AQA A level Chemistry 3.1.3 Bonding
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What is a covalent bond? (1)
A shared pair of electrons
What is meant by the term "electronegativity"? (2)
The power of an atom to attract a pair of electrons in a covalent bond
What is electronegativity measured on and what is the scale of it? (2)
- Pauling scale
- 0 to 4.0
Describe the general trend in electronegativites across a period (4)
- Increases
- Number of protons increases
- Same shielding
- Atomic radius decreases
Describe the general trend in electronegativites down a group (4)
- Decreases
- No. of principle energy levels increases
- Shielding increases
- Atomic radius increases
What is the most electronegative atom? (1)
F - Fluorine (compare all other elements to this if ever asked to rank the electronegativites of different elements)
What does a polar bond mean? (1)
A covalent bond where the electron density is unevenly distributed
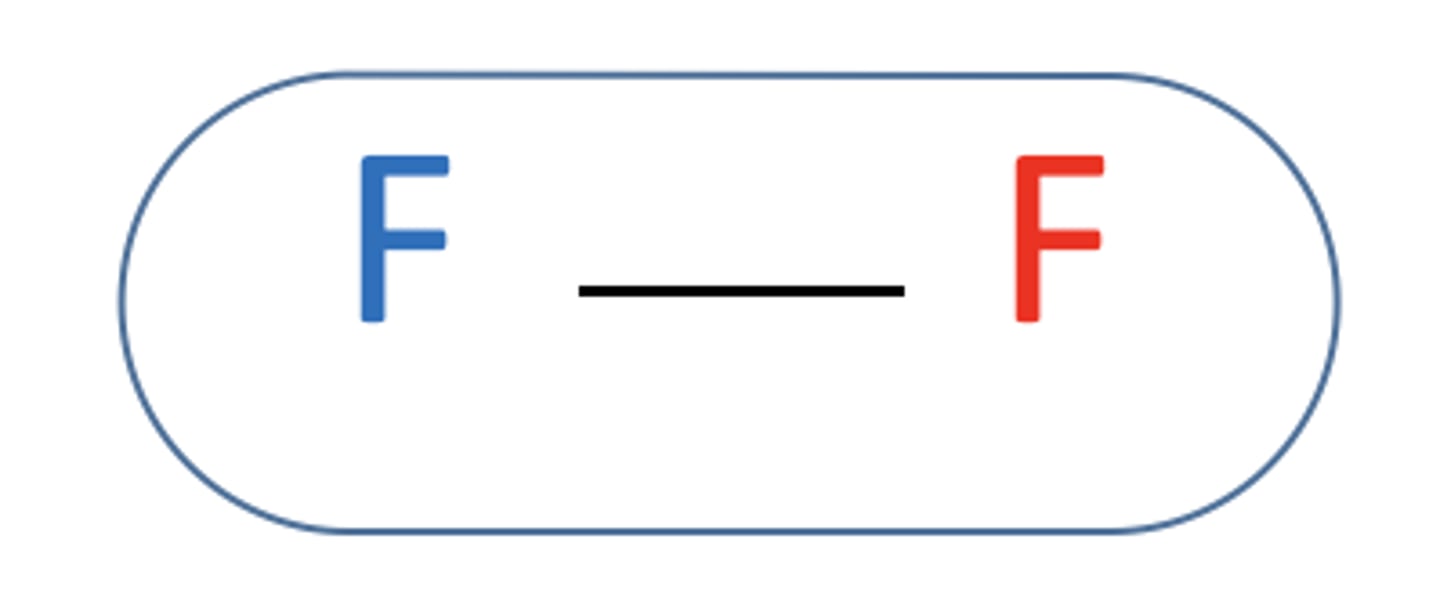
Are planar molecules such as F2 polar?
- No
- As the electron cloud is evenly distributed
Draw the polar bond and electron cloud formed by HF, showing clearly the partial charges (4)
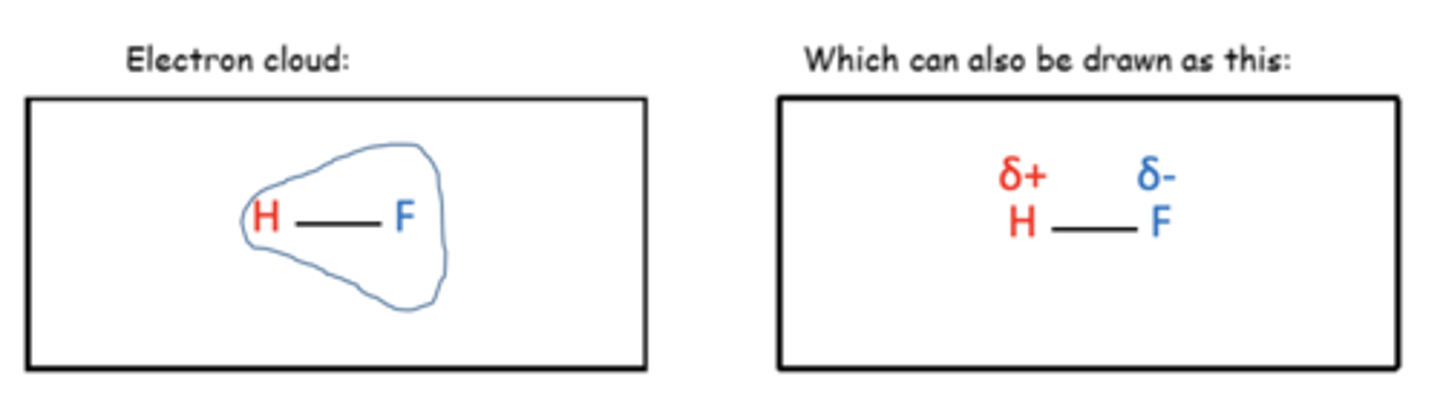
What partial charge does the more electronegative atom have in polar bonds? (1)
δ-
What partial charge does the less electronegative atom have in polar bonds? (1)
δ+
What happens when atom atoms with a very large difference in electronegativity form a polar bond? (2)
- Has such a strong pull of electrons that instead of sharing it, it will remove them completely
- Ionic bonding
What is an ionic bond? (2)
- A strong electrostatic attraction
- Between oppositely charged ions
What type of lattice structure do ionic compounds like NaF form? (1)
Giant ionic lattice
What are the properties of ionic compounds? (3)
- High melting and boiling points - due to strong electrostatic attraction
- They will not conduct electricity in solid states - As ions are not free to move or flow
- They are brittle and will shatter upon impact
What factors affect the strength of an ionic bond?
- Charge of ion (greater = better)
- Size of ion (smaller = better)
What is a metal? (2)
- A lattice of positively charged ions
- Attracted to a sea of delocalised electrons
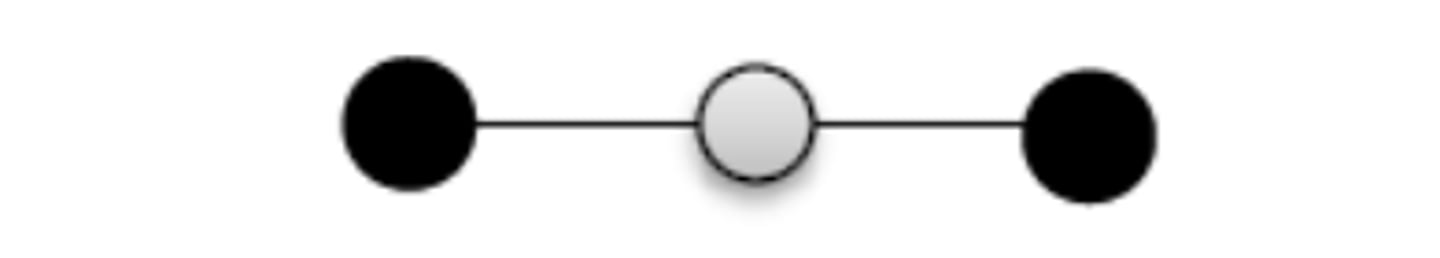
Draw a basic structure of a metal (3)
Ensure that the positive charge on the diagram metal ion reflects the charge of the metal ion e.g. Mg2+ would have 2+ ions, Al3+ would have 3+ ions drawn
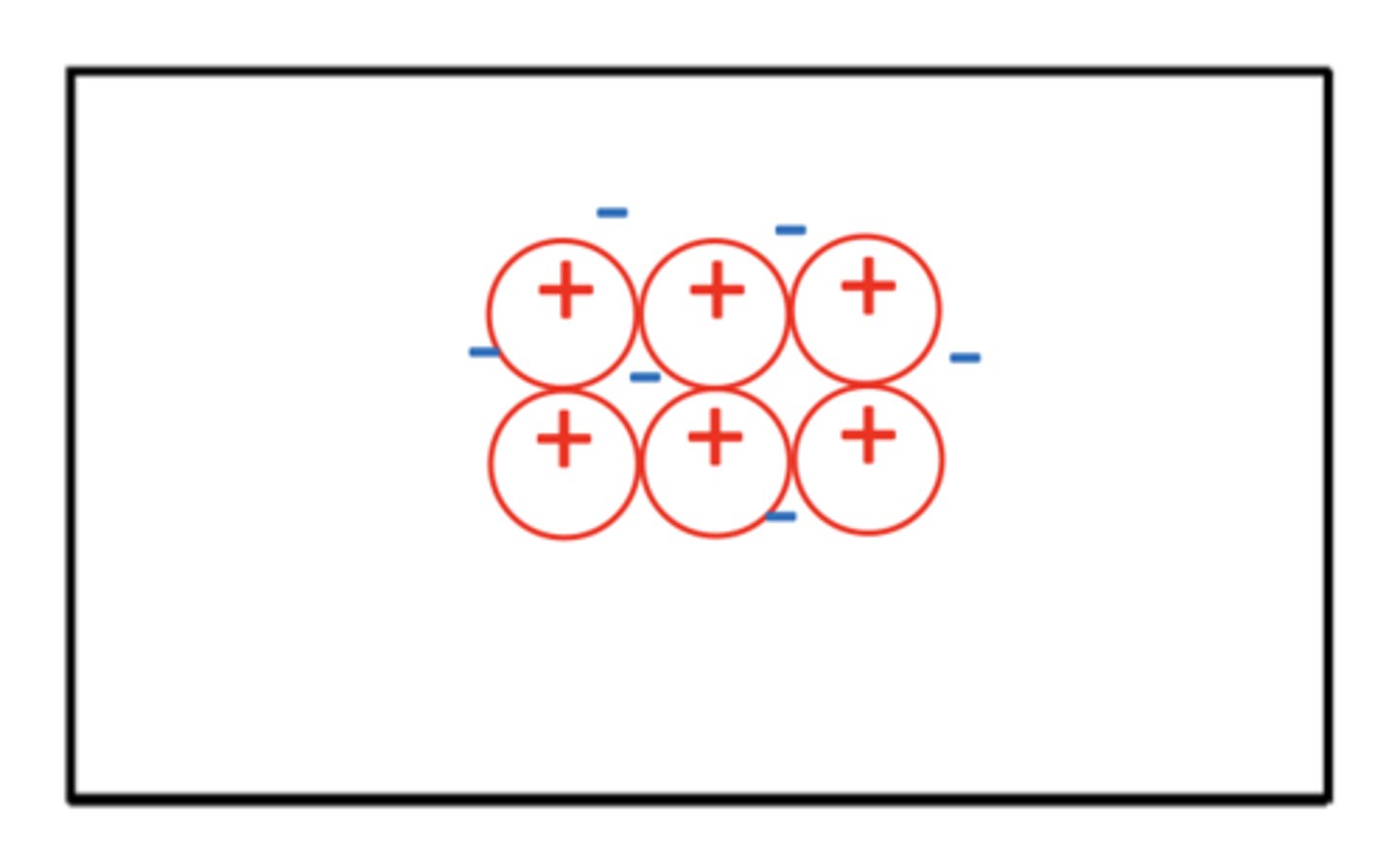
What are the three properties of metals? (3)
- Good conductors of electricity and heat
- High melting points
- Malleable and ductile
What factors affect the strength of a metallic bond? (3)
- Ionic charge (Higher = stronger)
- Number of delocalised electrons (Greater = stronger)
- The atomic radius (Smaller = stronger)
Why are metals good conductors of heat and electricity? (1)
The delocalised electrons are free to move and flow
Why are metals malleable and ductile ? (1)
There are layers of ions that cab slide over each other
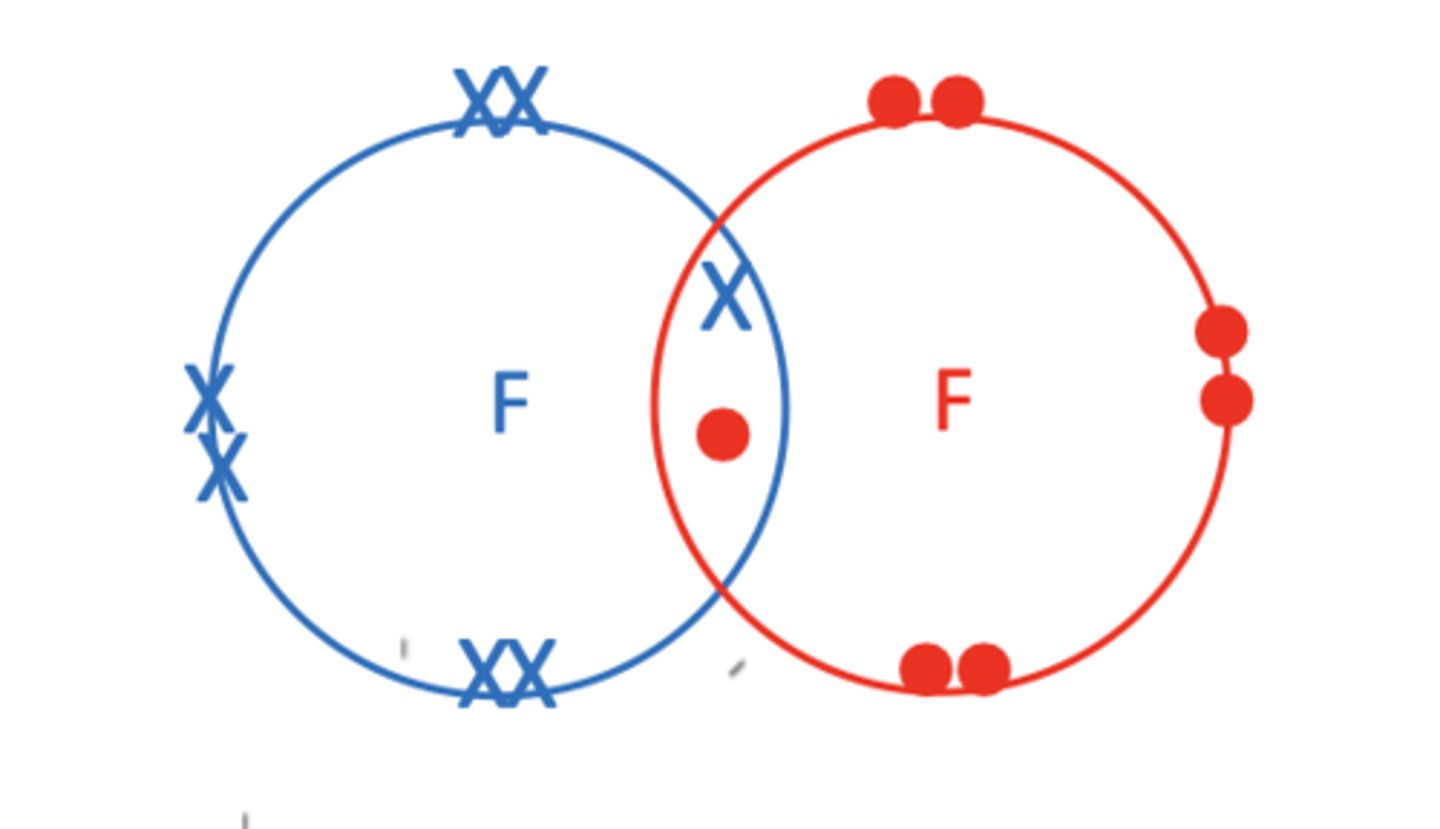
Draw a dot and cross diagram of F2 (3)
Draw a dot and cross diagram of O2 (3)
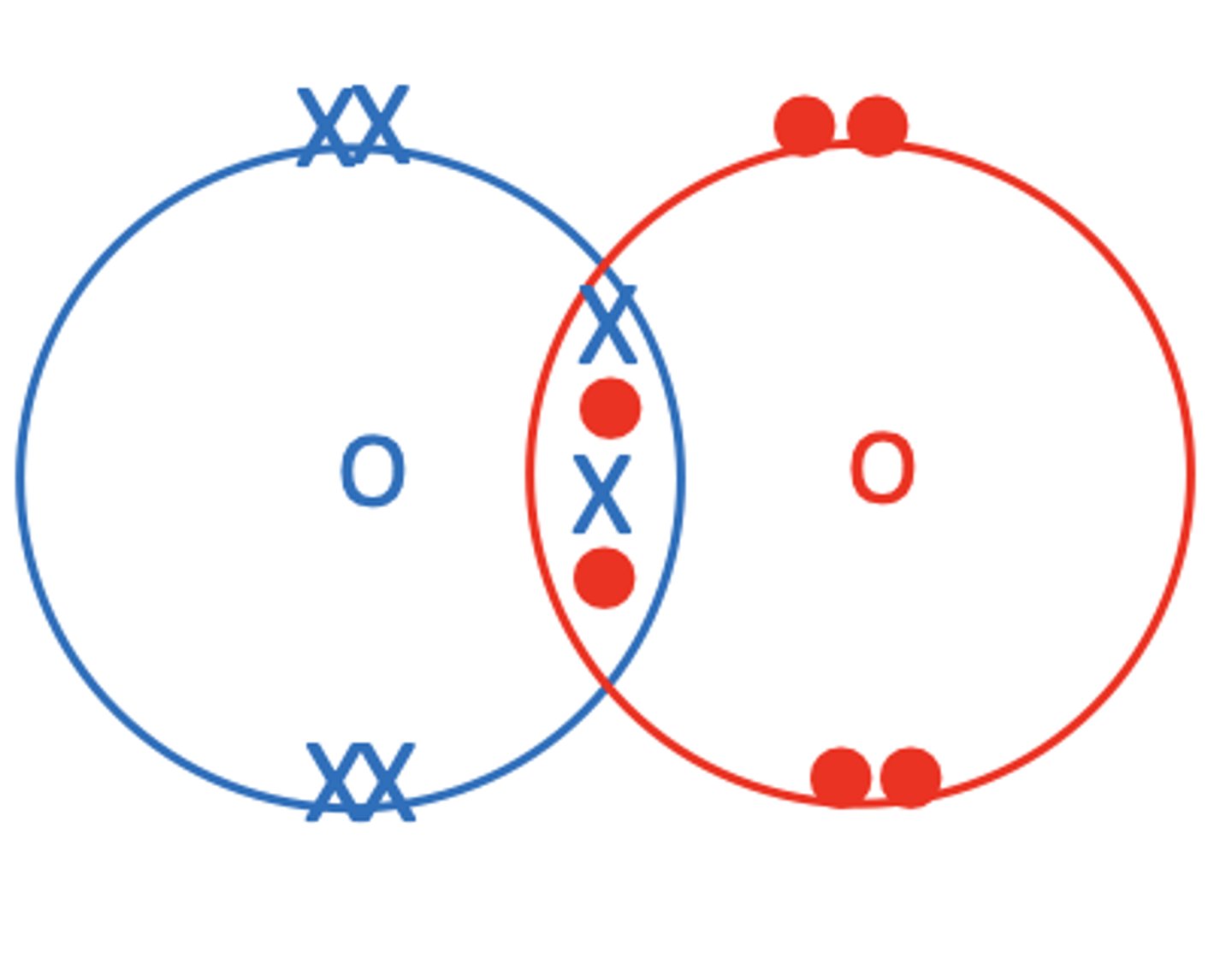
What is a co-ordinate bond? (2)
- When one atom provides both the electrons that are required
- To form a covalent bond
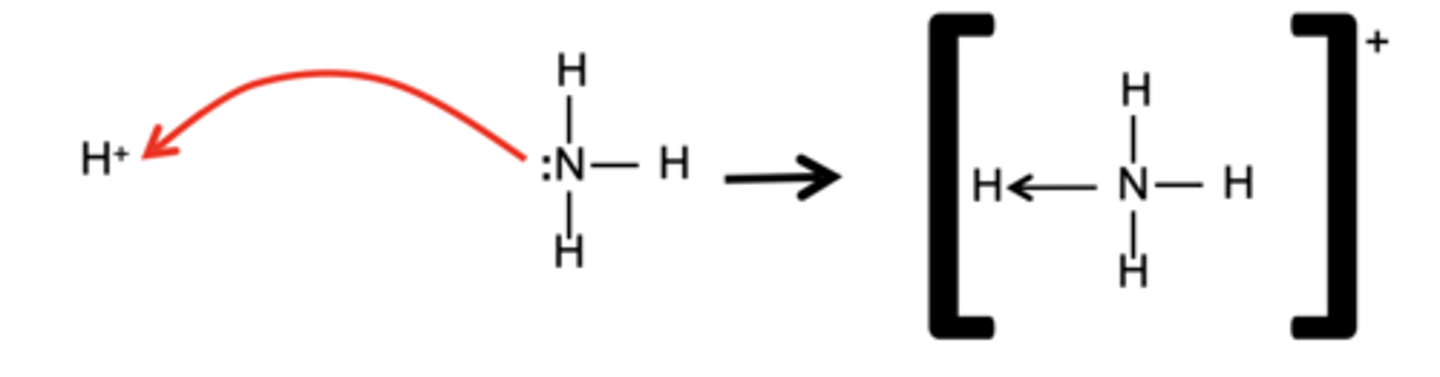
Draw and describe the formation of a an ammonium ion (3)
The nitrogen donates both electrons for the covalent bond
2BP 0LP
- Linear
- 180
3BP 0LP
- Trigonal planar
- 120
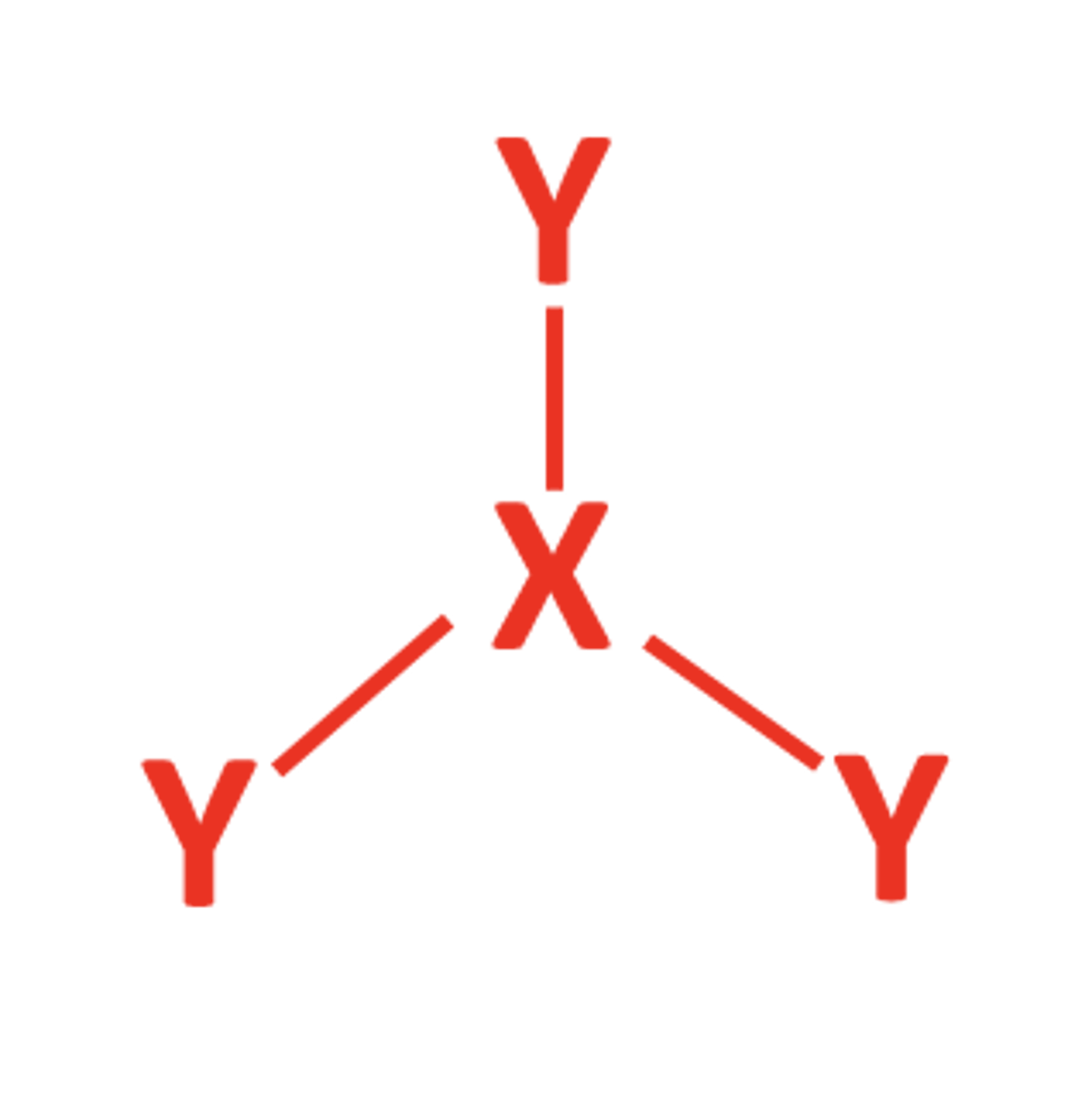
4BP 0LP
- Tetrahedral
- 109.5
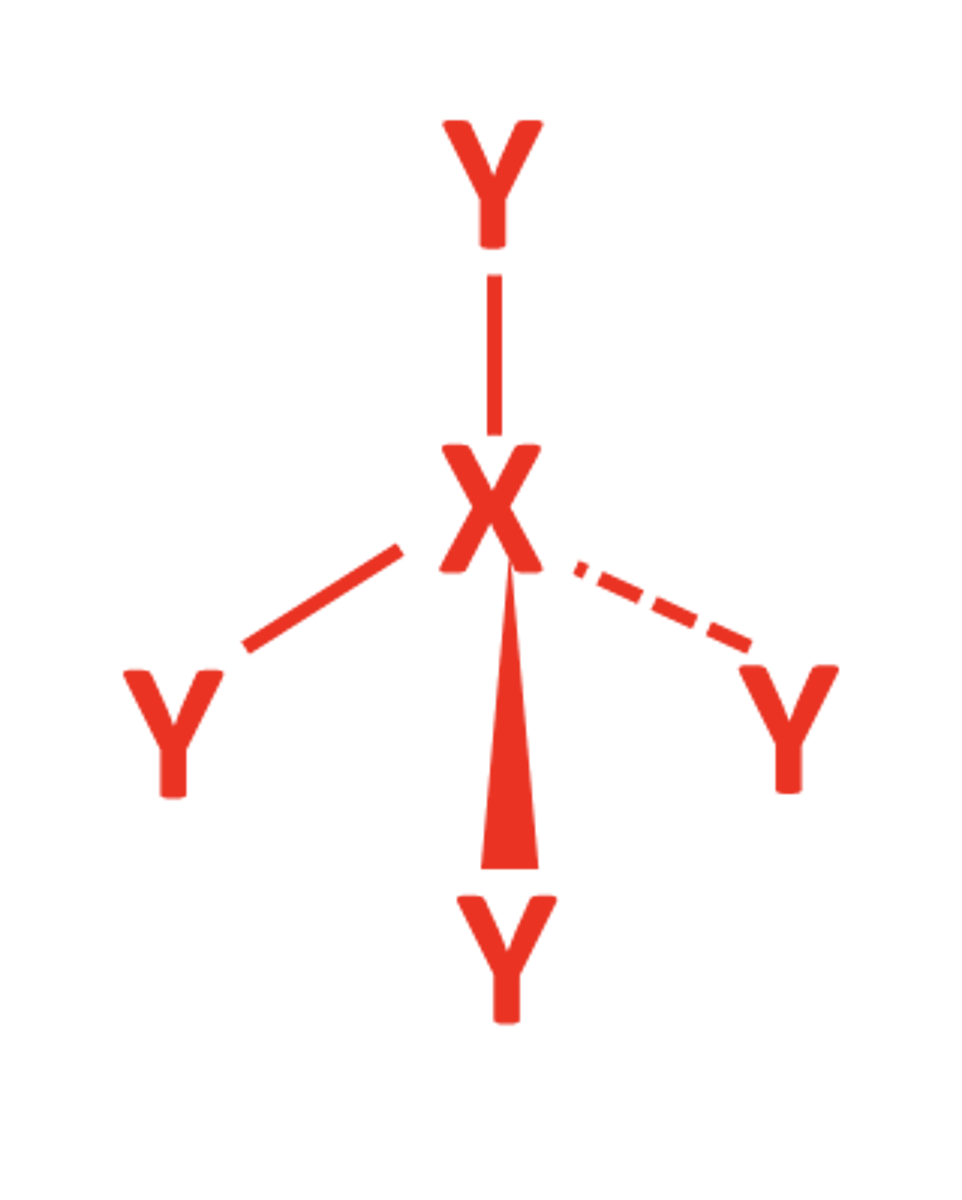
3BP 1LP
- Trigonal Pyramid
- 107
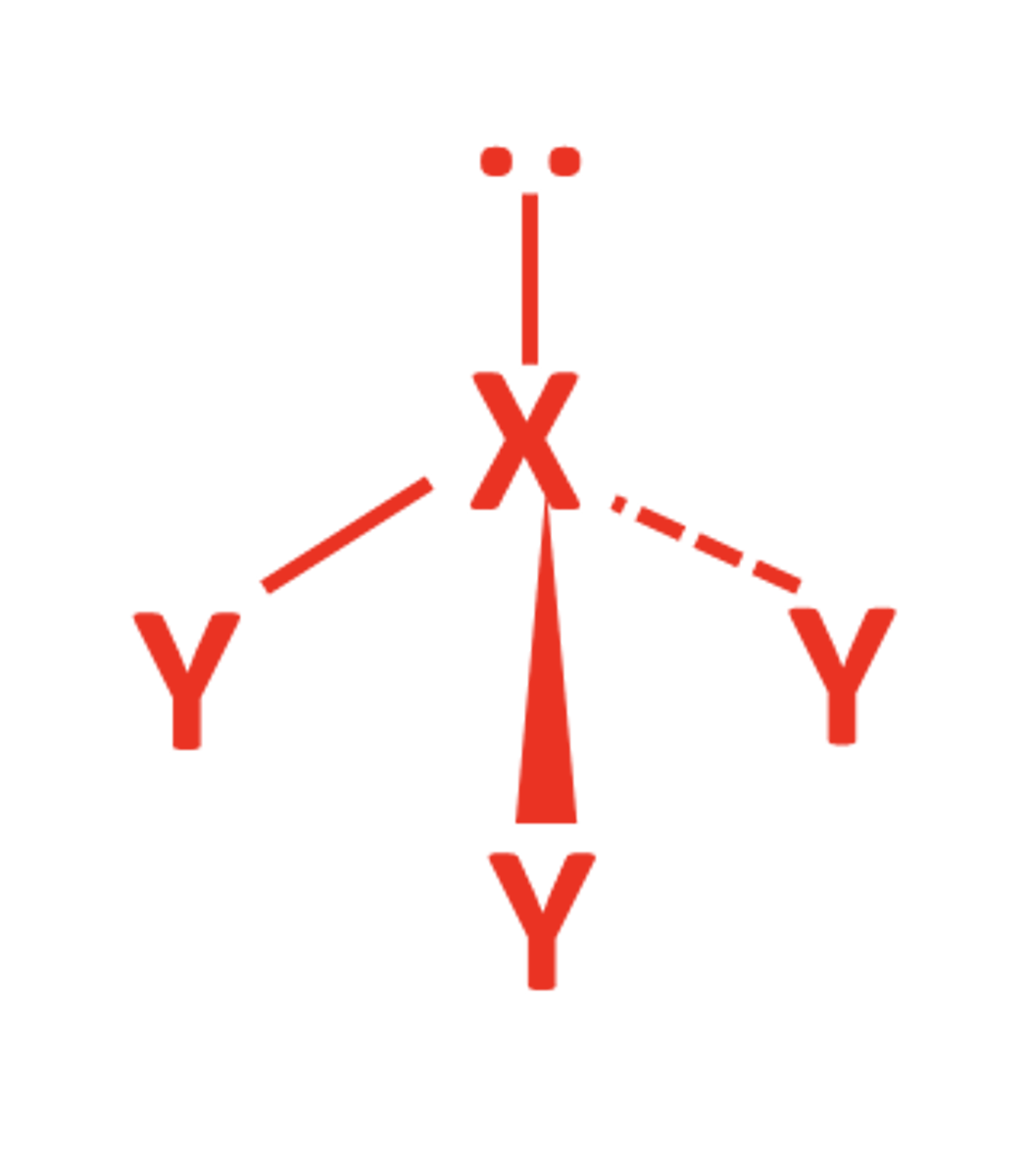
2BP 2LP
- V-shaped / Bent
- 104.5
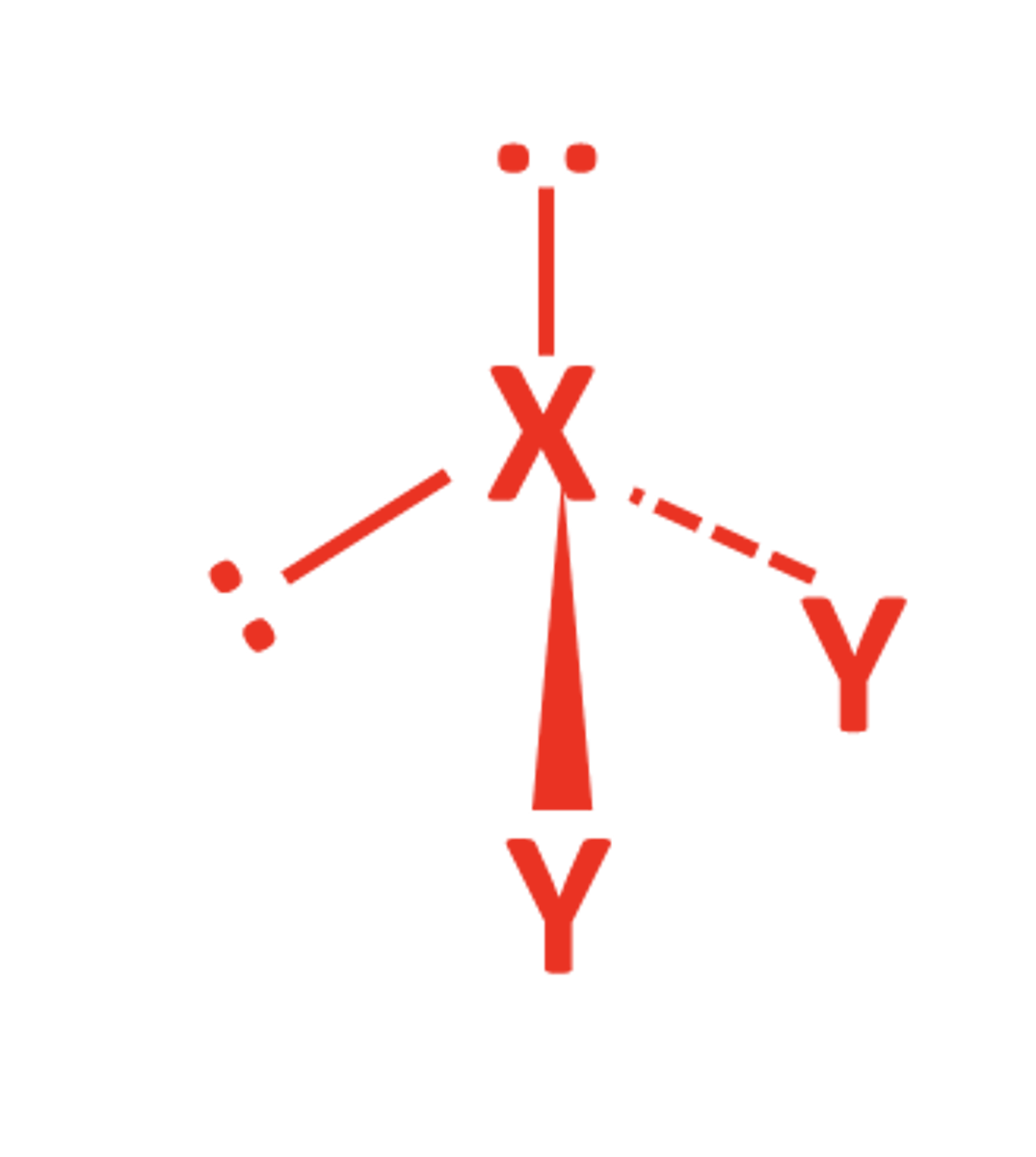
5BP 0LP
- Trigonal bipydramidal
- 120 and 90
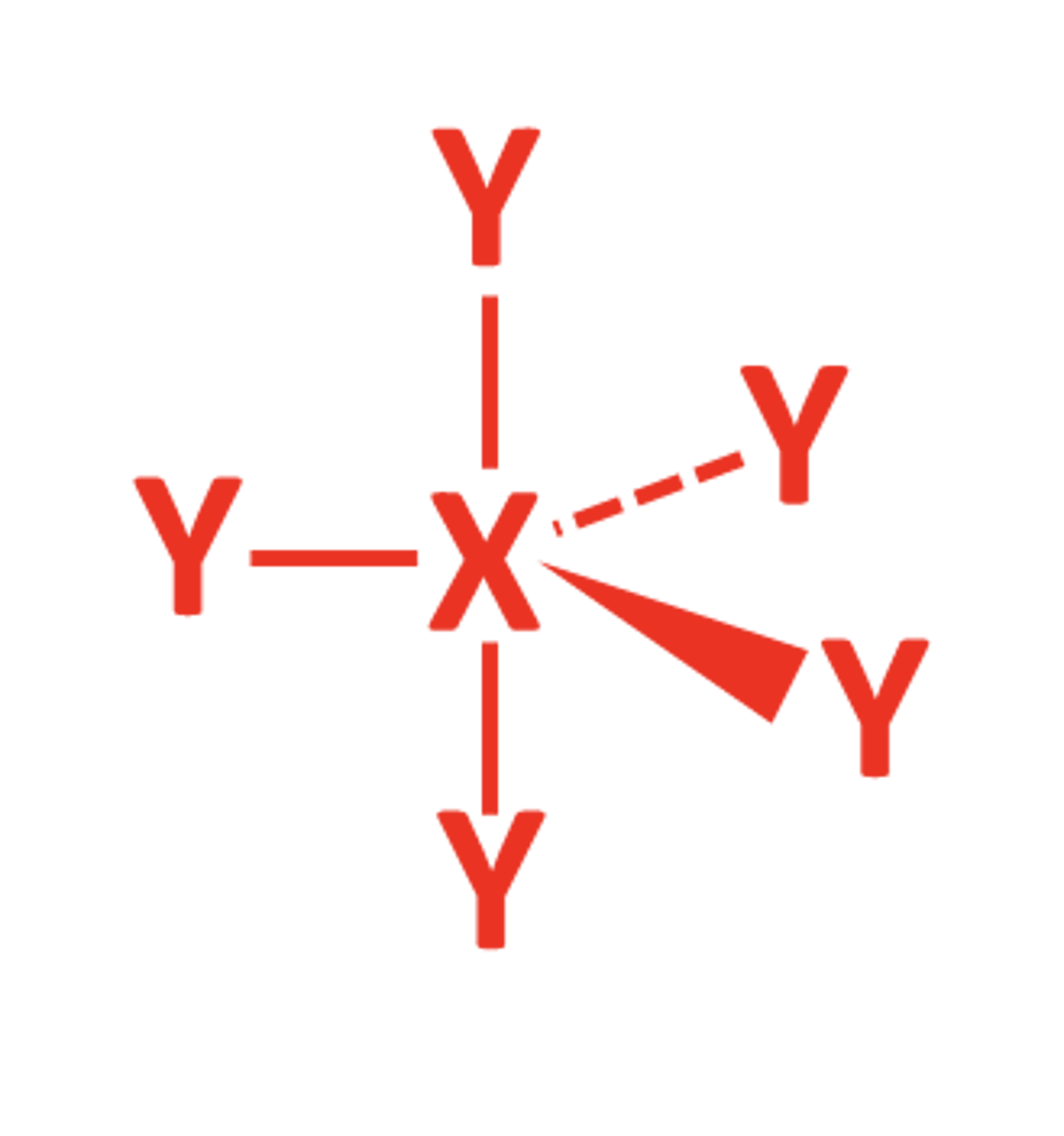
6BP 0LP
- Octahedral
- 90

4BP 2LP
- Square planar
- 90
What does the electron repulsion theory state?
- All electron pairs repel as far away as possible
- LP-LP Repulsion > BP-LP Repulsion > BP-BP Repulsion
NOTE: Use this for any question that asks you to explain the shape or bond angle of a molecule
What is the order of the strength of intermolecular forces? (3)
1. Van der waals (Weakest)
2. Permanent dipole-dipole
3. Hydrogen bonding (Strongest)
Where is van der waals found? (1)
Between ALL molecules
Where is permanent dipole-dipole found? (1)
Between polar molecules
Why do some molecules that posses polar bonds not possess permanent dipole-dipole forces? (2)
- The molecule itself is non polar
- Dipoles cancel out in planar molecules
Where is hydrogen bonding found? (1)
Only between polar molecules with a H-F/O/N bond
How do van der waals forces arise between molecules? (2)
- Uneven distribution of electrons in one molecule
- Induces dipole in a neighbouring molecule
What factors affect the strength of v.d.w? (3)
- Size of molecule (bigger = stronger)
- Mr of molecule (bigger = stronger)
- SA contact (more = stronger)
Why are straight chains of organic compounds harder to boil than branched organic compounds? (2)
- More SA contact
- More vdw forces between molecules
Draw a hydrogen bond forming between two NH3 molecules (3)
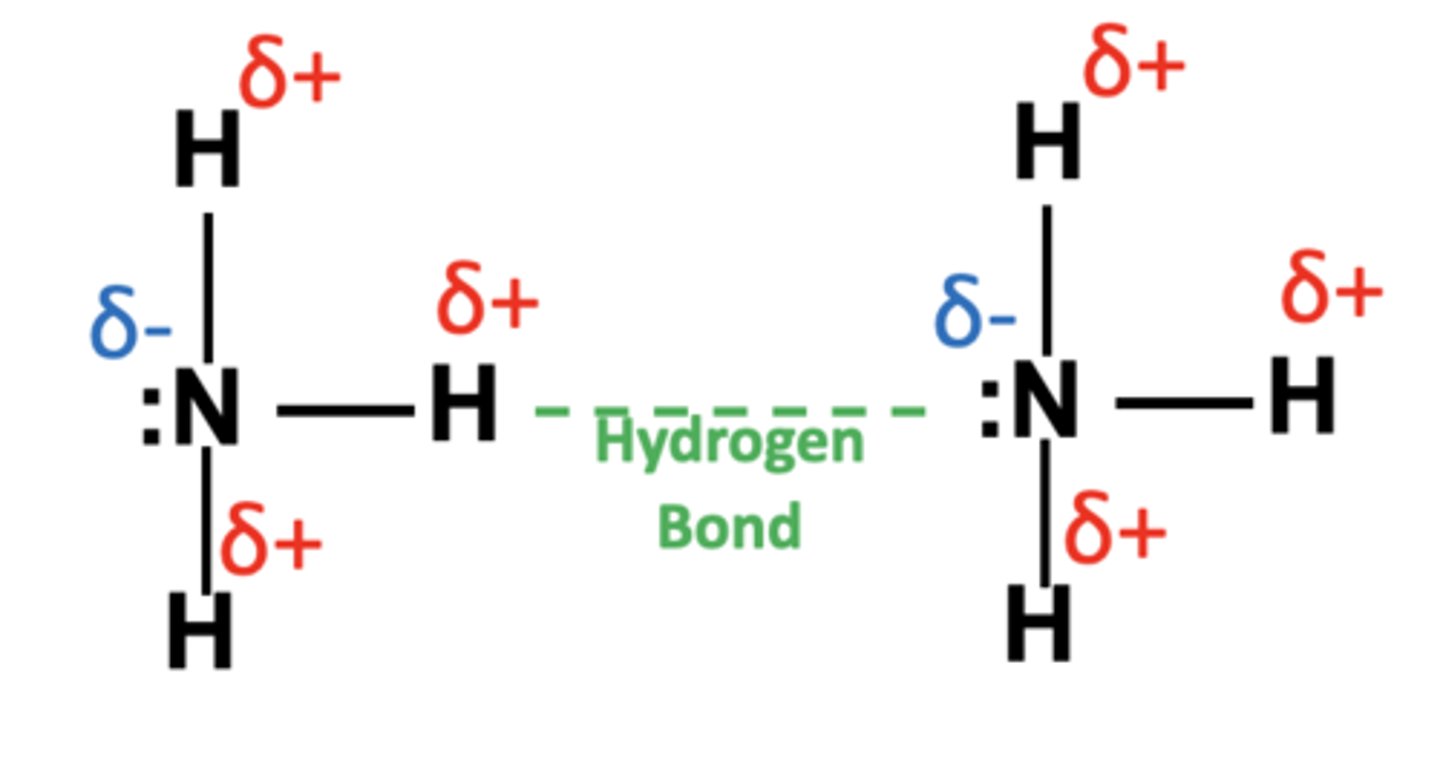
Why is ice less dense than liquid water? (1)
Water molecules are more spread out in ice compared to liquid water
Describe why macromolecular structures have high melting points (2)
- They possess strong covalent bonds between atoms
- Require a large amount of energy to break