enthalpy
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
what is enthalpy change
When chemical bonds break / form during a reaction, energy is often released
This heat energy change, which occurs at constant pressure is enthalpy (ΔH)
Measured in kJ mol-1
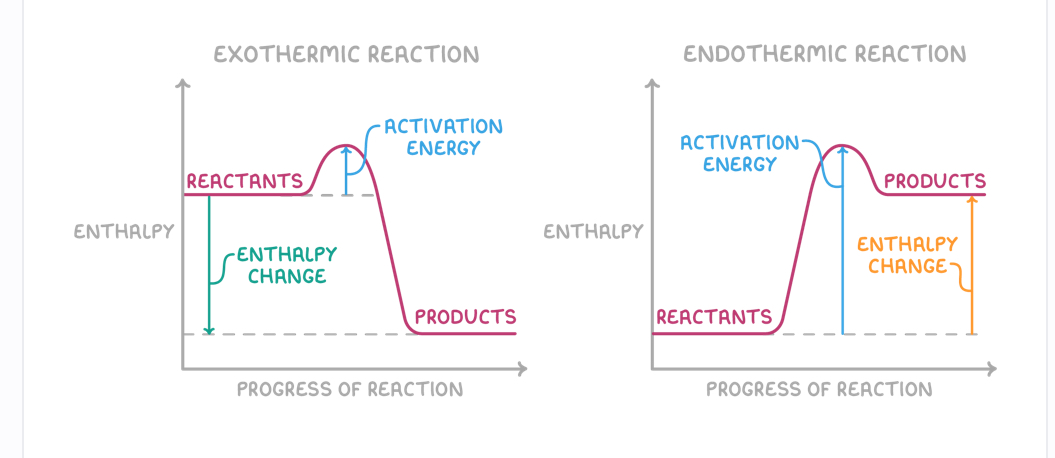
Exothermic reactions:
Release heat energy to surroundings
Negative enthalpy change
Temperature of surrounding increase
E.g combustion of methane
Endothermic reactions:
Absorb heat energy from surroundings
Have positive enthalpy change
Temperature of surroundings decrease
E.g thermal decomposition of calcium carbonate
Draw enthalpy profile diagrams of exo and endothermic reactions + key features of them
Activation energy is the minimum energy needed for a reaction to occur
Exo - reactants have higher energy than products
chants have lower energy than products
What standard conditions are used for enthalpy change
298K temperature
101kPa pressure
⦵ Used after ΔH to denote that enthalpy change refers to standard conditions
Standard enthalpy change of reaction ΔH⦵r
Enthalpy change when a reaction occurs in molar quantities shown in the chemical equation under standard conditions
Standard enthalpy change of formation ΔH⦵f
Enthalpy change when 1 mole of a compound forms from elements under standard conditions
Standard enthalpy change of combustion:
Enthalpy change when 1 mole of a substance combusts fully in oxygen under standard conditions
Standard enthalpy changes of neutralisation ΔH⦵neut
Enthalpy change when an acid and alkali neutralise under standard conditions to form 1mole of water
What are the two types of calorimetry?
We measure energy transferred in a reaction as heat using apparatus called a calorimeter, and we can deter,Ive enthalpy changes
Combustion calorimetry = used to find enthalpy change when a fuel is burned e.g combustion
Solution calorimetry = used to find enthalpy change for reactions occurring in aqueous solutions, neutralisation
Describe process of combustion calorimetry:
1, weighted fuel sample is burnt underneath a metal canister (calorimeter) containing a known mass of water
As fuel combusts, heat transferred to water, thermometer measures temperature change,
Temp change with mass and heat capacity of water used to calculate heat energy
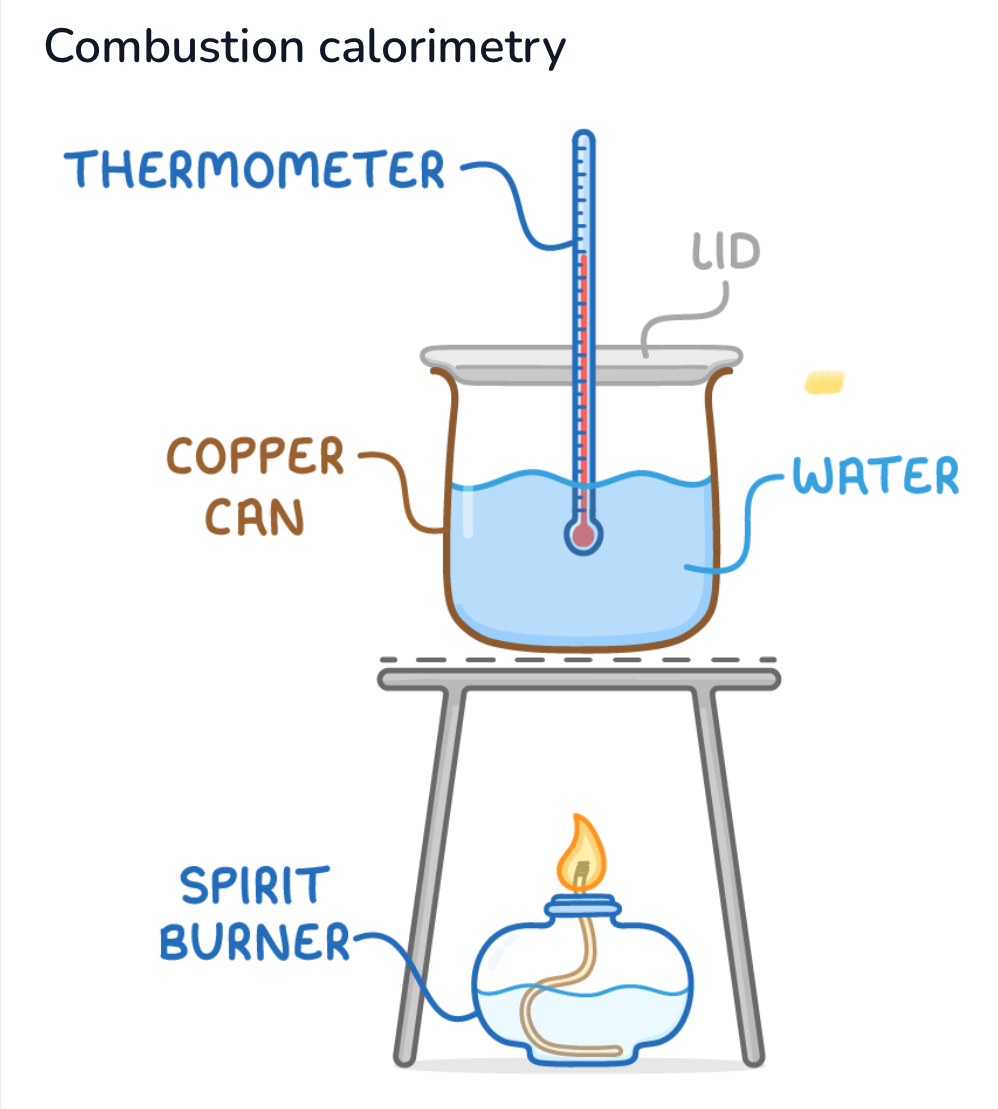
Common sources of error in calorimetry:
Heat loss = heat escaping to surrounding mean less is absorbed by water, lower enthalpy change
Fuel evaporation = if fuel evaporated before burning, reduces amount of heat generated, lower measured enthalpy change
Solution calorimetry process
Enthalpy changes for reactions that occur in aqueous solutions = neutralisation, dissolution, displacement
Measured volume of reactant to polystyrene cup calorimeter and record initial temp
Add measured volume of second reactant and seal container w lid
Stir moisture with thermometer and recor temp at intervals as heat is released / absorbed
why are polystyrene cups used as calorimeter for solution calorimetry?
Because it is a better insulation, lightweight, waterpolo
Reduce heat loss to surrounding
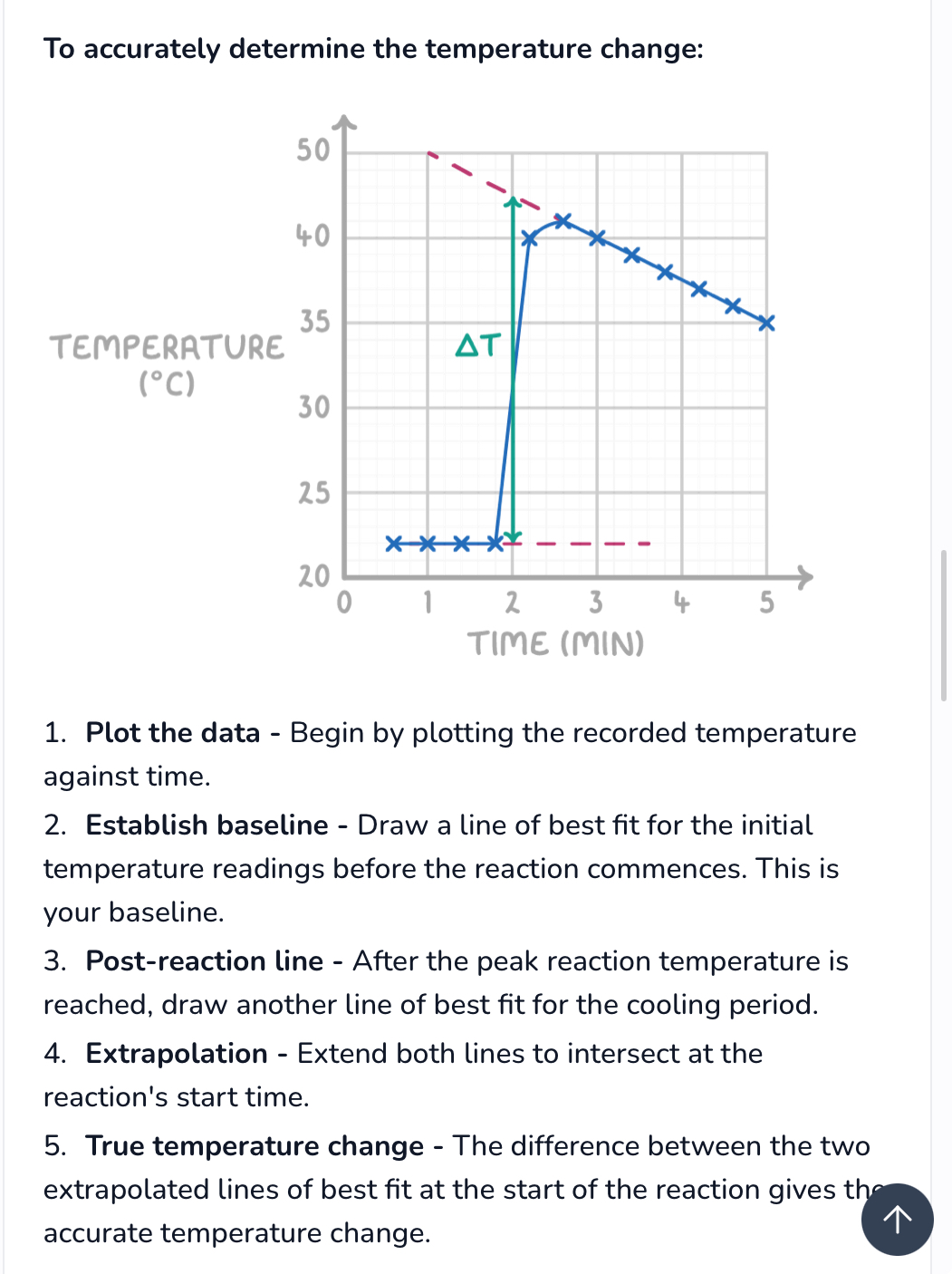
How to create accurate temperature time graph
Equation to calculate enthalpy change:
First calculate heat energy change, then use that to calculate enthalpy change per mole
Heat energy change: q=mcΔT
Q = heat energy (j)
M = mass of water (g)
C = specific heat capacity of water
ΔT = temperature change
Use value of q:
ΔH= -q/n
ΔH = enthalpy change kJ mol-1
q = heat energy (kJ)
n = number of moles of the limiting reactant (mol)
What is the overall enthalpy change of a reaction?
Combined effect of these two processes:
Breaking bonds in the reactant molecule = endothermic process that requires energy, breaking stronger binds requires more energy
Forming bonds to create the product molecules = an exothermic process that releases energy, formation of stronger bind releases more energy
when does a reaction have a positive / negative enthalpy change?
Bond breaking requires more energy than released, has positive enthalpy change e
If bond breaking requires less energy than is released during bond formation p, reaction has a negative enthalpy change
What is average bind enthalpy?
Average energy needed to break one mole a type of covalent bond in gaseous molecules
They are always positive (endo) because breaking bonds required energy
Represent average value across different molecule environments
How to calculate reaction enthalpy changes ΔHr?
ΔHr = Σ(bond enthalpies of bonds broken) Σ(bond enthalpies of bonds formed)
where Σ = sum of
or
ΔHr = Σ(bond enthalpies in reactants) Σ(bond enthalpies in products)
Why is Hess cycle more accurate than ΔHr values?
Calculating reaction enthalpy change with mean bond enthalpy is less accurate than Hess cycle because average bond enthalpjes don’t account for unique molecule environment of each bond
Hess is more precise
What Hess law states
Overal standard enthalpy change of a reaction. Is same, regardless of whether the reaction takes place in one stop or several steps
Allows you to break a reaction into component steps and add up their enthalpy changes to find total enthalpy change for reaction
How can standard enthalipes of formation be used with Hess law to determine unknown ΔH⦵ Values
These values are known for many compounds
standard enthalpy of formation for elements in their standard state is define as 0kj mol
To calculate = ΔH⦵r = ΣΔH⦵f(products) - ΣΔH⦵f(reactants)
where Σ = sum of
Using enthalpy of combustion to find enthalpy of reaction values
ΔH⦵r = ΣΔH⦵c(reactants) ΣΔH⦵c(products)
where Σ = sum of
How to form the Hess cycle for enthalpy of formation
Reacts → products
Elements at bottom
Arrow point up from elements
How to form Hess cycle for enthalpy of combustion
Reactant → products
Combustion products at bottom
Point towards combustion products