CHEM 103 exam 2
1/65
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
66 Terms
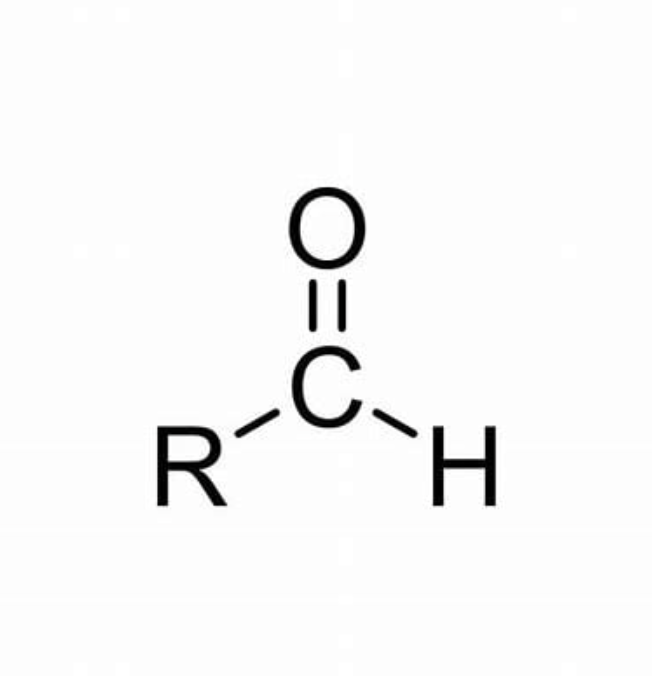
Aldehyde functional group
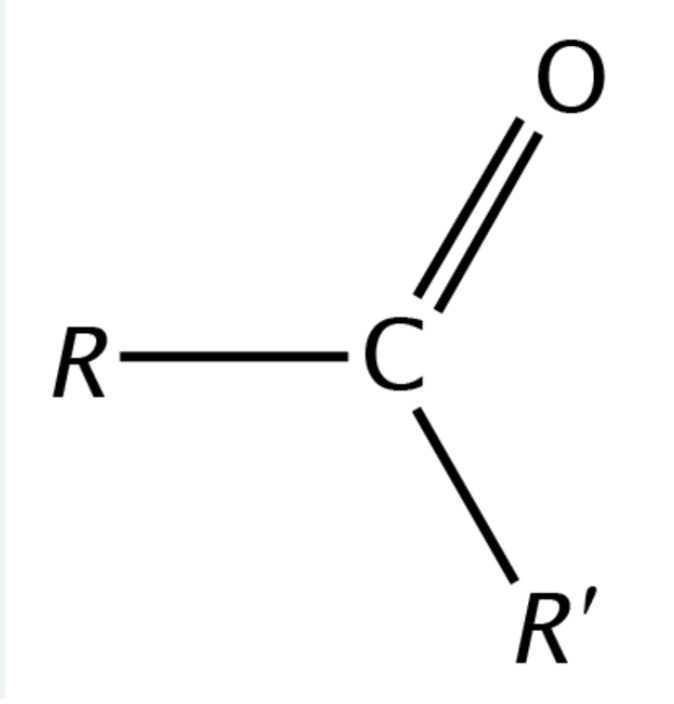
Ketone functional group
what is oxidation of alcohols
primary alcohol → aldehyde → carboxylic acid
what forms when primary alcohol is oxidized
makes carboxylic acid
what does oxidation of secondary alcohols makes
ketone
what happens when tertiary alcohols are oxidized
no reaction
Phenols
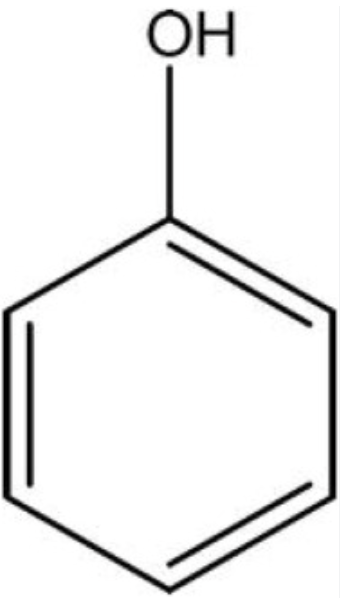
properites of phenols
weak acids
Pka value around 10
insoluble in water but reacts with strong bases
Ether
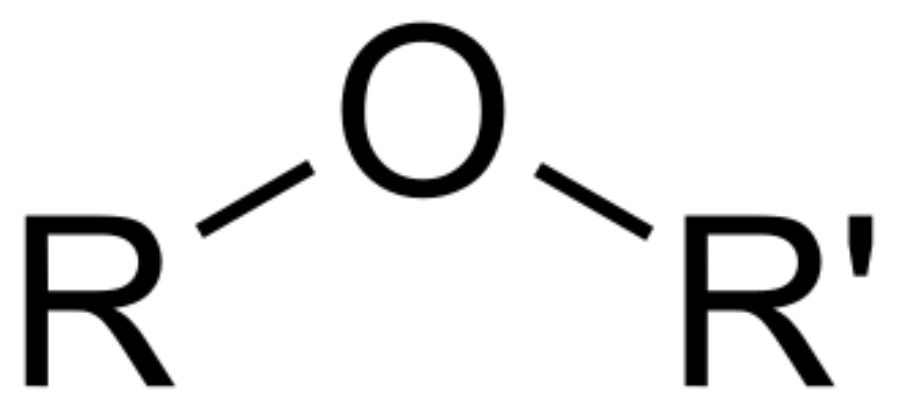
physical properties of ethers
polar compounds
only weak forces of attraction
lower boiling points than alcohols of same molecular formula
reactions of ethers
No oxidation
No reduction
Acid/base resistant
perfect solvents
how are peroxides formed
when ethers oxidize over time
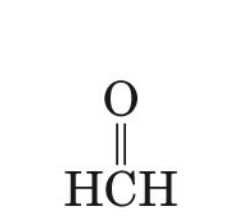
what is the common name of methanal
formaldehyde
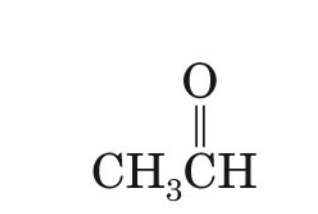
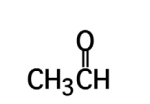
what is the common name of ethanal
acetaldehyde
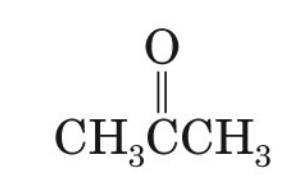
what is the common name of propanone
acetone
methanal
ethanal
propanone
Acetaldehyde
Acetic acid
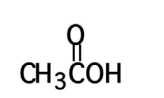
Methyl ethly ketone
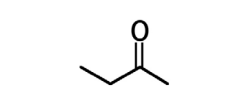
ethyl isopropyl ketone
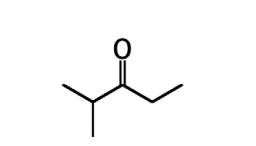
propionaldehyde
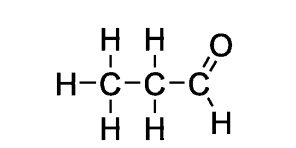
butyraldehyde
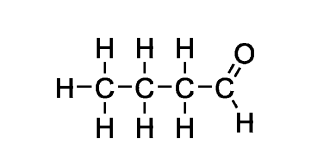
what does alpha mean
carbon 1
what does beta mean
carbon 2
what does gamma mean
carbon 3
what are physical properties of aldehydes and ketones
polar
weak intermolecular attractions (in liquid)
no hydrogen bonding
what is the polarity of carbonyl group
Oxygen is partially negative
carbon is partially positive Carbon
boiling point ranking (lowest to highest)
ether/ alkane → ketone/ aldehyde → alcohol → carboxylic acid
what are aldehydes oxidized to
carboxylic aicds
what does reduction of alcohol give
primary alcohol
what does reduction of ketone give
secondary alcohol
What are the two main ways to reduce aldehydes and ketones
Hydrogen gas with a metal catalyst.
Sodium borohydride
carboxylic acid function group
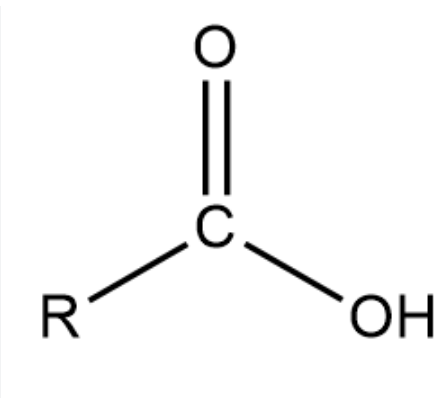
what is the common name of methanoic acid
formic acid
what is the common name of ethanoic acid
acetic acid
what is the common name of propanoic acid
propionic aicd
what is the common name of butanoic acid
butyric acid
physical properties of carboxylic acids
3 polar covalent bonds
much higher boiling points compared to other organic molecules
more soluble in water
weak acids
reactions of carboxylic aicids
all reacte with NaOH, KOH, and other strong bases to for water soluble groups
how many carbs need to be soluble in water
5 or more
what is the functional group of ester
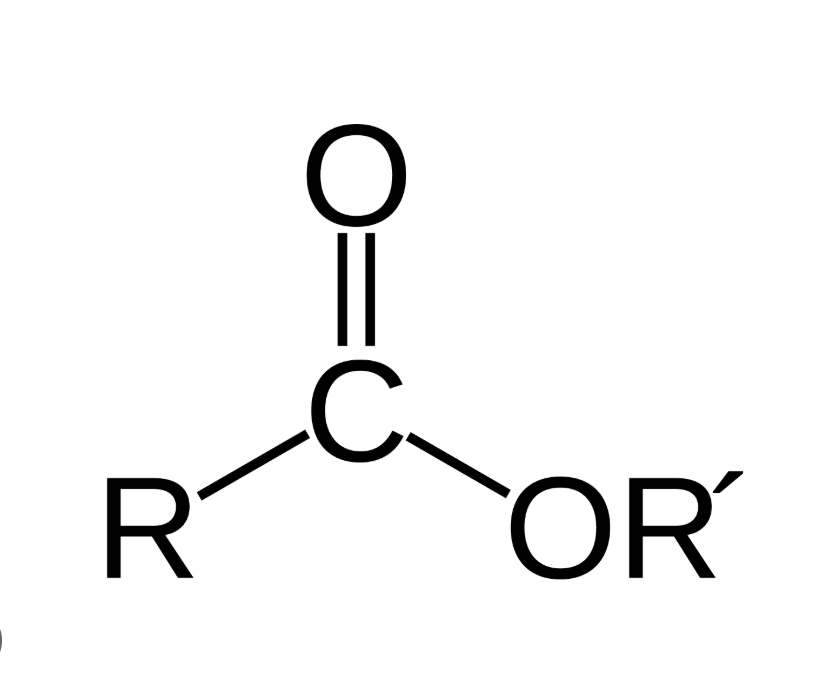
what happens during hydrolysis of ester
forms carboxylic acid and an alcohol
ionization of amines
reaction between an amine and water involves transfer of a proton from water to the amine
Neutralization of amines
whether soluble or insoluble in water, react quantitatively with strong acids to form water-soluble salts
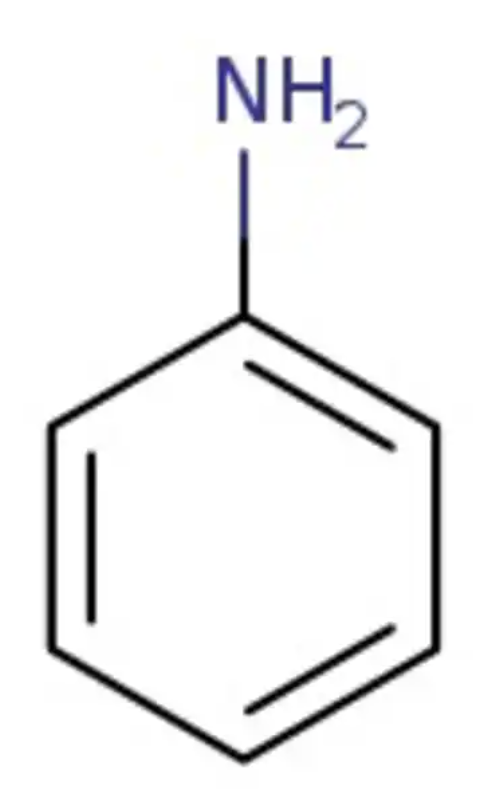
aniline
propylamine
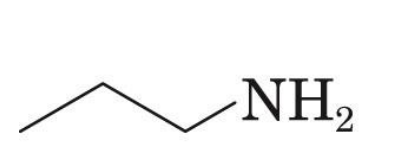
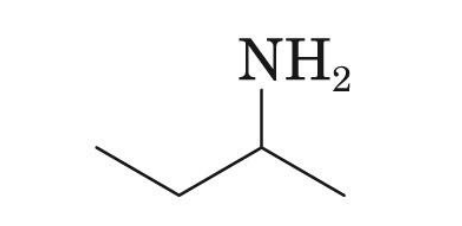
sec- Butylamine
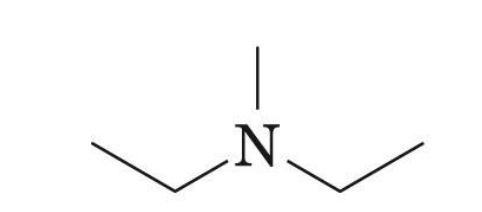
diethlymethlyamine
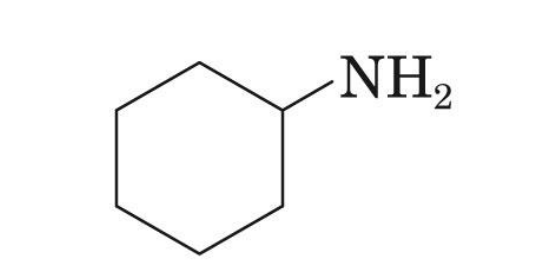
Cyclohexyamine
physical properties of amines
when low molecular weight strong order
polar
which degree of amines form hydrogen bonds
only 1 and 2
as molecular weight increases what happens
becomes less soluble in water
what is the common name for methanamide
formamide
what is the common name for ethanamide
acetamide
how to synthesize amides
treating a carboxylic acid with ammonia and heat
Hydrolysis of Amides
Amides require more vigorous conditions for hydrolysis in both acid and base than do esters
Hydrolysis in hot aqueous acid gives a carboxylic acid and an ammonium ion.
Thiol
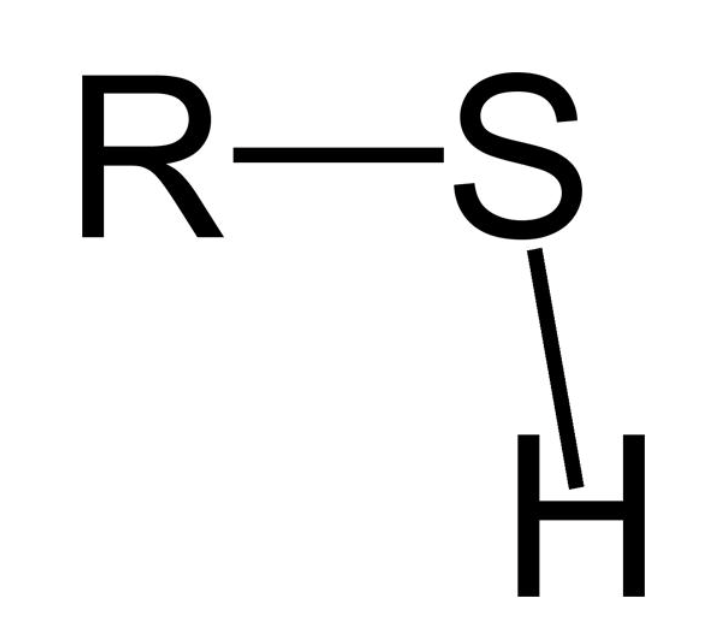
properties of thiol
very bad smell (ex skunks and onions)
Oxidation of Thiols
Thiols are readily oxidized to disulfides by O2
what is polymer
any long-chain molecule synthesized by bonding together many single parts
what is monomer
single unit
Addition Polymer
polymer that forms by simple linking of alkene monomers without the co-generation of other products
Low-density polyethylene (LDPE)
highly branched polymer
polymer chains do not pack well and London dispersion forces between them are weak
More flexible, more transparent
ex. ziplock bags
High-density polyethylene (HDPE)
Straight-chain polymer
chains pack well & London dispersion forces between them are strong
Stronger, more rigid, less transparent
ex. milk containers