Cations I
0.0(0)
Studied by 13 peopleCard Sorting
1/38
Earn XP
Last updated 12:10 AM on 3/2/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
1
New cards
Hg2 +2
Pb +2
Ag +
Pb +2
Ag +
Group I Cations
2
New cards
Group I Cations
Group of cations whose chlorides are relatively insoluble in dilute acids.
3
New cards
1 x 10-4
Solubility product of PbCl2
4
New cards
2 x 10-18
Solubility product of Hg2Cl2
5
New cards
1\.56 x 10-10
Solubility product of AgCl
6
New cards
0\.04 F
Solubility of PbCl2
7
New cards
7\.5 x 10-7 F
Solubility of Hg2Cl2
8
New cards
1\.3 x 10-5 F
Solubility of AgCl
9
New cards
Common-Ion Effect
refers to the decrease in solubility of an ionic precipitate by the addition to the solution of a soluble compound with an ion in common with the precipitate
10
New cards
Incompletely precipitated
Due to solubility, silver chloride and mercurous chloride are almost completely precipitated; however, lead chloride is ____
11
New cards
Increase in solubility
Lead chloride undergoes an _________ in hot water
12
New cards
0\.673g/100mL
solubility of pbcl2 in water at 0 degrees Celsius
13
New cards
3\.34g/100mL
solubility of pbcl2 in water at 100 degrees Celsius
14
New cards
White Precipitate
A portion of the undissolved PbCl2 may remain as a ____ that might cause confusion in the analysis of the silver and mercurous chlorides
15
New cards
Ammonia
Silver is separated from mercurous chloride through its solubility in ____
16
New cards
Ag(NH3)2 +
Silver Ammonia Complex Ion
17
New cards
Very Large
If the amount of AgCl is ____, some of it may not be dissolved in Ammonia water
18
New cards
Ammonia Water
Serves as a medium for the auto-redox reaction of Hg2Cl2
19
New cards
Mercury
Mercuric Aminochloride
Mercuric Aminochloride
Products of the auto-redox reaction of Hg2Cl2 that are both insoluble.
20
New cards
3F HCl
Group Precipitating Agent for Cations I
21
New cards
Yellow ppt
Confirmatory Result for Lead Ion
22
New cards
K2CrO4
Confirmatory Reagent for Lead Ion
23
New cards
3F HNO3
Confirmatory Reagent for Silver Ion
24
New cards
White ppt
Confirmatory Result for Silver Ion
25
New cards
White/Gray ppt
Confirmatory Result for Mercurous Ion
26
New cards
SnCl2 Solution
Confirmatory Reagent for Mercurous Ion
27
New cards
Precipitation with dilute HCl
PbCl2 is separated by leaching solution with hot water
Separation of AgCl from Hg2Cl2 with ammonia water
PbCl2 is separated by leaching solution with hot water
Separation of AgCl from Hg2Cl2 with ammonia water
Three principal steps of the procedures of the analysis of Group I Cations
28
New cards
Oxychlorides of Sb and Bi
A white precipitate from the start may indicate the presence of group i cations and the ________
29
New cards
HCl
SbOCl and BiOCl are soluble in ___ while group i cations are not.
30
New cards
Solubilities
A high concentration of HCl is to be avoided since it increases the ___ of the precipitated chlorides through the formation of the soluble complex ions
31
New cards
Dissolve
PbCl2 may ____ appreciably during the washing process
32
New cards
Lead Chloride
Fairly soluble in hot water but reprecipitates if the solution is allowed to cool.
33
New cards
Metallic Mercury
If the ammoniacal solution is left in contact with the precipitate of Hg and HgNH2Cl, the soluble silver complex ion react with ______
34
New cards
Ag(NH3)2 +
The solution must be acid to convert ____ to AgCl
35
New cards
Colloidal Mercury
If mercurous ions are present, the residue left from the ammonia treatment must be black or greatly discolored because of the precipitation of ______.
36
New cards
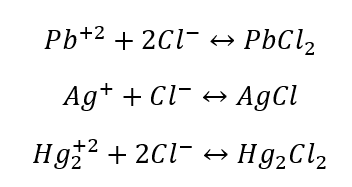
\-
Group ppt rxn
37
New cards
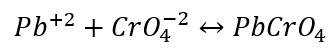
\-
Identification of Lead Ion
38
New cards
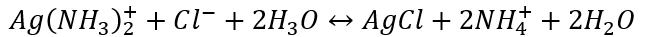
\-
Identification of Silver Ion
39
New cards
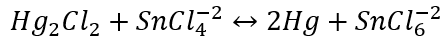
\-
Identification of Mercurous Ion