Tannins and Simple Phenolics Lecture Flashcards
1/100
Earn XP
Description and Tags
A set of 100 flashcards covering the definition, classification, properties, and sources of tannins and simple phenolics based on the PHARM 272 lecture.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
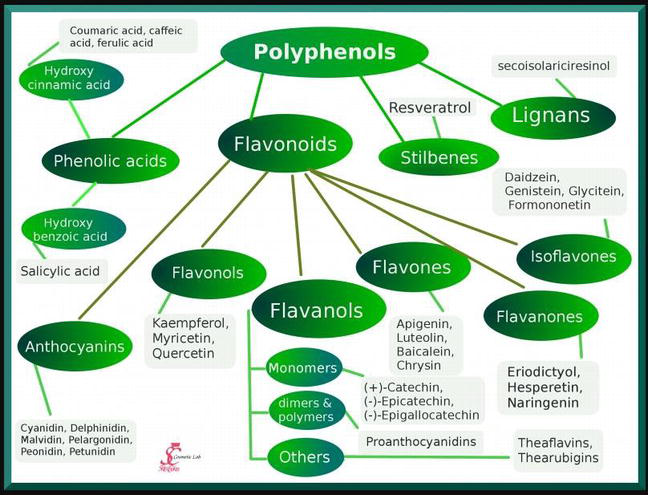
Plant phenols
Aromatic compounds characterized by an aromatic ring bearing a hydroxyl group.
Shikimic acid pathway
One of the biosynthetic pathways for plant phenols, also known as the Phenylpropanoid pathway.
Amino acid precursors
Aromatic amino acids such as phenylalanine, tyrosine, and tryptophan from which plant phenols are derived.
Examples of phenolic compounds
Simple phenols, flavonoids, quinones, lignins, and tannins.
Tannin (Historical Origin)
Term first used by Seguin to denote substances that react with animal hide protein to prevent decomposition and create leather.
Definition of Tannins
High molecular weight poly-hydroxy phenolic compounds capable of forming strong complexes with proteins and other macromolecules.
Nitrogen status of Tannins
They are non-nitrogenous compounds.
Chemical precursors (Definition 2)
Condensation products of Gallic acid or Flavan-3-ol, as well as their glycosides.
Plant distribution
Widely found in angiosperms and gymnosperms, specifically in the leaf, seed, root, bud, wood, and stem.
Tissue locations
Often found in secondary phloem, xylem, and between the cortex and epidermis.
Physiological role in growth
Tannins help regulate the growth of plant tissues.
Enzymatic inhibitory effect
Tannins inhibit many enzymes due to protein precipitation, providing a protective function in barks and heartwoods.
Hydrogen transfer role
Tannins play a role in plant cell hydrogen transfer due to their high affinity for oxygen.
Defensive roles
Act as deterring agents against predators and a barrier to microorganisms.
Astringent property
The ability to react with mucous membrane proteins to cause precipitation.
Physical appearance
Non-crystalline, generally powdery or flaky.
Solubility
Soluble in water, alcohol, dilute alkalis, glycerols, and acetone; sparingly soluble in other organic solvents.
General coloration
Generally dark yellow to reddish brown in color.
Precipitation with heavy metals
Tannins form precipitates with heavy metals such as Lead.
Precipitation with Nitrogenous salts
Tannins precipitate alkaloidal salts.
Precipitation with Proteins
Tannins precipitate proteins such as gelatin.
Iron salts reaction
Forms a colored reaction when treated with FeCl3.
Ammonia/Potassium ferricyanide reaction
Produces a deep red color.
Dietary sources of astringency
Tea, berries, and wine; causes a dry and puckery aftertaste.
Fruit sources
Strawberries and blueberries.
Nut sources
Almonds and walnuts.
Herb and spice sources
Clovie, vanilla, cinnamon, and cumin.
Styptics
A medicinal use of tannins for stopping bleeding.
Internal application
Used for the protection of inflamed surfaces of the mouth and throat.
Anti-diarrhoeal
A medicinal property of tannins used to treat diarrhea.
Antidote use
Employed as antidotes for poisoning by heavy metals, alkaloids, and glycosides.
Skin condition treatments
Used for bleeding abrasions, skin ulcers, and as a cicatrizant on gangrenous wounds.
Schistosomiasis
Tannins have molluscidal activity used to interrupt the transmission cycle of this disease.
Anti-viral effects
Reported medicinal property of tannins against viruses.
Treatment for stings/plants
Used to treat poisons from poison oak or bee stings for instant relief.
Manufacture uses
Used in the production of dyes, ink, wine processing, and leather.
True Tannins
High molecular weight poly-phenolic compounds (1000 to 5000Da) that convert animal hide to leather.
Goldbeater’s skin test
A qualitative test used to detect true tannins.
Hide powder test
A quantitative test for tannins based on their adsorption on standard hide powder.
1% Gelatin solution
A solution used to precipitate true tannins.
Pseudotannins
Low molecular weight monomeric polyphenols that do not respond to the Goldbeater’s skin test.
Pseudotannin examples
Eugenol, gallic acid, catechins, chlorogenic acid, phloroglucinol, and salicylic acid.
Gallic acid sources
Rhubarb and materials containing gallitannins.
Catechin sources
Catechu, acacia cutch, Australian kinos, cocoa, and guarana.
Chlorogenic acid sources
Mate, unroasted coffee, and a small quantity in Nux vomica.
Ipecacuanhic acid
A pseudotannin found in Ipecacuanha.
Hydrolysable Tannins
Tannins that can be broken down by enzymes (e.g., tannase) or acids.
Hydrolysable Tannin precursors
Phenolic acid (gallic or ellagic acid) and a glucose residue.
Structure of Hydrolysable Tannins
Phenolic acids united by ester linkages to a central sugar molecule, usually D-glucose.
Gallotannins
Hydrolysable tannins composed of gallic acid units.
Ellagitannins
Hydrolysable tannins composed of hexahydroxydiphenic acid units.
Depside bond
An ester bond linking two or more monocyclic aromatic units in polyphenolic compounds like gallotannins.
Ellagitannin linkage
Galloyl groups are linked through C-C bonds.
Tannic acid
An example of a hydrolysable tannin structure involving gallic acid units.
Gallitannin Hydrolysis
Produces Gallic acid upon acid hydrolysis.
Ellagitannin Hydrolysis
Yields ellagic acid upon acid hydrolysis.
Lactonization
The process by which hexahydroxydiphenic acid becomes ellagic acid during chemical hydrolysis.
Gallitannin Solubility
Rapidly soluble in water.
Ellagitannin Solubility
Slowly soluble in water.
Gallic acid + Iron salts
Produces a blue fluorescence.
Ellagic acid + Iron salts
Produces a blue fluorescence.
Hydrolysable tannin dry distillation
Produces pyrogallols and other phenolic compounds.
Gallitannin Plant Sources
Rhubarb, cloves, red rose petals, bearberry leaves, Chinese galls, Turkish galls, hamamelis, chestnut, and maple.
Ellagitannin Plant Sources
Pomegranate rind and bark, myrobalans, eucalyptus leaves, kousso, and oak bark.
Corilagin
A specific example of a hydrolysable tannin.
Geraniin
A specific example of a hydrolysable tannin.
Condensed Tannins
Derivatives of flavonols (catechin and flavonol-3-4-diol) that are not hydrolysable.
Condensed Tannin Synonyms
Non-hydrolysable tannins, pro-anthocyanidins, or catechol type tannins.
Condensed Tannin Biosynthesis
Formed by the condensation of flavanol units via C-C bonds to form polymers.
Sugar content in Condensed Tannins
They do not contain sugar residues.
Condensed Tannin reaction with Iron salts
Solutions turn green.
Phlobaphenes
Red insoluble compounds formed when condensed tannins are treated with acids or enzymes.
Condensed Tannin dry distillation
Produces catechol.
Proanthocyanidins (PAs)
Term for condensed tannins derived from their acid-catalyzed oxidation into red anthocyanidins.
Anthocyanidin pigment
Pigment responsible for colors in flowers, leaves, fruits, juices, and wines.
Condensed Tannin Bark sources
Cinnamon, wild cherry, cinchona, willow, acacia, and hamamelis.
Condensed Tannin Root sources
Krameria (rhatany) and male fern.
Condensed Tannin Seed sources
Cocoa, kola, and areca.
Condensed Tannin Fruit sources
Cranberries and grapes (red wines).
Condensed Tannin Leaf sources
Hamamelis and tea.
Condensed Tannin Extract/Juice sources
Catechu, mangrove, and eucalyptus.
Propelargonidin
A type of proanthocyanidin where R1=H and R2=H.
Procyanidin
A type of proanthocyanidin where R1=OH and R2=H.
Prodelphinidin
A type of proanthocyanidin where R1=OH and R2=OH.
Complex Tannins
Tannins biosynthesized from both a hydrolysable tannin (mostly C-glucoside ellagitannin) and a condensed tannin.
Anogeissusin A
An example of a complex tannin (R=H).
Anogeissusin B
An example of a complex tannin (R=OH).
Flavano-ellagitannin
A structural description for complex tannins like anogeissusin.
Gelatin test
A chemical test used to identify tannins by precipitation.
Catechin test
A specific chemical test for identify catechins.
Chlorogenic acid test
A chemical test used to identify chlorogenic acid.
Lead acetate test
A chemical test used to precipitate tannins using lead salts.
Precipitation of alkaloidal salts
A test used to detect tannins through the formation of precipitates with alkaloids.
Monomeric components
Low molecular weight units that make up pseudotannins.
Shikimic acid
The starting point of the pathway for tannin biosynthesis.
Poly-hydroxy phenolic
The chemical classification of tannins based on their multiple hydroxyl groups on aromatic rings.
Tannase
An enzyme capable of hydrolyzing hydrolysable tannins.
Hexahydroxydiphenic acid
The specific phenolic acid unit found in ellagitannins.
Catechol type tannins
Another name for condensed tannins based on their dry distillation product.
C-glucoside ellagitannin
A common hydrolysable component found within complex tannins.