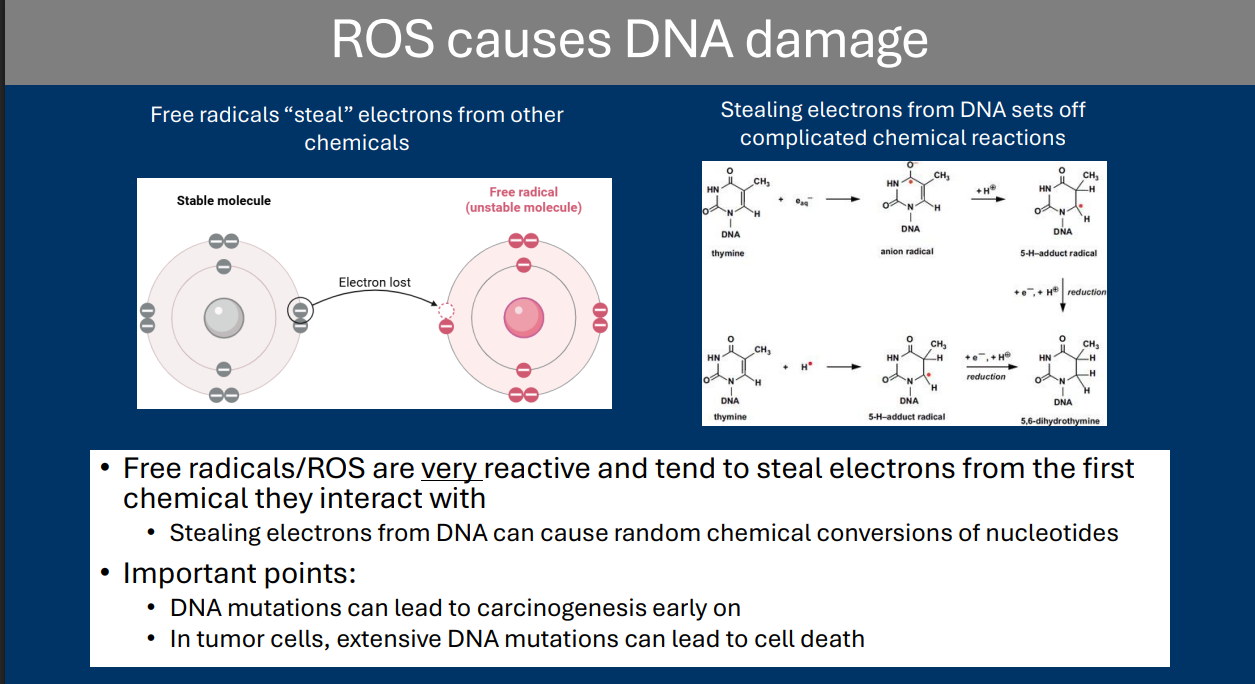
The dangers of oxygen metabolism
Oxygen: With great power comes great responsibility
• During aerobic respiration, oxygen serves as the final electron acceptor of the ETC
- Due to the high electronegativity of oxygen (loves to “steal” electrons)
• Oxygen’s high electronegativity makes oxygen very reactive
- Often “steals” electrons from other chemicals
• Reactive oxygen species (ROS) refers to a class of oxygen-based chemicals that can cause severe damage to cells
> DNA damage is a major target of ROS
The importance of lactate production
The Warburg Effect: The importance of lactate
During anaerobic respiration pyruvate is converted into lactate by the enzyme lactate dehydrogenase
1) Regeneration of NAD+
• Continual glycolytic flux requires a constant supply of NAD+ During anaerobic respiration pyruvate is converted into lactate by the enzyme lactate dehydrogenase
2) Acidification of the extracellular space
• Lactate export also exports H+ ions
• Acidification aids metastasis by facilitating ECM breakdown
3) Lactate induces angiogenesis
• Similar to VEGF, extracellular lactate can signal to endothelial cells to undergo angiogenesis
Gluconeogenesis
• Gluconeogenesis can generate glucose as a precursor for synthesizing other carbohydrates
Pentose Phosphate Pathway
• The pentose phosphate pathway (PPP) generates nucleotides for DNA synthesis
Lipogenesis
• Lipogenesis generates phospholipids from Glyceraldehyde-3-phosphoate (glycolysis) and Acetyl-CoA
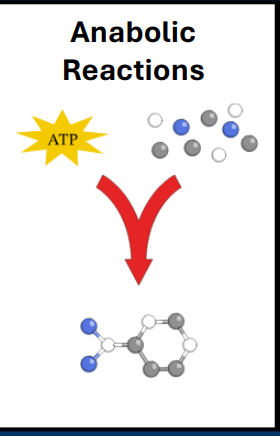
Anabolism
• Anabolic reactions
• Builds complex molecules using smaller molecules
• Requires energetic input (ATP)
- Continuous cell division of cancer cells requires a substantial amount of anabolism
- Anabolism requires a substantial amount of energy (e.g. catabolism)
Catabolism
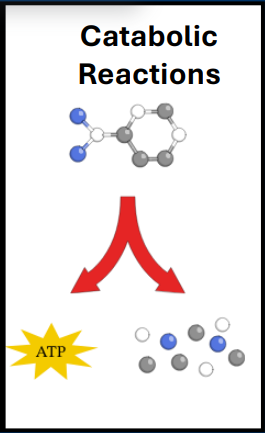
• Catabolic reactions
• Breaks down complex molecules into small molecules
• Releases energy that can be stored at ATP
-Anabolism requires a substantial amount of energy (e.g. catabolism)
Reactive Oxygen Species
Reactive oxygen species (ROS) refers to a class of oxygen-based chemicals that can cause severe damage to cells
• DNA damage is a major target of ROS
Nrf2
The Nrf2 pathways is a common pathway that regulates ROS scavenging
Pathway overview:
• Nrf2: Transcription factor, with expression regulated by ROS
• Cul3: Ubiquitin ligase that ubiquitinates Nrf2
• Keap1: Adaptor protein that bridges an interaction between Cul3 and Nrf2
The Nrf2 Signaling Pathway
• Under “normal” conditions:
• Nrf2 interacts with Keap1 which acts as a bridge between Nrf2 and Cul3
• Cul3 is a ubiquitin ligase that polyubiquitinates Nrf2 leading to its degradation
• End result: In the absence of ROS, Nrf2 expression remains low
• Under conditions of high ROS:
• ROS molecules modify the structure of Keap1
• Keap1 releases Nrf2
• Nrf2 is no longer poly-ubiquitinated/degraded by Cul3
• Nrf2 translocates into the nucleus and transcribes genes needed for the removal of ROS
> Example genes = Catalase, SOD1, SOD2, Glutathione
End result: In the presence of ROS, Nrf2 expression increases and protective genes are expressed
Keap1
The Nrf2 Signaling Pathway
• Keap1: Adaptor protein that bridges an interaction between Cul3 and Nrf2
• Under “normal” conditions:
• Nrf2 interacts with Keap1 which acts as a bridge between Nrf2 and Cul3
• Under conditions of high ROS:
• ROS molecules modify the structure of Keap1
• Keap1 releases Nrf2
Cul3
The Nrf2 Signaling Pathway
• Cul3: Ubiquitin ligase that ubiquitinates Nrf2
• Under “normal” conditions:
• Nrf2 interacts with Keap1 which acts as a bridge between Nrf2 and Cul3
• Cul3 is a ubiquitin ligase that polyubiquitinates Nrf2 leading to its degradation
• Under conditions of high ROS:
• Nrf2 is no longer poly-ubiquitinated/degraded by Cul3
GLUT1
• HIF-1α promotes the expression of:
• GLUT1: A membrane transport protein for glucose
mTOR
• The mTOR signaling pathways is a master regulator of anabolic metabolism
• Revolves around activation of mTOR, a protein kinase with many regulatory targets in the cell
• mTOR activation increases nearly all anabolic pathways
• Pathway activity is upregulated in >50% of cancers
• mTOR: Protein kinase that promotes anabolic metabolism when activated
• Rheb is an activator of mTOR (Inactive GDP-Rheb can no longer activate mTOR)
• mTOR signaling is repressed by AMPK
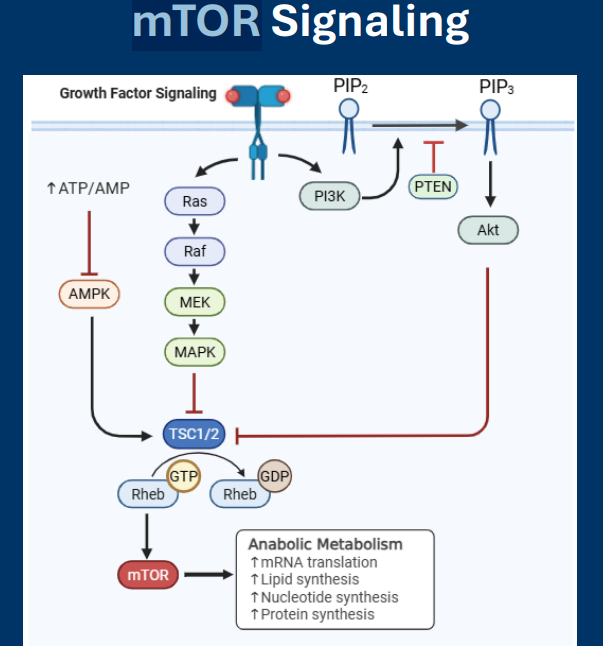
TSC1/2
Regulation of metabolic pathways: mTOR
• TSC1/2: GTPase-activating protein that induces Rheb to convert GTP (active) to GDP (inactive).
RheB
Regulation of metabolic pathways: mTOR
• Rheb: G-protein that activates mTOR
• TSC1/2: GTPase-activating protein that induces Rheb to convert GTP (active) to GDP (inactive).
• Rheb is an activator of mTOR
Akt
• Activation PI3K pathway via activation of AKT
AMPK
Known pathways that inactive mTOR:
• AMPK
- mTOR signaling is repressed by AMPK
• AMPK is an “energy sensor” kinase:
- Activated by high AMP/ATP ratio
- Inactivated by high ATP/AMP ratio
• Low energy availability (i.e. high AMP/ATP) activates AMPK
• AMPK activates TSC1/2
Critical thinking questions
1.) How would the regulation of the enzymatic activities of Pyruvate Dehydrogenase and Lactate Dehydrogenase influence a cell’s tendency to utilize oxygen for metabolism?
2.) What is the difference between anabolic and catabolic pathways? Why are each important for carcinogenesis?
3.) What is the Warburg effect? What are some possible reasons why cancer cell metabolism follows this phenomenon?
1.)
2.)
3.)
Critical thinking questions
4.) In what ways does HIF-1α signaling help activate the metabolic pathways responsible for the Warburg effect?
5.) What is the function of Nrf2? How can we explain the observation that Nrf2 can function as either a tumor suppressor or a proto-oncogene depending on context?
6.) What is mTOR signaling and how does it affect cellular metabolism? What is the rationale for the suppression of mTOR signaling by the activation of AMPK?
7.) What are some ways in which our diet can either increase or decrease our chances of developing cancer?
4.)
5.)
6.)
7.)