SCC MODULE 4
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
explain kinetic theory of matter
attempts to explain the difference between solids, liquids and gases
explain solids:
particles are closely packed and held together very strongly. have fixed shape and a fixed volume.
explain liquids:
particles are held together fairly strongly, but can slip past each other and change position. have no fixed shape, but fixed volume.
explain gases:
particles are far apart, attractive forces between gas particles are negligible. have no fixed shape or volume.
what are the many similar physical properties gases share?
consist of tiny particles in constant motion
particles move in rapid, random, straight-line motion; results in collusions with one another and container walls.
pressure of a gas is due to collision of particles with container walls
collisions are elastic - no energy loss
volume of particles themselves is very small as compared to the volume occupied by the gas
no attractive or repulsive forces between particles
average kinetic energy is proportional to absolute (kelvin) temperature
what are gas measurements:
temperature (T):
celsius (C)
kelvins (K)
absolute zero = OK = -273.15C
100C = 373.15K
pressure (P):
pascals (Pa)
mm Hg
atmospheres (atm)
what is 1 atm equal to in kPa and mm Hg:
101.3kPa and 760 mm Hg
what are Volume measurements:
1 cubic metre = 1000L
unit for volume = L
what is the measurement for standard temperature and pressure =
0 C (273.15K) and 101.3kPa
describe Boyles law:
as pressure of a gas increases its volume decreases and vice versa. only true at constant temperature
equation that explains boyles law:
P1V1=P2V2
describe Charles law:
as volume of a gas increases , the temperature is increased. true at constant pressure
equation that explains Charles law:
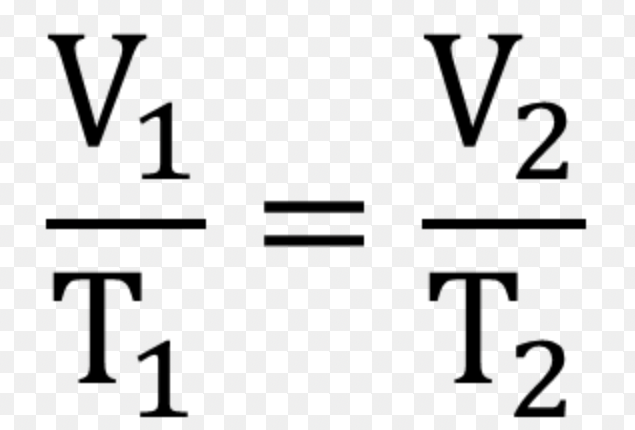
what is absolute zero
the lowest possible temperature where nothing could be colder and no heat energy remains
point where the fundamental particles have minimal vibrational motion, retaining only quantum mechanical, zero point energy induced particle motion
how is absolute zero defined as:
0K = -273.15 C
Combined Gas Law:
described the relationship between the pressure, volume and temperature for a fixed amount of gas
equation for combined gas law:
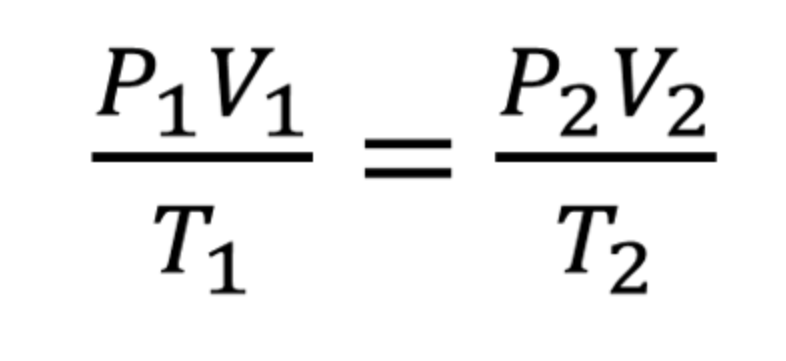
describe avogadros hypothesis:
equal volumes of all gases, measured under the same conditions of temperature, pressure and volume contain the same number of molecules, regardless of the type of gas.
how is avogadros hypothesis shown by
one mole of gas (6.022 ×10²³) has a volume of 22.4L at STP (101.3kPa and 273.15K)
formula to find moles with volume
Volume/22.4
explain ideal gas law & the equation
express the relationship between moles, volume, temperature and pressure using ideal gas law equation:
PV = nRT
what is ‘R’ ?
ideal gas constant:
if pressure is in kPa= 8.31 L kPa mol-1 K-1
if pressure is in atm = 0.0821 atm mol-1 K-1
if pressure is in mm Hg= 62.4 mm Hg mol-1 K-1
explain daltons law of partial pressures: & the equation
total pressure in a mixture of non reacting gases = sum of partial pressures of individual gas in the mixture.
P = p1 + p2 + p3
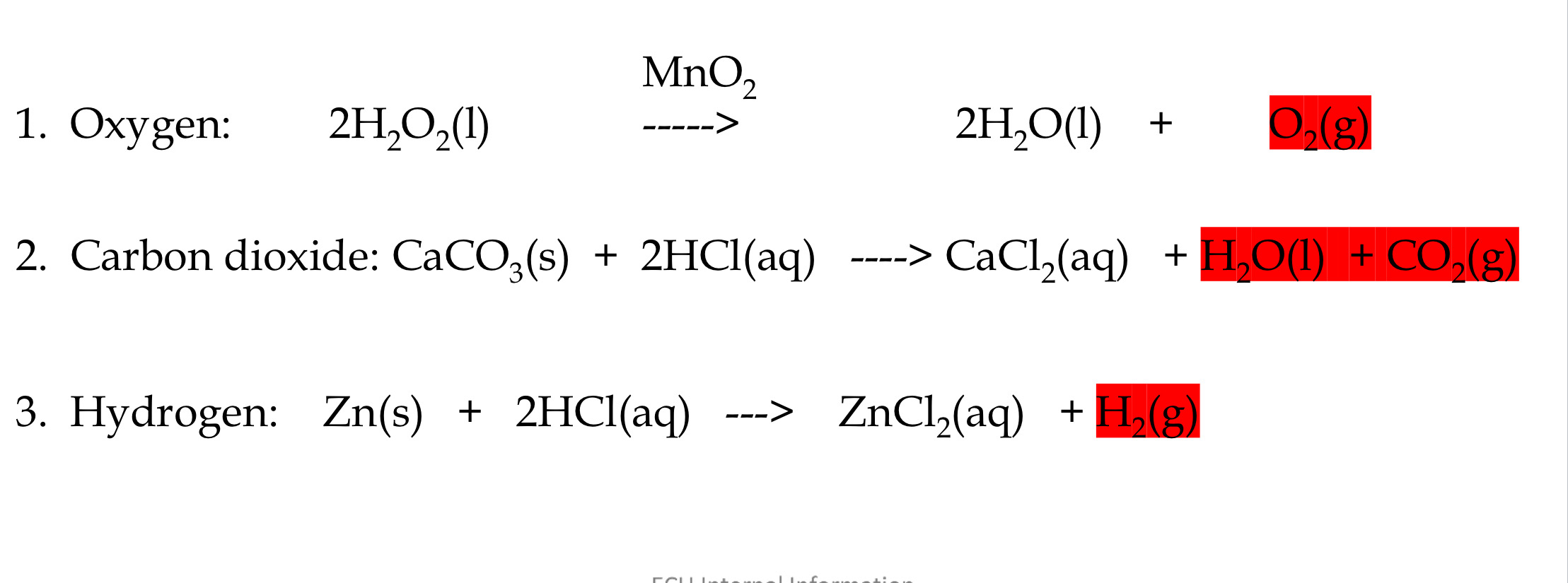
what is the laboratory preparation of the important gases; hydrogen, oxygen and carbon dioxide:
what is the composition of unpolluted dry air:
nitrogen = N2 = 78.1%
oxygen = O2 = 20.9%
argon = Ar = 0.93%
carbon dioxide = CO2 = 0.035%
others = <0.01%
what are 4 atmospheric issues
ozone depletion
photochemical smog
acid rain
greenhouse effect
explain ozone and its production & equation:
colourless, highly reactive gas with a distinct odour
stratosphere contains ozone gas which is formed from dissociation of molecular oxygen by UV radiation;
O2 —> O. + O. and then O. + O2 —> O3
what does decrease of ozone concentration lead to:
attributed to chloroflurocarbon of CFC’s, found in aerosols and refrigerators
why are CFC’s bad:
chemicals are very stable and remain in the environment for many years and eventually enter the stratosphere where UV radiation breaks up the CFC’s producing chlorine atoms that break up the ozone molecule
equation for chlorine breaking up ozone molecule:
Cl. + O3 —> ClO + O2 and ClO + O. —> Cl. + O2
what are processes of phasing out CFC’s:
using alt technologies
replacement chemicals:
hydrocarbons
HCFC’s
HFC’s
define smog
term used for collection of pollutants; ozone, particulate matter and humidity mixed together.
what is photochemical smog:
when the ozone layer is in the wrong place (due to high levels of ozone produced due to reactions of pollutants), in contrast with stratospheric ozone depletion.
requirements for photochemical smog to occur:
hydrocarbons
nitrogen oxides
sunlight
results of photochemical smog:
brown haze, ozone, oxidised organic compounds
what is acid rain
refers to precipitation that is slightly more acidic than usual
how is acidic rain produced
burning of sulfur containing fossil fuels results in the production of sulfur oxides, then reacting with water in air to produce acidic molecules (sulphuric acid)
what are the 2 predominant acids in acid rain and why
sulphuric acid and nitric acid due to the primary pollutants: sulfur oxides and nitrogen oxides
what are properties of nitric and sulphuric acid:
very soluble in water and strong acids
impact of acid rain on earth:
damages forests, aquatic ecosystems, buildings and statues.
what are greenhouse gases (gg):
in the troposhpere are essential as they trap radiant heat and help keep the temperature of the lower atmosphere warm and relatively constant
why is the increase of gg a problem?
increased CO2 has prevented infra red radiation from escaping since CO2 molecules absorb IR, this causes an increased warming of earth
how are the concentration of gg increased
due to combustion of fossil fuels, coal, oil and natural gas, also when forests are cleared.