Chemistry Week 13
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Pressure
Represented by P
Caused by collisions of molecules with wall of container
Must be in atm when using the ideal gas law
Volume
Represented by V
Must be in liters when using the ideal gas law
Moles of gas
Represented by n
Must be in moles when using the ideal gas law
Temperature
Represented by T
Must be in K when using the ideal gas law
- Convert C to K by adding 273.15
The Ideal Gas Law
Used to solve for the behavior of an ideal gas, relating pressure, volume, amount of gas, and temperature
Combination of Avogadro’s Law, Charles’ Law, Amonton’s Law or Gat-Lussac’s Law, and Amonton’s Law or Gat-Lussac’s Law
Formula: PV=nRT
Ideal gas constant
Represented by R
Value: 0.08206 (L*atm)/(mol*k)
Derived from the four k constants
Initial Condision - Final Condition
Step 1: Place initial conditions equal to final conditions
Step 2: Remove any variables that remain constant
Note: A variable being constant can be implied
- Ex: Gas inside a container is heated, the volume is constant because there is no mention of the container being changed
Note: A variable being constant can be explicitly stated
- Ex: The temperature does not change

Standard temp. and pressure
0 °C or 273 K and 1 atm pressure
One mole of grass takes up roughly 22.4L of space at this temperature
Molar Volume
Volume occupied by 1 mole of a gas at STP
Formula: V = nRT/P
About 22.4 L per mole
Density
How compact something is
Formula: d = mass/V
Ideal Gas
Gas that doesn’t interact with any molecules in any way, except physically colliding with them
Normal Atmospheric Pressure
Pressure of the air at sea level
Value: 760mmHg = 760torr = 1atm
Amonton’s Law or Gat-Lussac’s Law
States that pressure (P) is directly proportional to its temperature (T)
- Assuming V and n are constant
Charles’ Law
States that volume (V) is directly proportional to its temperature (T)
- Assuming P and n are constant
Avogadro’s Law
States that volume (V) is directly proportional to the amount of moles of gas (n)
- Assuming T and P are constant
Relating Molar Mass to Density
Molar Mass from Ideal Gas Equation
Step 1: Redefine molar mass equation (M = grams/mole) as M = m/n
Step 2: Rearrange redefined equation (M = m/n) into n = m/M
Step 3: Plug rearranged formula into Ideal Gas Law (PV = nRT)
- Resulting formula: PV = (m/M)RT
Molar Mass Equation
Molar mass equation: M = grams/mole
Room mean square speed
The average kinetic energy of a molecule
Represented by variable Urms
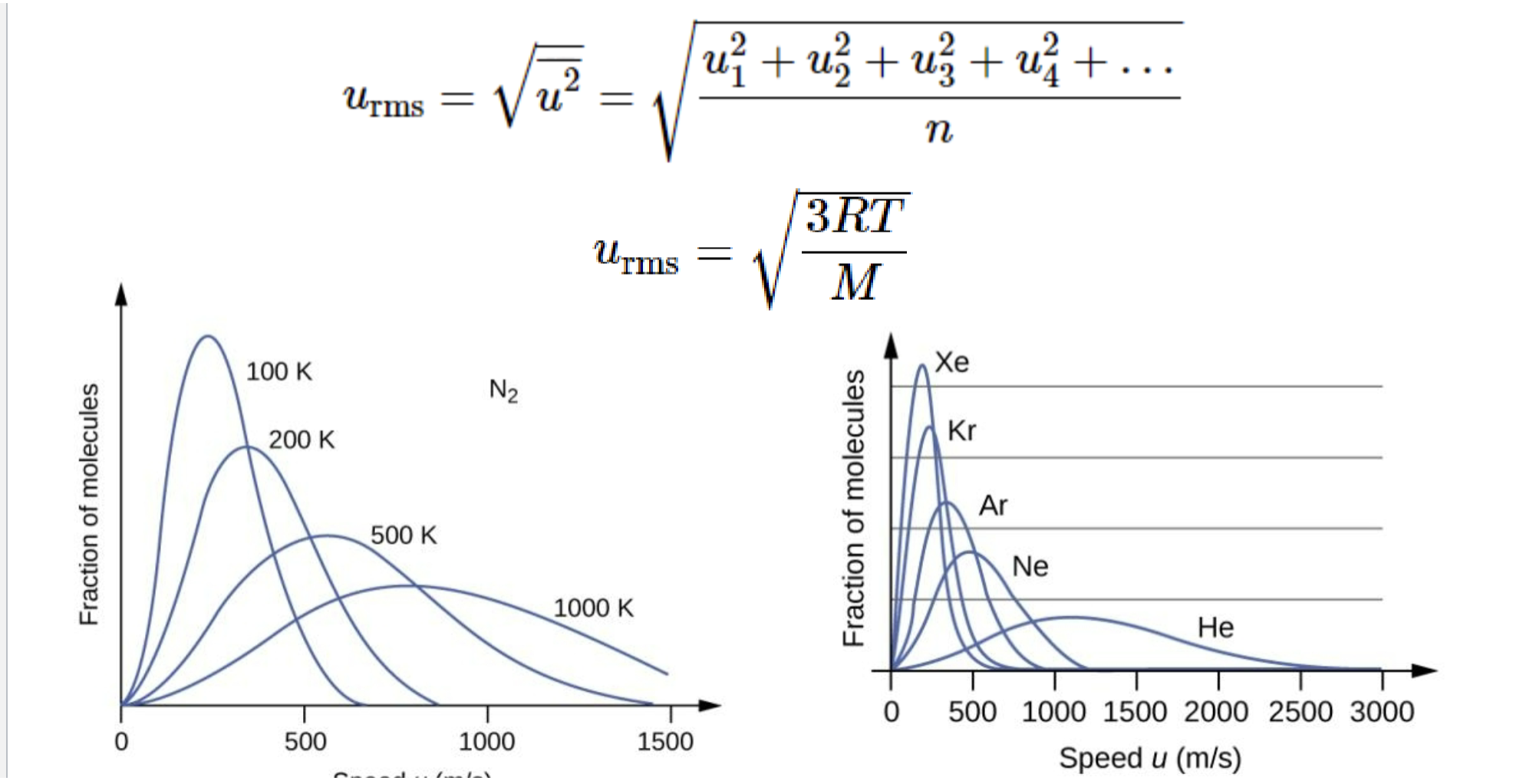
Dalton’s Law
The sum of the partial pressures of all ideal gases in a container equals the total pressure of the mixture
Diffusion
The unrestricted dispersal of molecules through space, completely random
Effusion
Diffusion through a very small opening in a physical barrier
Larger molecules have trouble passing through opening