Reactions of Amides and Enols/Enolates
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
How do you make an amine with an alkyl halide (a carbon chain with a Br, I, Cl, I)?
Use NaCN/Sn2 (This makes C-N bond) —>
Use 1. xs LAH/ 2. H2O (This makes RNH2) —>
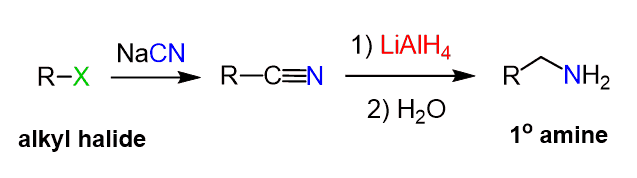
How do you make an amine from a carboxicilic acid (COOH)?
Use 1. SoCl2/ 2. xs NH3 (This makes an acid chloride) —>
Use 1. xs LAH/ 2, H2O (This makes RNH2)—>
How do you make an amine from benzene (6 membered ring with double bonds)?
Use HNO3/H2SO4 (this creates NO2 substitiuent)
Use H2/Pt OR 1. Fe, Zn, Sn, SnCl2/ 2. NaOH (This makes RNH2)
How do you make an amine using an azide (N=N=N)?
Use Na3N (This attaches N3 to end of chain)
Use H2/Pt OR 1. LAH/ 2. H2O (This makes RNH2)

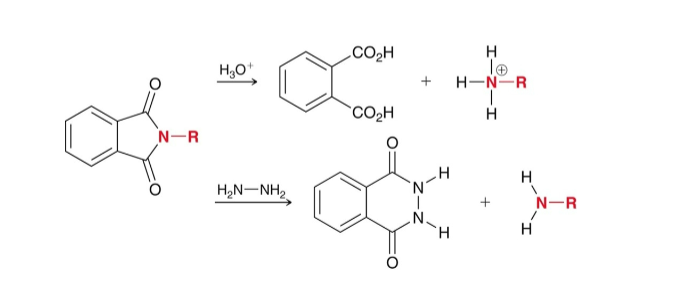
How do you make an amine using Gabriel synthesis (pthalimide)?
First make Potassium Pthalimide by using KOH (gets rid of Hydrogen on pthalimide)
Potassium Pthalimide is alkylated with RX/Sn2 (This adds an R group to the N on the Pthalimide)
Liberate the amine with H3O+ or 2NH2 (This creates RNH2)
How do you make a primary amine with reductive amination (starting from a ketone)?
Use NH3/H+, NaBH3CN
How do you make a secondary amine with reductive amination (starting from a ketone)?
Use RNH2/H+, NaBH3CN
How do you make a tertiary amine with reductive amination (starting from a ketone)?
Use R2NH/H+, NaBH3CN
What three ways can you make a primary amine?
Use Gabriel Synthesis (Potassium Pthalimide Group—> 1. RX/NH2NH2)
Use Azide Synthesis (N=N=N —> 1. RX/ 2. H2, Pt
Use Reductive Amination (NH3 —> ketone + NaBH3CN)
How do you add an alkyl group to an amine?
Use an acyl chloride (ketone with a chlorine)
What should you do if you want to brominate or do a friedal crafts on an amide ring?
Use an acyl chloride to protect (This turns NH2 into a NH)
Then use Br2 (This adds Br on correct position)
Use NaOH to return NH2 to normal
Friedal Crafts is same thing except CH3Cl
What is the sandmeyer reaction?
A diazonum salt (R-NN) is replaced with (Br, CN, I, or Cl) with the reagent Cu(Br, Cl, I, CN)
How can you use a sandmeyer reaction to install a carbox. acid (COOH)?
First use HNO3/H2SO4 (This makes NO2)
Next use Fe, H3O+ and NaOH (This inevitably makes NH2)
Then use NaNO2, HCl (This makes the diazonium salt)
This where the intimediary is
Finally, use 1. CuCn/ 2. H3O+, heat to add COOH
How do you add a F starting with a diazonium salt (NN)?
Use HBF4 (This replaces NN with F)
How do you add an OH starting with a diazonium salt?
Use H2O/heat (this replaces NN with OH)
How do you add H starting with a diazonium salt?
Use H3PO2 (This replaces NN with H)
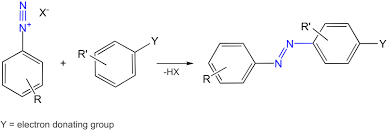
How does azo coupling work/ what does it look like?
It adds an R group to one of the N’s, and the N triple bond becomes an N double bond
How do you make a mixture of ketone and enolate from a ketone (reversible)?
Use NaOH
How do you make an enolate from a ketone (irreversible)?
Use LDA or NAH
What reagents do you use to halogenate a ketone?
Use H3O+/Br2
Will the enolate form under acidic or basic conditions?
Basic (-OH)
What do you use for an aldol condensation reaction/what does it look like?
Use H or OH/heat
The E isomer will be favored
THIS ONE GETS DRAWN WEIRD REFER TO NOTES
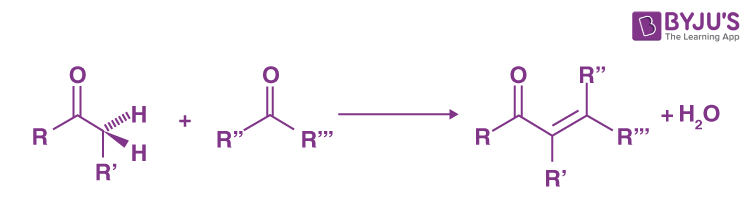
How do intramolecular aldol reactions work?
Use NaOH, H2O/heat
COUNT FROM ONE C=O TO END OF CHAIN
CREATE A RING AND INSTALL DOUBLE BOND
IF GOING IN REVERSE YOU CUT THE DOUBLE BOND
What do you use for claisen condensation and what does it look like?
Use NaOEt/H3O
SAME THING AS CONDENSATION PRODUCT YOU PUT THE TWO MOLECULES TOGETHER JOINED BY DOUBLE BOND
What do you use for a dieckmann reaction and what does it look like?
Use NaOEt/H3O
SAME THING AS INTRAMOLECULAR
YOU CREATE A RING
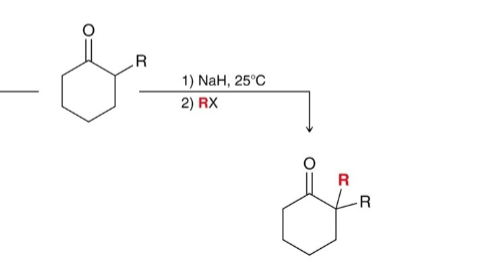
What do you use for alkylation of the alpha position?
Use LDA/RX
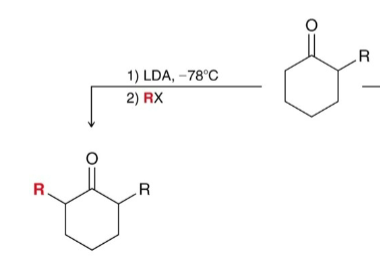
How do you get the thermodynamic product from alkylation (less substitued)?
Use LDA at low temp (-78)
How do you get the kinetic product from alkylation (more substituted)?
Use NaH at 25