lipids
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
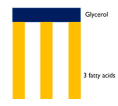
Triglyceride
Formed by the condensation of one
molecule of glycerol and three
molecules of fatty acids
forming 3 ester bonds
Fatty acid
structure
carboxyl group and a long
hydrocarbon chain
can be saturated or unsaturated
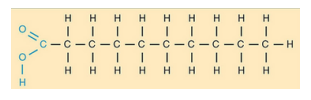
Ester bond
–COO– chemical bond
formed between glycerol and fatty
acids
Describe how an
ester bond form?
A condensation reaction between
glycerol and a fatty acid (RCOOH)
What small molecule is released when an ester bond forms?
Water
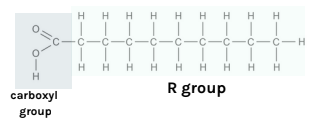
What is the R
group of a fatty
acid?
the hydrocarbon chain.
it can be saturated or unsaturated
How is the fatty acid generally represented in the specification?
As RCOOH
What functional group do fatty acids contain?
The carboxyl (–COOH) group.
What does the R in the fatty acid formula RCOOH represent?
The hydrocarbon side chain, which can vary between fatty acids
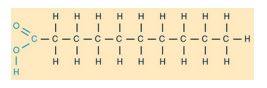
Saturated
fatty acid
a long hydrocarbon chain with a
carboxyl group at one end
only single bonds between carbon
atoms
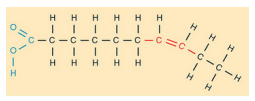
Unsaturated
fatty acid
a long hydrocarbon chain with a
carboxyl group at one end
at least one double bond between
carbon atoms
What specific bond is present in the hydrocarbon chain of an unsaturated fatty acid?
A C=C double bond.
What is the difference
between saturated and
unsaturated fatty
acid?
A saturated fatty acid has no
double bonds between carbon
atoms
whereas unsaturated fatty acids
have at least one double bond
between carbon atoms
What effect do double bonds in fatty acid chains have on the shape of the chain?
They introduce kinks, so the chain is not straight
How do double bonds in fatty acids affect melting point?
They lower the melting point, making the lipid more likely to be liquid (oils).
Why are triglycerides used as energy storage molecules?
They contain a high proportion of energy-rich C–H bonds and can be oxidised to release lots of energy.
How does the structure
of a triglyceride relate
to its function?
high ratio of C–H bonds to carbon
atoms → lots of energy released
when broken so more ATP can be generated during respiration
High hydrogen to oxygen ratio →
release water when oxidised
(metabolic water source)
Large, non-polar → insoluble in
water, doesn’t affect water potential
Low mass to energy ratio → energy-
rich storage molecule making them efficient energy stores
Why is it important that triglycerides are insoluble in water?
They do not dissolve in cytoplasm or blood plasma.
How are triglycerides arranged in cells due to their insolubility?
They form insoluble droplets or globules within cells.
Phospholipid
Formed by the condensation of one
molecule of glycerol and two
molecules of fatty acid
held by two ester bonds
a phosphate group is attached to
the glycerol

How does the structure of
a phospholipid relate to
its function?
phospholipids have two charged
regions, so they are polar
In water, they are positioned so
that the heads are exposed to
water and the tails are not.
This forms a phospholipid bilayer
which makes up the plasma
membrane around cells.
Polar molecule
A molecule that has an uneven
distribution of charge
Explain the
arrangement of
phospholipids in a cell-
surface membrane
In water, phospholipids form a
bilayer
Hydrophobic (fatty acid) tails point
away as they are repelled from
water
Hydrophilic (phosphate) heads
point to and are attracted to water
Hydrophobic
The tendency to repel and not mix
with water
e.g. tail region on a phospholipid
Hydrophilic
The ability to mix, interact or
attract water
e.g. head region on a phospholipid
Describe the
structure of a
phospholipid
bilayer
a phospholipid has:
A hydrophilic (polar) head that is
attracted to water
Two hydrophobic (non-polar)
tails that repel water
In a bilayer, phospholipids form two
layers with heads on the outside and
tails on the inside.
Plasma
membrane
phospholipid bilayer
cell surface membranes and
organelle membranes
Why is the phospholipid bilayer a useful barrier in cells?
It forms a partially permeable barrier between cell contents and the external environment.
How does the presence of a charged phosphate group affect interactions with water and ions?
The charged head can interact with water and charged particles, helping stabilise the bilayer at the water interface
How do the structural differences between triglycerides and phospholipids explain their different roles?
Triglycerides, with three hydrophobic tails, are suited to energy storage, while phospholipids, with hydrophilic heads and hydrophobic tails, are suited to forming membranes
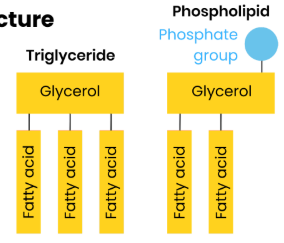
How does the
structure of a
triglyceride and
phospholipid differ?
Triglyceride: one glycerol + three fatty
acids
Phospholipid: one glycerol + two fatty
acids + one phosphate group
Triglycerides are non-polar and
hydrophobic
Phospholipids have a polar
(hydrophilic) head and non-polar
(hydrophobic) tails
Test for lipids
Add ethanol and shake
to dissolve lipids
then add water
milky white emulsion forms
What is the first step in the emulsion test for lipids?
Mix the sample with ethanol.
Why is ethanol added in the emulsion test?
Ethanol dissolves lipids, allowing them to be carried into solution
What would the result look like if no lipid is present?
The solution remains clear and colourless.
Why does a cloudy white emulsion form if lipid is present?
Lipid droplets come out of solution when the ethanol–lipid mixture is added to water and scatter light, producing the cloudy appearance
Why must the sample and ethanol be shaken thoroughly in the emulsion test?
To ensure any lipid present dissolves fully in the ethanol.
Why is water added after dissolving the sample in ethanol?
It forces the dissolved lipids to form an emulsion if present
How could you increase the reliability of the emulsion test results?
Use a control with no lipid, replicate samples, and standardise volumes and times
Exam insight: common mistakes ❌