AP BIO UNIT 1: Chemistry of life
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What are atoms made up of
Protons, neutrons, electrons
What is a proton
Subatomic particle with a positive charge— found in the nucleus of the atom
What is a neutron
Subatomic particle with neutral/no charge— found in the nucleus of the atom.
What are electrons
Subatomic particles with a negative charge— found in orbitals around the nucleus.
What are valence electrons?
Electrons located in the outermost shell of an atom, responsible for bonding and chemical reactions. They determine an atoms reactivity and stability.
What will atoms do in order to complete their valence shell?
Atoms will gain, lose, share, or take electrons to complete their valence shell.
What is electronegativity
An atom’s tendency to attract electrons to itself in a chemical bond, influencing polarity and bond strength.
What are bonds between elements?
Interactions between different elements and their electrons that result in the formation of molecules.
What is an ionic bond
When one element wants to get rid of its electrons, and the other element wants to gain electrons, resulting in a transfer of electrons and the formation of charged ions. This is due to the attraction of oppositely charged ions
What are covalent bonds?
Covalent bonds are formed when two atoms share one or more pairs of electrons, allowing them to achieve a full outer electron shell and become more stable.
What determines if a covalent bond is nonpolar?
If the elements have the same electronegativity(both equally as attracted to the electron), sharing electrons equally.
What determines if a covalent bond is polar?
If the atoms involved have significantly different electronegativities—resulting in an unequal sharing of electrons, leading to the formation of a partial positive and partial negative charge.
What are functional groups?
Specific groups of atoms within molecules that determine the characteristic chemical reactions of those molecules.
What is the hydroxyl functional group
Oxygen bonded to a hydrogen
—OH or —HO
Polar, because of oxygen's electronegativity
Make any region where hydroxyl is attached hydrophilic and increases water solubility
Found in alcohols, sugars
What is the Carbonyl functional group
carbon double bonded to oxegyn
C=O
Polar & hydrophilic because of oxegyn’s electronegativity
found in sugars
highly reactive, helps form ring structures
What is the carboxyl functional group?
double bonded carbon group attached to a hydroxyl
—COOH
Polar and acidic; donates protons (when dissolved in water, H+ ion is breaks off)
leaves ionized carboxyl
Found in amino acids and fatty acids
What is the amino functional group?
Nitrogen attached to two hydrogens
—NH2
Polar
Makes the molecule it is attached to a base by accepting protons and increasing PH
If this happens, it will have THREE hydrogens and an ionized charge.
Found in amino acids and nucleotides
what is the sulfhydryl functional group?
Sulfur atom bonded to a hydrogen
—SH
Polar
Important in determining the shape of proteins
Stabilizes protein structure through disulfide bonds
Forms disulfide bonds when they come close to one another that bend/turn into a protein chain that determine the shape
Found in amino acids and proteins
What is the phosphate functional group?
One phosphorus bonded to 4 surrounding oxegyns
Contains one double bonded oxygen and three single bonded oxygens
—PO₄²⁻
Ionized and Non ionized
Polar and negatively charged
Found in nucleotides and phospholipids
Make molecules acidic
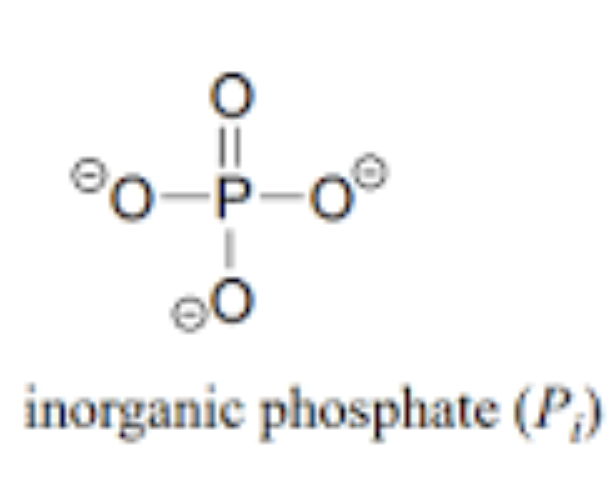
What is an ionized phosphate group
Also known as inorganic phosphate
No hydrogen ions
Found in cells
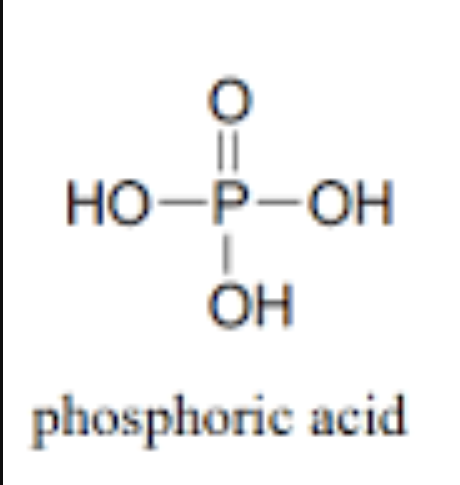
What is a non-ionized phosphate group
Also known as organic
Oxygen atoms are bonded to hydrogens
Neutral charge
What is a methyl functional group?
A carbon atom bonded to three hydrogen atoms
—CH₃.
Adding methane groups is called methylation
Creates a nonpolar region in whatever molecule it is attached to
Turns gene expression OFF
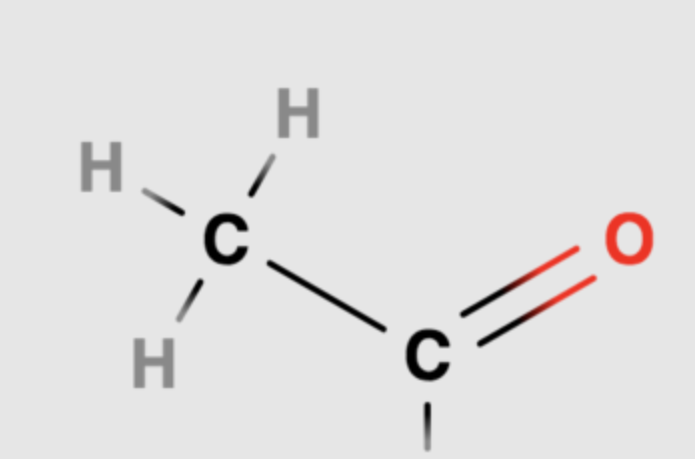
What is an acetyl functional group?
A carbon bonded to three hydrogens, attached to another carbon double bonded to an oxygen
Methyl group attached to a carbonyl
Adding acetyl groups is called acetylation
Turns gene expression ON
Polar because of the carbonyl group
What kinds of bonds do water have?
INSIDE the water molecule:
Polar covalent bonds between hydrogen and oxygen atoms
Polarity allows for hydrogen bonding between water molecules.
BETWEEN the water molecules
hydrogen bonds between the positive hydrogen end of one water molecule to the negative oxygen end of another.
What is a hydrogen bond?
An attractive bond between the positive side of one hydrogen atom, and the negative side of another electronegative atom, typically oxygen or nitrogen.
W