Chemistry - Test 1
1/87
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
88 Terms
Accuracy
How close numbers are to a target.
Precision
How close values are to each other.
How do you read a graduated cylinder?
Read at eye level and read the meniscus.
Certain Digits
What you can read
Uncertain Digits
The last digit, an estimate.
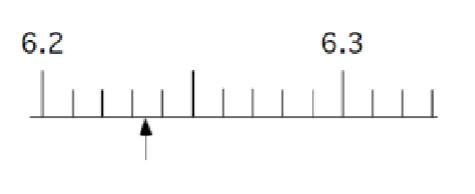
What is this measurement?
6.235 cm (5 is the estimate/uncertainty)
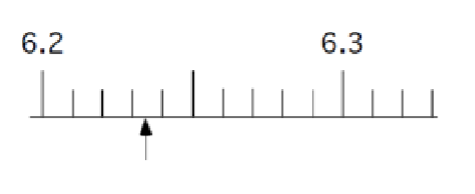
What are sig figs?
All certain digits + one uncertain digit.
What are the sig fig rules?
Any non-zero number is significant
Sandwiched zeros are significant.
Zeroes to the left of the first significant number are not significant.
Zeroes at the end are only significant after a zero and a decimal.
Give sig figs for each number:
13476
1087
0.78
.00657
43.0
67.00
1900
6000
0150.0
1500.0
5
4
2
3
3
4
2
1
4
5
What rules do you following when adding and subtracting decimal numbers?
Add them all up then round to the least amount of decimals based on the added/subtracted numbers.
Add these numbers:
32.567 + 135.0 + 1.4567
169.0237 → 169.0
What is the rule for multiplying and dividing decimals?
Go off of the lowest number of sig figs
Multiply these:
23.7 × 3.8
43.678 × 64.1
90.06 → 90.
2799.7598 → 2.80 × 10³
What is scientific notation?
A shorthand to express large or small numbers.
What is the scientific notation format?
This
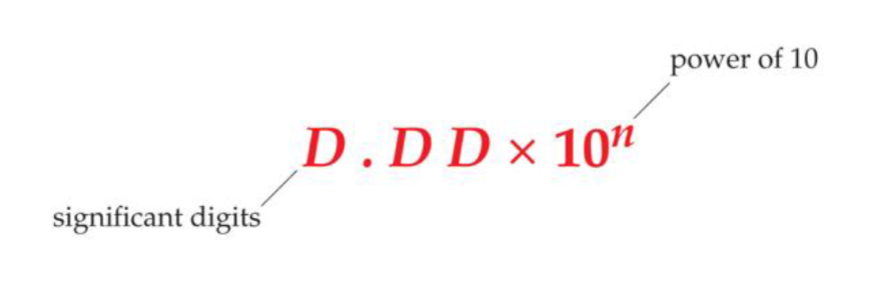
Write these in scientific notation:
142.63
1,500,000
0.00336
1.4263 × 10²
1.5 × 106
3.36 × 10-3
What are the base units for length, mass, and volume?
Meter, grams, liter.
What is a difference between metric and english units?
Metric units are power 10 based. English units are definition based.
When changing units using the metric system, which should have the one and which should have the power?
The prefix should have one and the base should have the power.
Change 2.6 mg to g
2.6 × 10^-3 g
Convert 7.00 in to cm
17.78 = 17.8 cm
What type of property is density?
A physical property of matter and an intensive property.
What is density?
The amount of mass in a specific volume
Density equation with units?
D = M/V
Mass: Kg, g
Volume: cm³ , mL, L
1 mL = ?
1 cm³
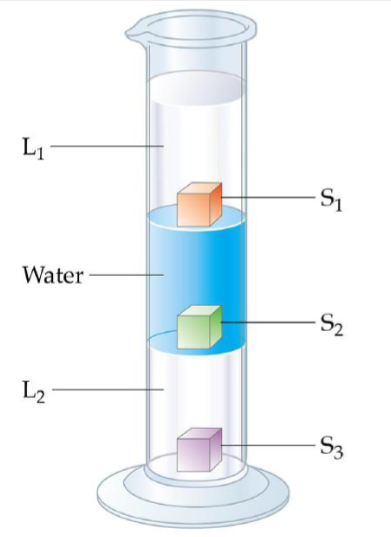
What happens to the most dense?
Rank the liquids from most to least dense
Rank the cubs from most to least dense
It will sink.
L2 > H2O > L1
S3 > S2 > S1
H2O = S2
What is an intensive property?
It does not depend on the amount of material
Why is the volume displacement method used?
To measure density/volume of irregularly shaped objects.
What two things must be followed to make sure volume displacement method works?
The object cannot float and it cannot dissolve in water.
How do you convert C to F?
F = (9/5)C + 32
How do you convert F to C?
C = 5/9(F-32)
How to convert C to K?
K = C +273
How to convert K to C?
C = K - 273
A bracelet has a mass of 50.901 g. When the bracelet is placed in a graduated cylinder containing water, the water level rose from 15.5 mL to 20.3 mL. What is the density of the bracelet? Is it silver or platinum? (d of silver = 10.5 g/mL) (d of plat = 21.45 g/mL)
10.6 or 11 g/mL. Silver.
What is chemistry?
The study of matter and its interactions
What is matter?
Anything with mass and volume (density).
Matter has density. It must have both.
What is a substance?
A type of matter with specific composition and properties.
Ex) Water, air, carbon dioxide.
What is mass?
How much matter an object has. It is constant.
What is weight?
Attraction for a body. It varies.
Gravity. Greater pull = greater weight
What does definite mean?
Does not change
Is shape definite or indefinite for solids, liquids, and gases?
Solids: Definite
Liquid: Indefinite
Gas: Indefinite
Is volume definite or indefinite for solids, liquids, and gases?
Solids: Definite
Liquids: Definite
Gases: Indefinite (They can be compressed)
Rank the states of matter from lowest to highest energy
Lowest: Solid
Higher: Liquid
Highest: Gas
Rank the density of the states of matter from highest to lowest
Highest - Solid
Lower - Liquid
Lowest - Gas
Rank the speed of particles of each state of matter from lowest to highest
Lowest (only vibrates) : Solids
Faster : Liquids
Fastest : Gases
What are the solid → liquid and liquid → solid changes called?
Solid → liquid: Melting
Liquid→ Solid: Freezing
What are liquid → gas and gas → liquid called?
liquid → gas: Evaporating
Gas → liquid: Condensing
What are solid → gas and gas → solid called?
Solid → gas: Sublimation
Gas → Solid: Deposition
When is deposition used?
Coat a metal with a metal. Typically in lab settings.
What are physical properties?
Describe the characteristics of a substance.
Ex) Boiling pt, color, shape, size, shape
What are chemical properties?
Describe change in properties or resistance to change.
Ex) Reactivity, oxidation, pH

Answer these: (Physical or chemical)
Physical
Chemical
Chemical
Physical
Chemical (corrosion)
Physical
Chemical
Chemical
Physical
Physical changes of matter?
Do not alter the properties or identity
Ex) Ice → water
Chemical changes of matter?
Affect the identity.
Ex) Water hydrolyzed (decompose)
-Get something new
-Usually chemical reactions
What are pure substances?
One type of substance
Have fixed composition
Cannot be separated by physical processes.
What are elements?
Elements are pure substances
They cannot be separated into simpler substances by chemical means
One type of atom
Ex) copper, aluminum
What are compounds?
A type of pure substance
They can be separated into simpler substances by chemical means
Combined atoms
Ex) Salt (NaCl) and Water (H2O)
What are mixtures?
Two or more types of substances
Variable compositions
Can be separated by physical processes
What are homogenous mixtures?
Have a uniform composition
They look the same, you only see one thing
Ex) Salt water (NaCl, H2O), brass (Cu , Zn)
What are heterogenous mixtures?
Nonuniform composition
See what is what
Ex) Pizza, water and sand
What are all compounds by definition?
Homogenous

Do this
Heterogenous mixture
Homogenous mixture
Homogenous mixture
Heterogenous mixture
Heterogenous mixture
Pure element
Heterogenous mixture
Pure element
Compound
Homogenous
Pure element
What did Democritus do?
The atom is indivisible called “atomos”
What did Dalton come up with? What are the four statements?
Atomic theory
Matter is composed of particles called atoms
Atoms of the same element have the same properties
Combining atoms makes a compound with different properties
Atoms cannot be created or destroyed (No longer valid because of nuclear)
What did J.J. Thompson do?
Cathode ray tube, discovered electrons.
Used a cathode ray where a negative electron beam was attracted to a positive plate
Came up with the plum model
What did Rutherford do?
Gold foil experiment
Go through → atom is mostly empty space
Bounce back → Dense center
Deflected → Positive center
Discovered nucleus/positive particle (proton).
What are the particle charges of protons, neutrons, and electrons.
Proton (+)
Neutron (o)
Electron (-)
What is the relative mass of the three particles?
Proton (1 amu)
Neutron (1 amu)
Electron (0 amu)
What is the location of the three particles?
Proton: Nucleus
Neutron: Nucleus
Electron: Outside
How do you calculate protons?
The atomic number
How do you calculate neutrons?
Mass number - protons = neutrons
How do you calculate electrons?
Electrons = protons in neutral atoms
What is important about atomic number?
It is unique to each atom and gives the identity
What is the mass number ?
Protons + neutrons.
Average rounded
Nuclear charge is
how many protons are in nucleus
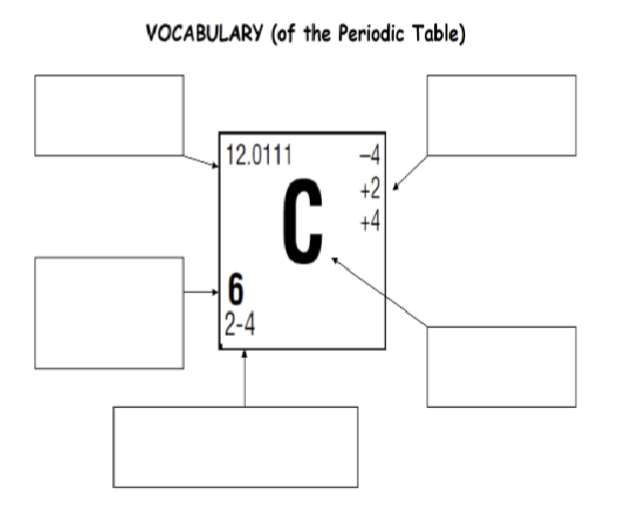
Do this
Top left: Atomic mass
Below top left: Atomic number
Below that: Electron cofiguration
Top right: Oxidation states
Bottom right: Symbol
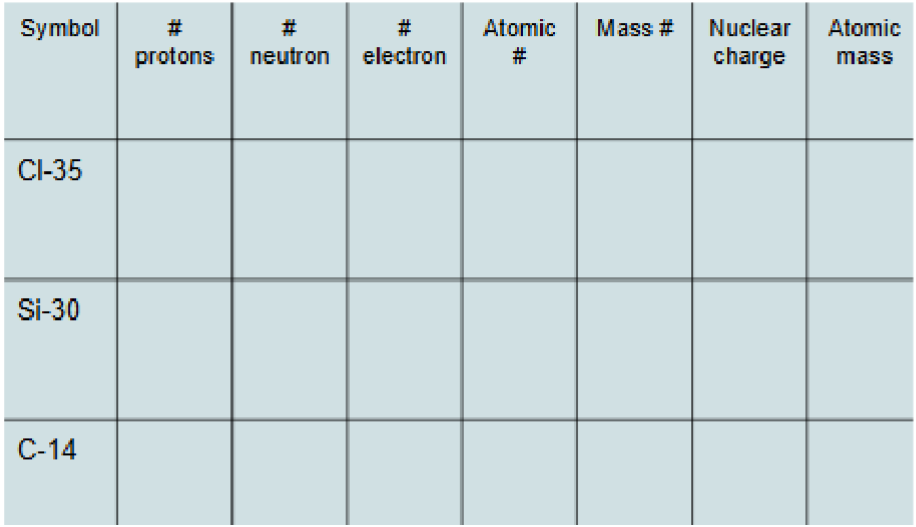
Do this
Cl-35: 17 18 17 17 35 +17 35.456
Si-30: 14 16 14 14 30 +14 28.085
C-14: 6 8 6 6 14 +6 12.011
What are ions?
Atoms with electron charge
What is a neutral atom?
When protons = electrons. It has no charge. Na, Cl, C
What is a negative atom?
Anion. number of electrons > number of protons
Ex) Cl^- and O^-2
What is a positive atom?
Cations
Number of electrons < number of protons
Ex) Na^+ and Mg^+2
For Cl^- how many protons, neutrons, and electrons?
Protons: 17
Electrons: 18
Neutrons: 18
For Mg^+2 how many protons, neutrons, and electrons?
Protons: 12
Electrons: 10
Neutrons: 12
What are isotopes?
Atoms of the same element with different numbers of neutrons. (Diff mass numbers)
What are the two representations of isotopes?
Mass number on top
Atomic number on bottom
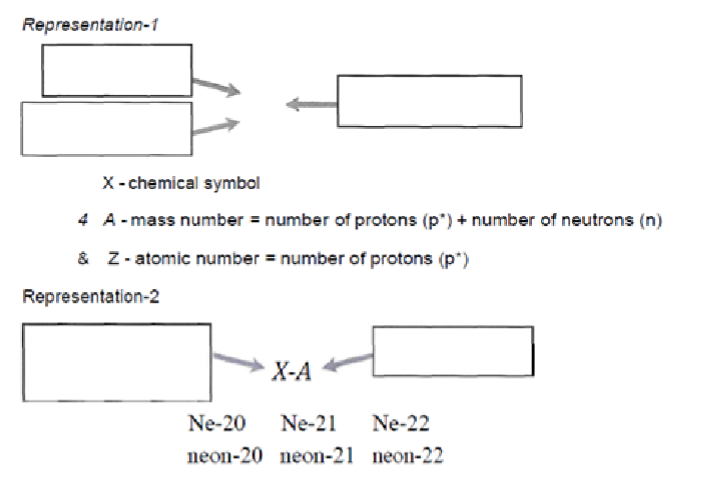
What is weighted atomic mass?
Average mass of isotopes that compose that element weighted based on natural abundance.
What is the weighted atomic mass formula?
Average atomic mass = (fractional abundance) x (isotopic mass)
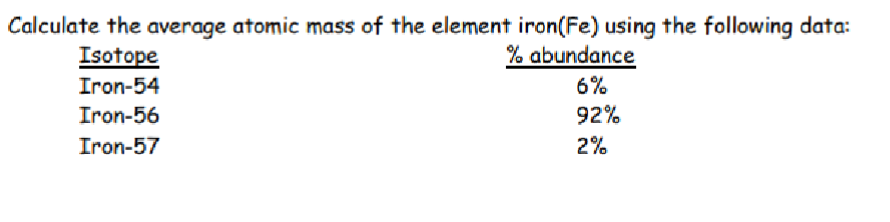
This
55.9 amu