Final Lab Exam Practice: Chem II
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
Expanded structures
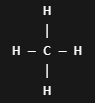
condensed structures
CH₃CH₃, CH₃CH₂OH
line-angle/skeletal structures
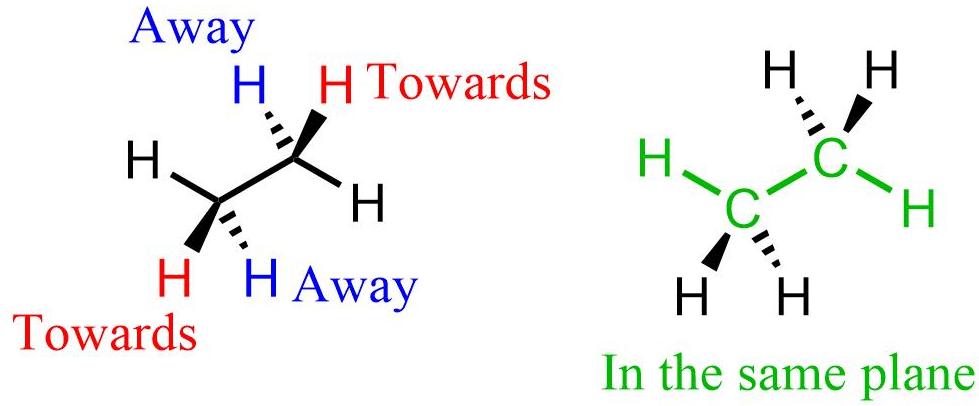
3D structures
alkene
C=C
alkyne
C≡C
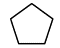
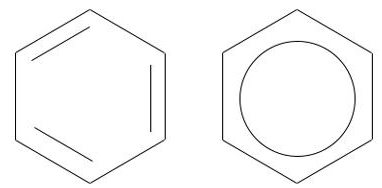
cycloalkane
aromatic
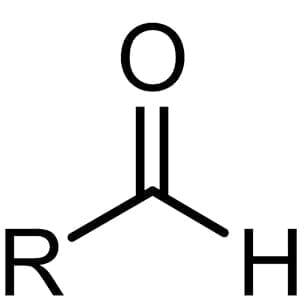
aldehyde
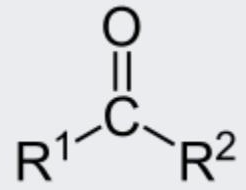
ketone
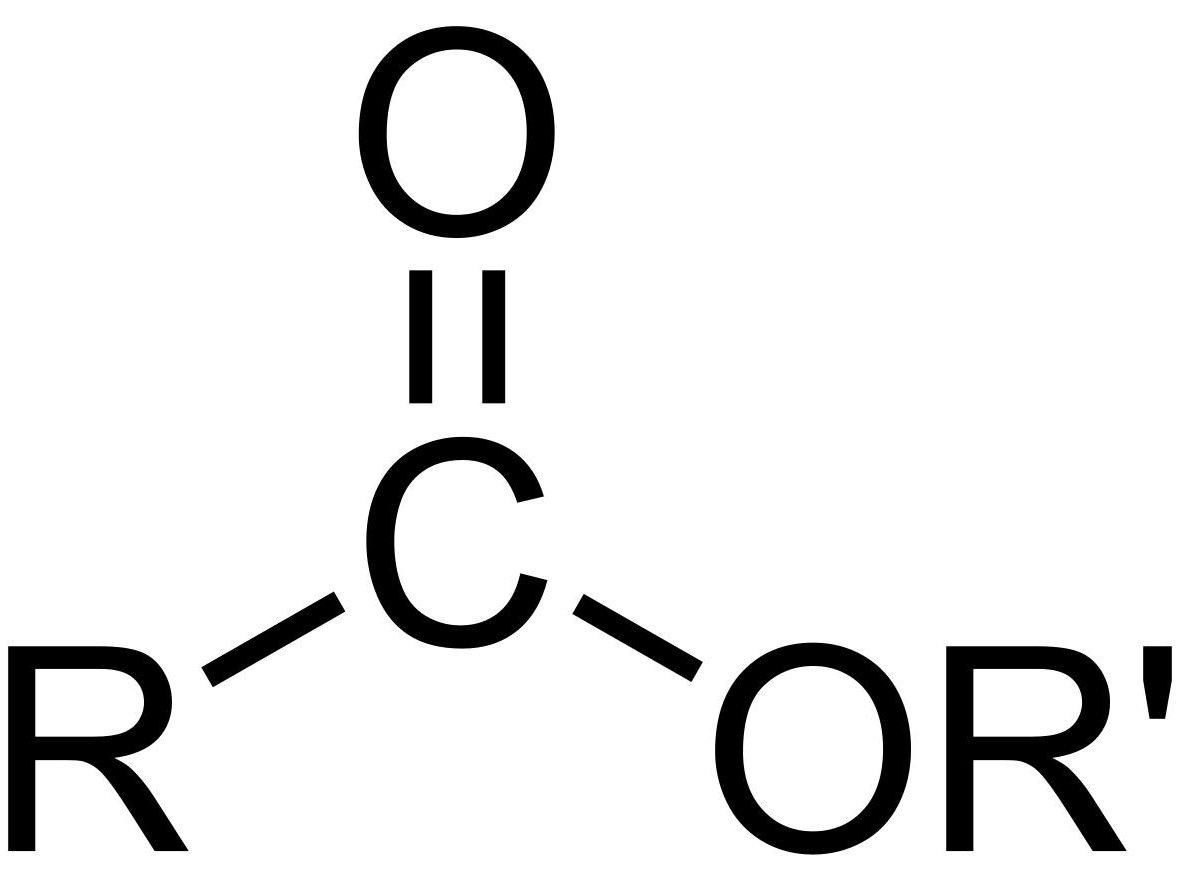
ester
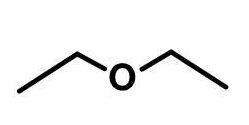
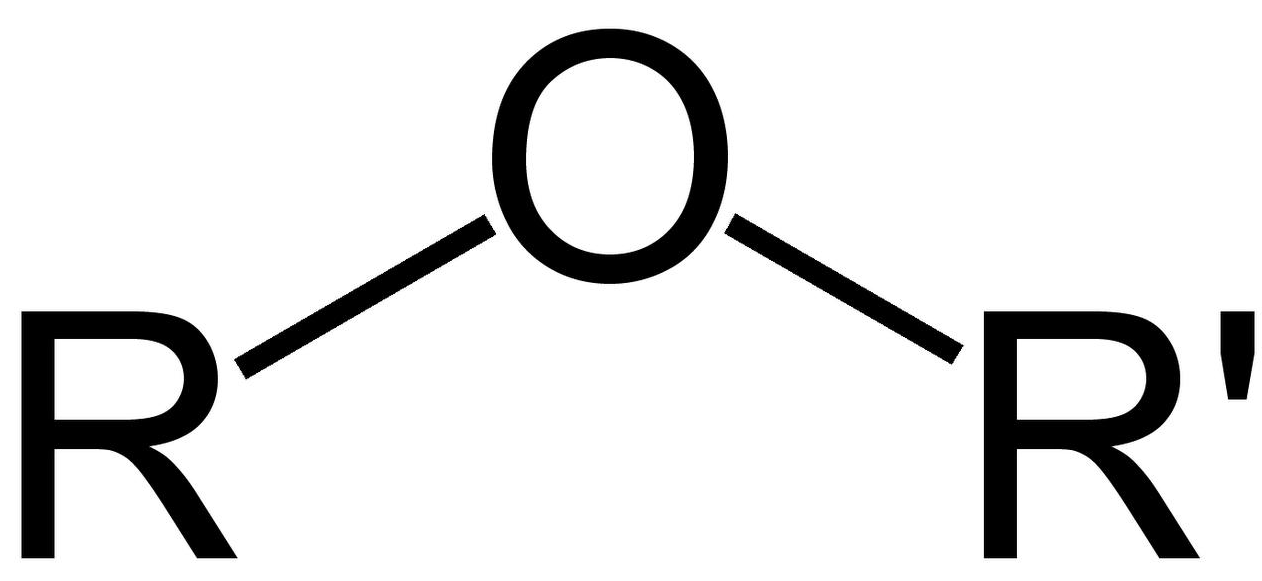
ether
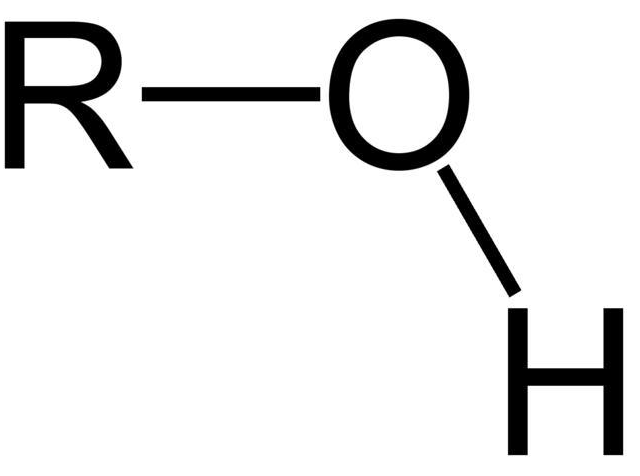
alcohol
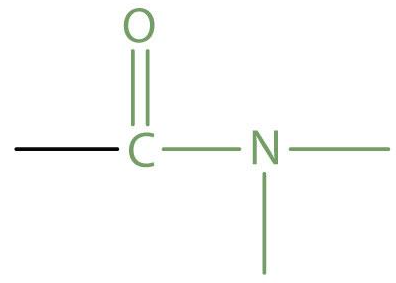
amide
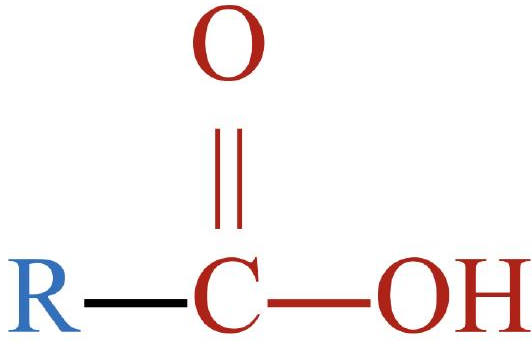
carboxylic acid
functional group isomers
same molecular formula but different functional group and properties
chain isomers
isomers that have different structures
positional isomers
isomers that have R groups in different positions
geometric isomers
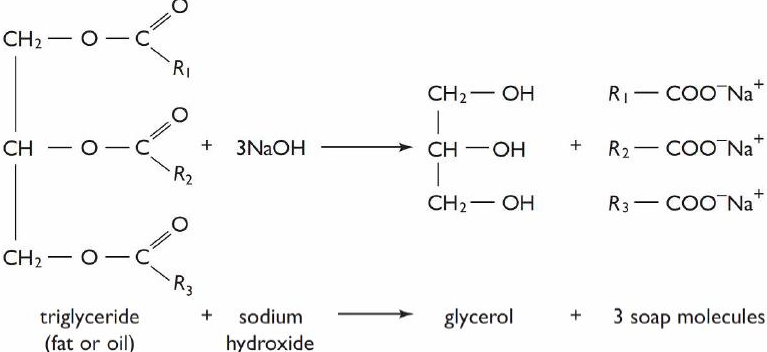
CH2O-C=O—R + 3NaOH > CH2OH + R-C=O O-Na+
Hard water
water that contains calcium and magnesium ions
calcium and magnesium turn it into an insoluble solid (soap scum).
Why does soap not dissolve/precipitate in hard water?
pH, foam (lather), cleansing efficiency, hardness, total fatty matter, moisture content, free alkali
What test can be done to test quality of soap?
the polar head dissolves in water and nonpolar tail that attaches to grease, allowing it to surround and wash away nonpolar substances
How does soap clean in terms of chemical structure of soap, which makes it soluble in water and able to remove non-polar substances?
monosaccharides and disaccharides such as maltose and lactose (reducing sugars) will answer positive; sucrose and polysaccharides such as starch (non-reducing sugars) will answer negative
Benedict’s test
glucose is oxidized to a carboxylic acid while Cu²⁺ is reduced to Cu⁺, forming a red precipitate
What chemical changes happen Glucose in the Benedict’s test?
acts as a reducing agent by donating electrons and being oxidized to a carboxylic acid
The aldehyde group of glucose does what?
forms a red precipitate
The calcium ions of Benedict’s solution does what?
gold, green, orange, red
What is the color change for a positive Benedict’s test?
deep blue
What is the color change for I2 test with starch?
waxes, triacylglycerols, glycerophospholipids, sphingolipids
Types of lipids
liquid state, unsaturated, linked structure, low melting point, insoluble in water
Physical properties of liquid lipids (oils)
solid state, saturated, straight structure, high melting point, insoluble in water
Physical properties of solid lipids (fats)
Bonds: single bonds only
shape: straight
melting point: high
room temp state: solid
Common sources: animal fats
Saturated lipids
bonds: at least 1 double bond
shape: linked/bent
melting point: low
room temp state: liquid
common sources: plant oils
Unsaturated lipids
brown/yellow-brown
Color change for I2 test for unsaturated lipids
Regular: heavy processing (heat); mono + poly fatty acids; lowest health value
Virgin: moderate processing; mostly mono fatty acids; good health value
Extra virgin oils: minimal processing; mostly mono (intact) fatty acids; highest health value
compare fatty acid contents of regular, virgin, extra virgin oils, and suitability for human consumption
peptide; hydrogen; disulfide (covalent), ionic, hydrogen, hydrophobic
Chemical bonds responsible for primary, secondary, tertiary structures of proteins
heat, pH, salt concentration, organic solvents, heavy metals, mechanical agitation
factors that denature a protein
Basic: accept H+, positively charged, contain amino groups
Acidic: donate H+, negatively charged, contain extra carboxyl groups
Basic and acidic amino acids
secondary, tertiary, and quaternary lose shape and function
What structures of a protein are changed in protein denaturation?
Biuret test
Which test indicates Prescence of protein?
ninhydrin test
Which test indicates presence of amino acid?
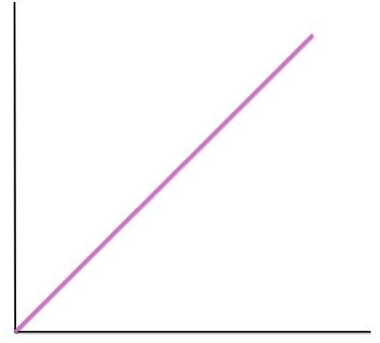
reaction rate vs enzyme concentration
linear shape
reaction rate vs substrate concentration
rate increases with [][][] until it reaches a maximum when all enzyme active sites are saturated
![<p>rate increases with [][][] until it reaches a maximum when all enzyme active sites are saturated</p>](https://assets.knowt.com/user-attachments/05bed19b-d97d-42d8-92f4-2feff6954e2d.png)
reaction rate vs temperature
rate increases with [][][] up to an optimum, then decreases as enzymes denature
![<p>rate increases with [][][] up to an optimum, then decreases as enzymes denature</p>](https://assets.knowt.com/user-attachments/d1b2bdae-2309-4d3d-ba40-a207430591b9.png)
reaction rate vs pH of enzymatic reaction
rate increases to an optimal [][][] and then decreases as [][][] changes disrupt the enzyme’s structure
![<p>rate increases to an optimal [][][] and then decreases as [][][] changes disrupt the enzyme’s structure</p>](https://assets.knowt.com/user-attachments/1791e5ae-020a-4a60-874c-e8e7eadc15ac.png)
bicarbonate buffer system (H2CO3 / HCO3-)
Which is the most important buffer system in human blood?
excess fat breakdown producing acidic ketone bodies, usually due to lack of insulin
Ketoacidosis is result of:
blood pH increases
How does blood pH change during hyperventilation?
equilibrium shifts to the left
During hyperventilation, HCO3- equilibrium shifts:
Breathing into a paper bag increases CO₂ in the blood, which lowers pH back toward normal
How does breathing into a paper bag help return pH levels to normal?
Blood pH increases
How does blood pH change when someone overdoses on antacids?
equilibrium shifts to the left
In overdose of antacid, which direction does equilibrium change?
soluble
Hydrocarbons (hexanes, methylene chloride) are _____ in non-polar solvents
decreases
Length of carbon chain ____ solubility in polar solvents
hydrogen bonds
Alcohols, amines, and carboxylic acids form ____ ____
Ionic: strongest, +/-
Hydrogen: moderately strong
dipole-dipole: partial +/-, weak (HCl and HCl)
van der Waals (London dispersion): weakest, present in all
Difference of intermolecular interactions
weight (g) / volume (mL)
Density formula
lower and wider range of melting point
2 most common effects on melting point if organic compound is impure