Acids and Bases Gen Chem
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
Common strong acids
HI, HBr, HCl, HClO4, H2SO4, HNO3
Bronsted Lowry
Acids: Proton donors
Bases: Proton acceptors
Lewis
Acids: Electron acceptors (electrophiles)
Bases: Electron donors (nucleophiles), lone pairs or - charge
Amphoteric
Can act like an acid or a base H2O
Periodic table
BEARN
Basic, Acidic
Ka=
Products over reactants
Ka=[H3O+][A-]/[HA]
Ka>1 strong acids
Strong bases
Group 1 hydroxides like NaOH, group 1 oxides like Li2O
Weak bases: NH3
pH=
-log[H+]
Example for math
pH=-log(0.025)=-log(2.5 × 10^-2)
-log=exponent so pH=-(-2) this is the max minimum is max-1
Note bigger concentration closer to minimum
For bases
Get pOH range then convert to pH using pH=14-pOH
pOH=
pOH=-log[OH]
pH + pOH =
14
Pka=
Pka=-logKa
Low Pka=strong acids
Pka + pKb =
14
Weak acids
HCN—H+ and CN-
0.2 M 0M 0M
(0.2-x)M xM xM
x²/0.2-x = Ka
Math example for weak acids
x2/0.2-x = 6 × 10^-10
=x²/0.2=6 × 10^-10
x²=1.2 × 10^-10
square roots is exponent divided by power so -10/2 =-5
x=10^-5 so pH = 4-5
Acidic salt vs basic salt
Acidic salt:
Ignore anions focus on cations for acidic
Group 1 and 2 metals=neutral (Na etc.)
All else acidic
Basic salt:
Ignore cations, focus on anions for basic
Cl-, Br-, I- neutral
All else basic
HCl, HBr, HI all strong acids so conjugated bases are very weak (neutral)
Diprotic acids
Step wise 0.1 M
H2SO4 diprotic strong acids—H^+ and HSO4-
then HSO4—-H^+ and SO42-
After first H+ and HSO4- both 0.1 M but after second H+ more than and then HSO4 is less than because consumed again
Percent ionized
[H+] or [OH]/original M
if Pkb is 6 and Pka is 8 than bases dominate in thsi case
Henderson Hasselbalch equation
For acids: PH=pka + log ([conjugate base]/[weak acid])
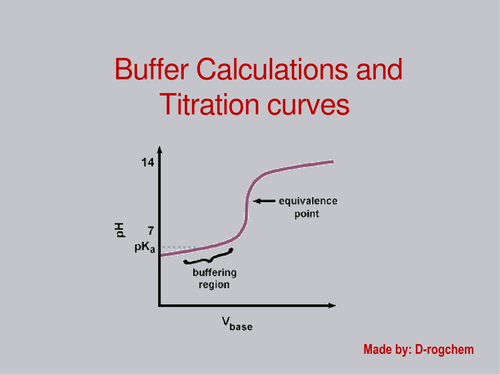
Buffers
Buffering capacity concentrations that can be absorbed, if change concentration carrying capacity does not change
PH does not change with dilution since it is based on ratios!!!
PH and pKa comparisons
if pH and pKa the same then 50 % HA and 50% A-
3.5 pH and 4.5 Pka then 90% HA and 10% A-
if pH<pka the acid form dominates (pH lower so more acidic)
if pH>pka than acid deprotonated
Titration curves
For strong acid and strong bases pH=7 at equivalence point and ½ equivalence point pH=pka
For weak acid equivalence point pH>7 strong conjugate base
For weak base pH<7 strong conjugate acid
Diprotic acids have 2 equivalents points pHequiv point=average around it