3.5 - 3.7 - chemical bonding
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
VSEPR theory
The valence shell electron pair repulsion theory (VSEPR) predicts the shape and bond angles of molecules
what does VSEPR theory state
that the shape of a molecule or ion is caused by the repulsion between pairs of electrons
In a molecule, bonding and lone pairs of electrons around the central atom will repel each other, so they arrange them as far apart as possible to minimize the repulsion forces, forcing the molecule to adopt a different shape
When determining the shape and bond angles of a molecule, the following VSEPR rules should be considered:
Valence shell electrons are those electrons that are found in the outer shell
Electron pairs (whether they are bonding or lone pairs) repel each other as they have similar/like charges
Lone pair electrons repel each other more than bonded pairs
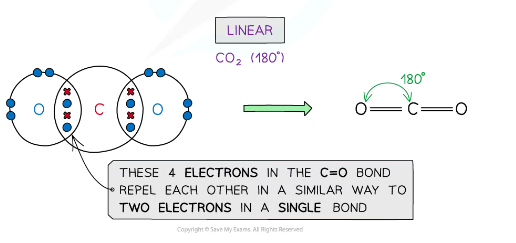
Repulsion between a multiple (double and triple) and single bonds is treated the same as for repulsion between two single bonds
Repulsion between pairs of double bonds are greater
The most stable shape is adopted to minimize the repulsion forces
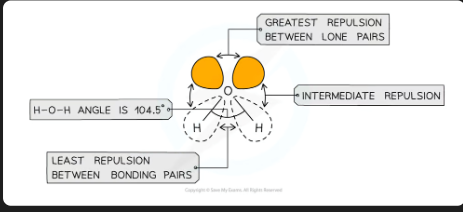
Different types of electron pairs have different repulsive forces
Lone pairs of electrons have a more concentrated electron charge cloud than bonding pairs of electrons
The cloud charges are wider and closer to the central atom’s nucleus than bonding pairs (which are between two nuclei)
The order of repulsion is therefore: lone pair – lone pair > lone pair – bond pair > bond pair – bond pair

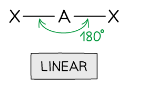
what is the shape of a molecule with 2 bonding pairs of electrons and zero lone pairs and what are the angles between each atom
linear, 180
what is shape of a molecule with 2 bonding pairs of electrons and 2 lone pairs and what are the angles between each atom
v-shape/non-linear/bent, 104.5
what is the shape of a molecule with 3 bonding pairs of electrons and zero lone pairs and what are the angles between each atom
trigonal planar, 120
what is the shape of a molecule with 4 bonding pairs of electrons and zero lone pairs and what are the angles between each atom
tetrahedral, 109.5
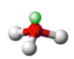
what is the shape of a molecule with 3 bonding pairs of electrons and one lone pair and what are the angles between each atom
pyramidal, 107
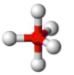
what is the shape of a molecule with 5 bonding pairs of electrons and zero lone pairs and what are the angles between each atom
trigonal bipyramid, 120 and 90
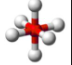
what is the shape of a molecule with 6 bonding pairs of electrons and zero lone pairs and what are the angles between each atom
octahedral, 90
example of a molecules with a linear shape
CO2
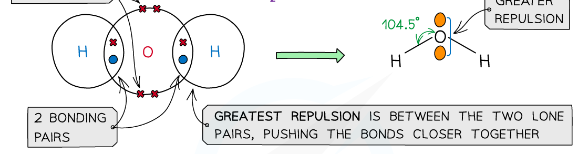
example of a molecules with a bent/non-linear/v shape
H2O, H2S
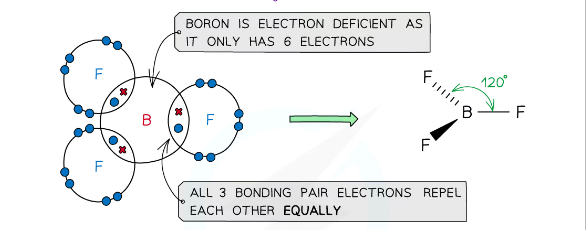
example of a molecules with a trigonal planar shape
BF3, AlF3
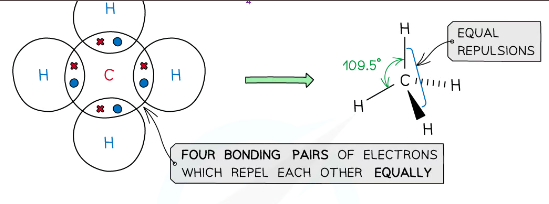
example of a molecules with tetrahedral shape
CH4, NH4+
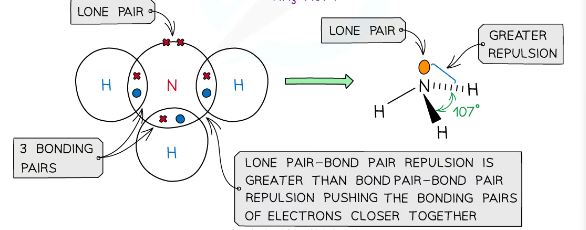
example of a molecules with pyramidal shape
NH3, PH3
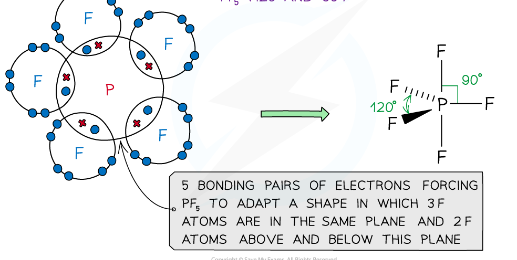
example of a molecules with a trigonal bipyramid shape
PF5, PCl5
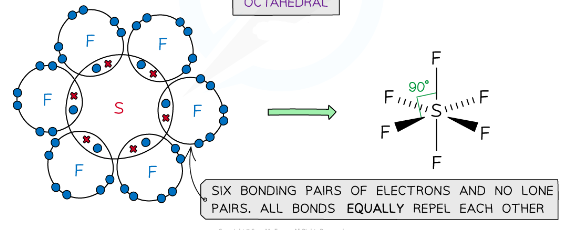
example of a molecules with an octahedral shape
SF6, PCl6-
what type of bonding is hydrogen bonding
the strongest form of intermolecular bonding
Intermolecular bonds are bonds between molecules
Hydrogen bonding is a type of permanent dipole – permanent dipole bonding
What is hydrogen bonding/where does it occur
It only occurs in molecules where hydrogen is covalently bonded to either O, N or F. These three elements are so electronegative that they withdraw the majority of the electron density in the covalent bond with hydrogen, leaving the H atom very electron-deficient. The H becomes so δ+ charged that it can form a bond with the lone pair of an O, N or F atom in another molecule
For it to happen, the following is needed:
A species which has an O, N or F (very electronegative) atom with an available lone pair of electrons
A species with an -OH or -NH or -FH group
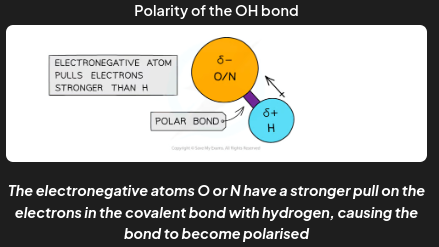
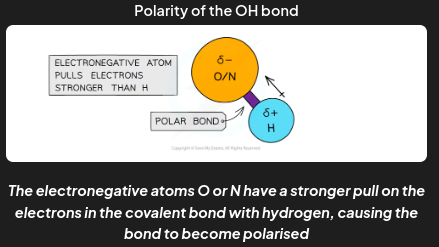
When hydrogen is covalently bonded to an electronegative atom, such as O, N or F, the bond becomes
very highly polarised
The H becomes so δ+ charged that it can form a bond with the lone pair of an O or N atom in another molecule
For hydrogen bonding to take place, the angle between the -OH/-NH/-FH and the hydrogen bond is
180o
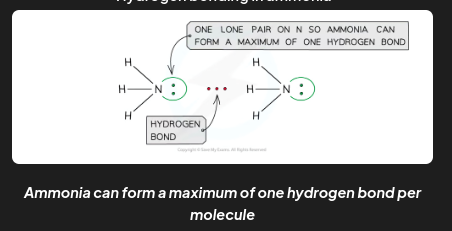
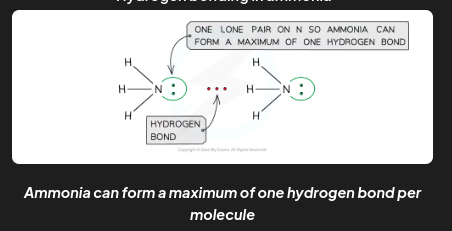
The number of hydrogen bonds depends on:
The number of hydrogen atoms attached to O or N in the molecule
The number of lone pairs on the O or N
properties of water - summary
Hydrogen bonding in water, causes it to have anomalous properties such as high melting and boiling points, high surface tension and anomalous density of ice compared to water
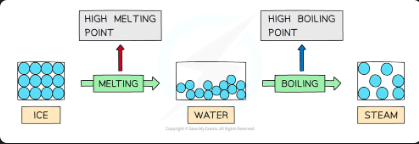
properties of water due to hydrogen bonding - high mp and bp
Water has high melting and boiling points because of the strong intermolecular forces of hydrogen bonding between the molecules
In ice (solid) and water (liquid) the molecules are tightly held together by hydrogen bonds
A lot of energy is therefore required to break the water molecules apart and melt or boil them
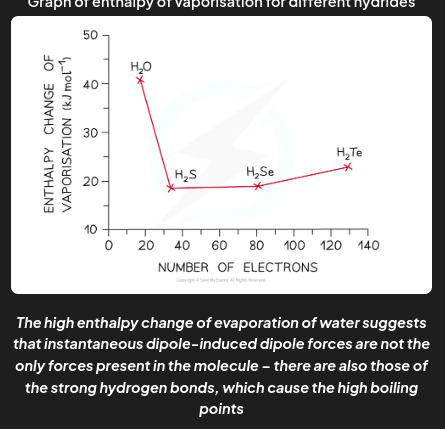
The graph below compares the enthalpy of vaporisation (energy required to boil a substance) of different hydrides. Explain why H2O has the highest enthalpy of vaporization.
As you go from H₂S to H₂Te in Group 16 hydrides, the enthalpy changes increase because the molecules get larger and contain more electrons. This leads to stronger instantaneous dipole-induced dipole forces (also called London dispersion forces). Since H₂O is smaller, you’d expect its enthalpy change to be lower, around 17 kJ mol⁻¹. However, water’s enthalpy of vaporization is almost three times higher due to the strong hydrogen bonds that are unique to water among these hydrides.
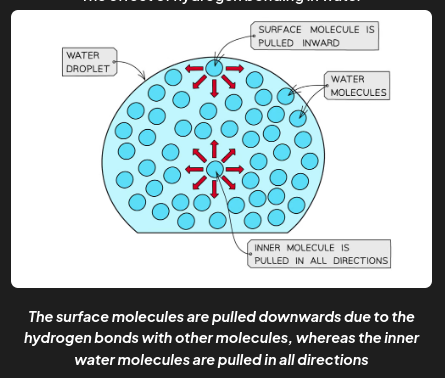
properties of water - high surface tension
Surface tension is the ability of a liquid surface to resist any external forces (i.e. to stay unaffected by forces acting on the surface)
The water molecules at the surface of liquid are bonded to other water molecules through hydrogen bonds
These molecules pull downwards on the surface molecules, causing them to become more compressed and tightly packed at the surface
This increases water’s surface tension
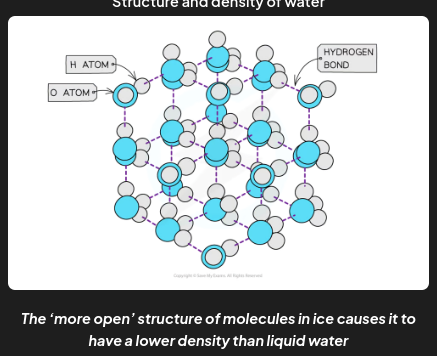
properties of water - density
Solids are denser than their liquids as the particles in solids are more closely packed together than in their liquid state
In ice however, the water molecules are packed in a 3D hydrogen-bonded network in a rigid lattice
Each oxygen atom has a lone pair of electrons causing it to be slightly negative and attracted to the slightly positive hydrogen atoms on adjacent water molecules.
This happens many times so the water molecules are pushed apart into a hexagonal structure which has large spaces within it.
This way of packing the molecules in a solid and the relatively long bond lengths of the hydrogen bonds means that the water molecules are slightly further apart than in the liquid form.
Therefore, ice has a lower density than liquid water
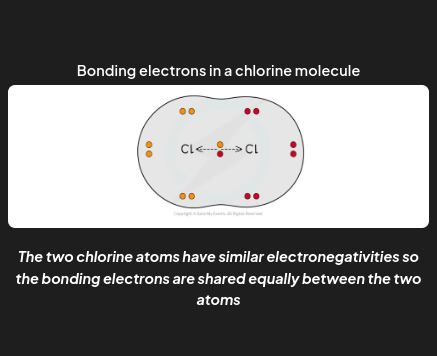
nonpolar covalent bond
When two atoms in a covalent bond have the same electronegativity the covalent bond is nonpolar

polar covalent bond
When two atoms in a covalent bond have different electronegativities the covalent bond is polar and the electrons will be drawn towards the more electronegative atom

As a result of electrons being draw towards the more electronegative atom in a polar covalent bond,
The negative charge centre and positive charge centre do not coincide with each other
This means that the electron distribution is asymmetric
The less electronegative atom gets a partial charge of δ+ (delta positive)
The more electronegative atom gets a partial charge of δ- (delta negative)
The greater the difference in electronegativity the more polar the bond becomes

dipole moment
a measure of how polar a bond is
The direction of the dipole moment is shown an arrow which points to the partially negatively charged end of the dipole:
To determine whether a molecule with more than two atoms is polar, the following things have to be taken into consideration:
The polarity of each bond
How the bonds are arranged in the molecule
Some molecules have polar bonds but are overall not polar because
the polar bonds in the molecule are arranged in such a way that the individual dipole moments cancel each other out - symmetrical non-polar molecules
The polar molecule is an asymmetrical molecule with polar
bonds.
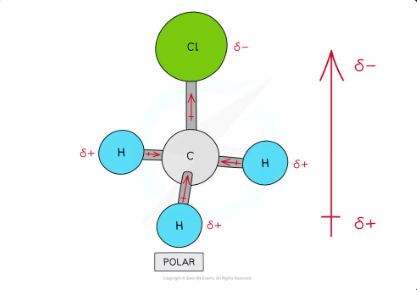
polarity in chloromethane
There are four polar covalent bonds in CH3Cl which do not cancel each other out causing CH3Cl to be a polar molecule; the overall dipole is towards the electronegative chlorine atom
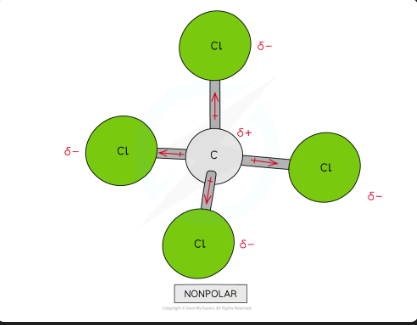
polarity in tetrachloromethane
Though CCl4 has four polar covalent bonds, the individual dipole moments cancel each other out causing CCl4 to be a nonpolar molecules
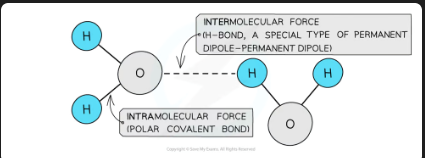
covalent bonds are
strong intramolecular forces
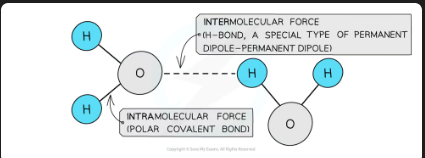
Molecules also contain weaker intermolecular forces which are
forces between molecules
These intermolecular forces are called Van der Waals’ forces
There are two types of van der Waals’ forces:
Instantaneous (temporary) dipole – induced dipole forces also called London dispersion forces
Permanent dipole – permanent dipole forces
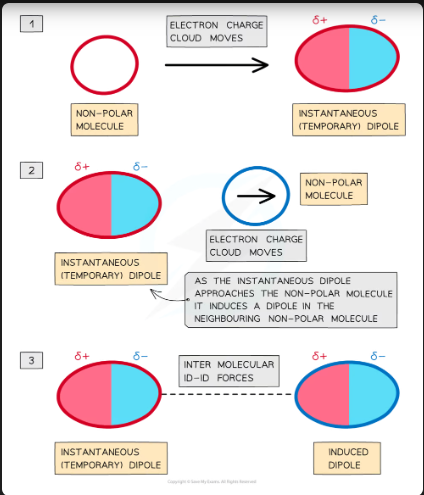
Instantaneous dipole - induced dipole (id - id)
Instantaneous dipole - induced dipole forces or London dispersion forces exist between all atoms or molecules.
The electron charge cloud in non-polar molecules or atoms are constantly moving
During this movement, the electron charge cloud can be more on one side of the atom or molecule than the other
This causes a temporary dipole to arise
This temporary dipole can induce a dipole on neighbouring molecules
When this happens, the δ+ end of the dipole in one molecule and the δ- end of the dipole in a neighbouring molecule are attracted towards each other
Because the electron clouds are moving constantly, the dipoles are only temporary
Larger and heavier atoms and molecules exhibit stronger dispersion forces than smaller and lighter ones.
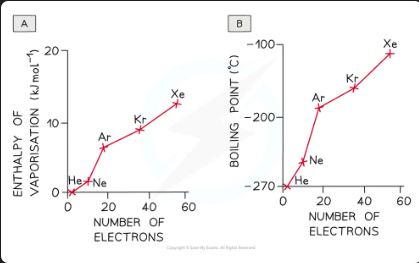
Id-id forces increase with:
Increasing number of electrons (and atomic number) in the molecule
Increasing the places where the molecules come close together
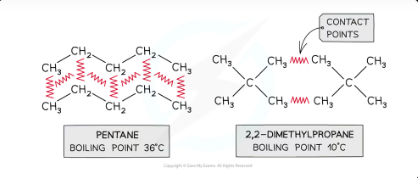
why does pentane have a higher boiling point
The increased number of contact points in pentane means that it has more id-id forces and therefore a higher boiling point
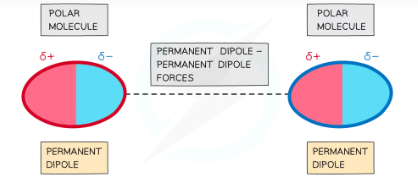
Permanent dipole - permanent dipole (pd - pd)
Polar molecules have permanent dipoles
The molecule will always have a negatively and positively charged end
Forces between two molecules that have permanent dipoles are called permanent dipole - permanent dipole forces
The δ+ end of the dipole in one molecule and the δ- end of the dipole in a neighbouring molecule are attracted towards each other
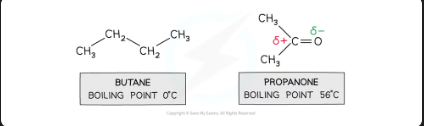
pd-pd vs id-id forces for small molecules with the same number of electrons,
pd - pd forces are stronger than id - id
Butane and propanone have the same number of electrons
Butane is a nonpolar molecule and will have id - id forces
Propanone is a polar molecule and will have pd - pd forces
Therefore, more energy is required to break the intermolecular forces between propanone molecules than between butane molecules
So, propanone has a higher boiling point than butane
hydrogen bonding as a permanent dipole
Hydrogen bonding is an intermolecular force between molecules with an -OH/-NH group and molecules with an N/O atom
Hydrogen bonding is a special case of a permanent dipole - dipole force between molecules
Hydrogen bonds are stronger forces than pd - pd forces
explanation of hydrogen bonding as a permanent dipole
The hydrogen is bonded to an O/N atom which is so electronegative, that almost all the electron density from the covalent bond is drawn towards the O/N atom
This leaves the H with a large delta positive and the O/N with a large delta negative charging resulting in the formation of a permanent dipole in the molecule
A delta positive H in one molecule is electrostatically attracted to the delta negative O/N in a neighbouring molecule
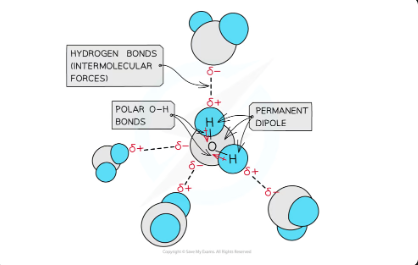
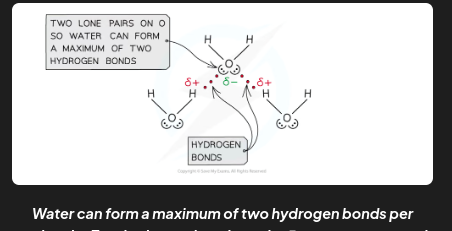
hydrogen bonds in water molecules
Hydrogen bonding in water occurs between the oxygen lone pair of one water molecule and the δ+ hydrogen atoms of another water molecule
intramolecular forces
forces within a molecule
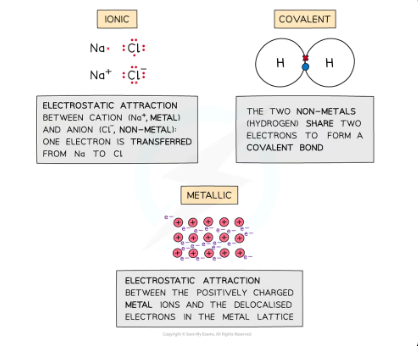
examples of intramolecular forces
ionic bonding, covalent bonding and metallic bonding
intermolecular forces
Intermolecular forces are forces between molecules and are also called van der Waals’ forces
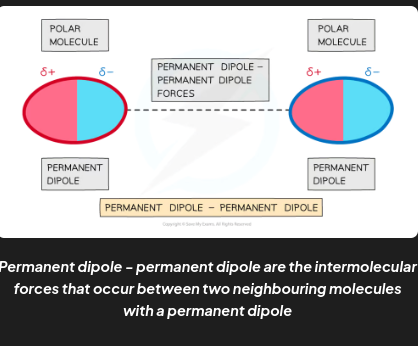
example 1 of intermolecular forces
Permanent dipole - permanent dipole are the attractive forces between two neighbouring molecules with a permanent dipole
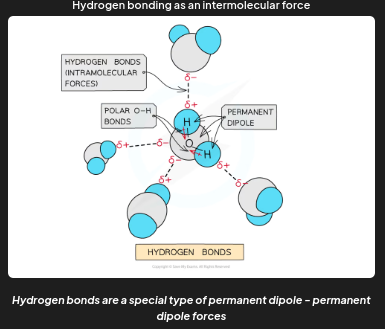
example 2 of intermolecular forces
Hydrogen bonds are a special type of permanent dipole - permanent dipole forces
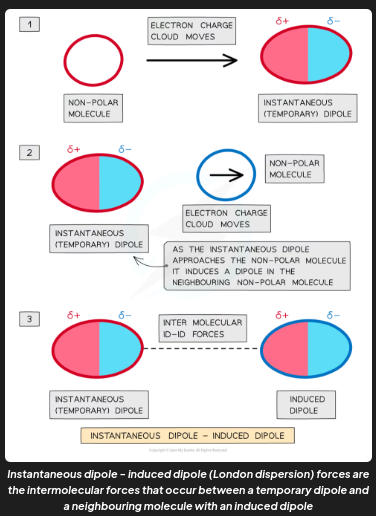
example 3 of intermolecular forces
Instantaneous dipole - induced dipole (London dispersion) forces are the attractive forces between a temporary dipole and a neighbouring molecule with an induced dipole
in general, intramolecular forces are
stronger than intermolecular forces
order the strengths of the types of bonds/forces (pd-pd, hydrogen, covalent, id-id and ionic) from strongest to weakest
ionic, covalent, hydrogen, pd-pd, id-id
dot and cross diagrams
diagrams that show the arrangement of the outer-shell electrons in an ionic or covalent compound or element
The electrons are shown as dots and crosses
in a dot and cross diagram
Only the outer electrons are shown
The charge of the ion is spread evenly which is shown by using brackets
The charge on each ion is written at the top right-hand corner
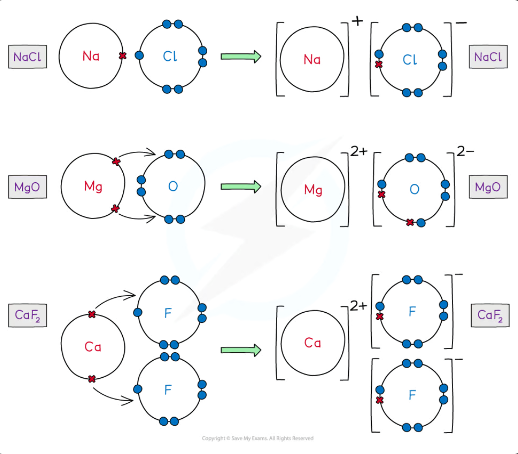
how are ionic bonds formed
Ionic bonds are formed when metal transfer electrons to a non-metal to form a positively charged and negatively charged ion
The atoms achieve a noble gas configuration
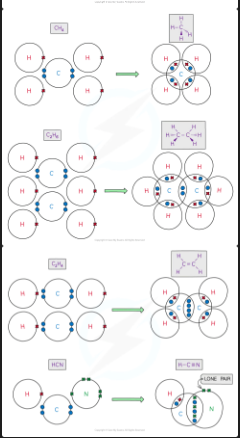
how are covalent bonds formed
when two atoms share their outer valence electrons to achieve a noble gas configuration
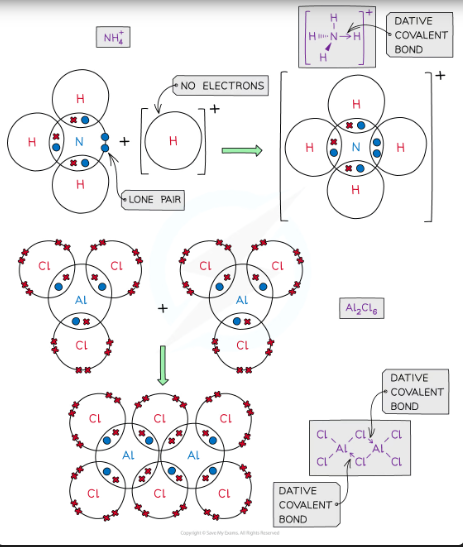
coordinative/dative covalent bonding
A covalent bond in which both shared electrons are donated by the same atom.
In a displayed formula, the dative covalent bond is represented by an arrow
The head of the arrow points away from the lone pair that forms the bond and towards the electron-deficient atom that accepts the electrons
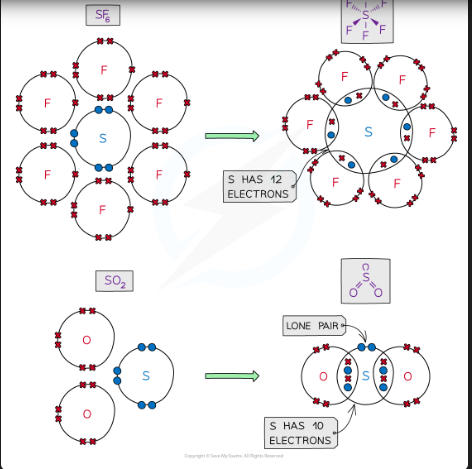
expanded octet rule
Elements in period 3 and above have the possibility of having more than eight electrons in their valence shell
This is because there is a d-subshell present which can accommodate additional pairs of electrons
This is known as the expansion of the octet
The concept explains why structures such as PCl5 and SF6 exist, which have 5 and 6 bonding pairs of electrons respectively, around the central atom
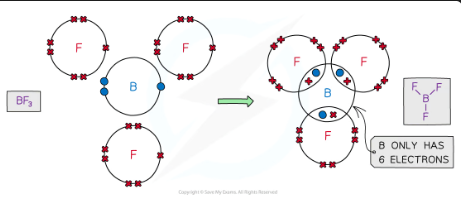
incomplete octet rule
An incomplete octet happens when an atom (in period 1 and 2) has fewer than 8 electrons in its valence shell after bonding. Some elements are stable with fewer than 8 electrons because they naturally don’t have enough valence electrons to form a full octet.
eg - 2⃣ Boron (B) → Only 3 valence electrons, so it forms 3 bonds and has only 6 electrons.
Example: BF₃ (Boron trifluoride)
3⃣ Radicals (Odd Electron Molecules) → Some molecules have an odd number of electrons, so at least one atom can’t get 8 electrons.
Example: NO (Nitrogen monoxide) → Nitrogen has only 7 electrons.