Forensic Toxicology Final
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
90 Terms
An illicit drug user taking opioids requires higher doses to reach the same pain relief after a period of usage. This phenomenon is known as?
Tolerance
What items must be present on the Chain of Custody forms according to ASTM guidelines?
Signatures of sampling personnel and signatures of all personnel handling/receiving samples, Preservation of sample with amount and type, Field notes, and Sampling location ID, date, and time interval
What biological specimen type has the shortest detection window for analysis post storage in preservative?
Breath
Which of the following factors influences the LD50 value of a substance?
Species of the test animal, route of administration, age of the test animal, sex of the test animal
Who is referred to as the "father of toxicology" for his contributions to the field?
Paracelsus
What is the best sample preparation technique for this diazepam
Liquid-Liquid extraction using chloroform (CHCl3) as the solvent
What is the most common method of toxicant absorption
Oral ingestion
10 mg of heroin and 10 mg of benzos were taken together resulting in death. Doses of 30 mg of heroin do not induce death and doses of 20 mg of benzos do not induce death. What effect cause increased sedation and respiratory depression that resulted in fatality?
Synergism
A drug with a high volume of distribution is most likely to be?
Lipophilic and widely distributed in tissues
List each area present in the pharmacokinetics acronym ADME
Absorption, distribution, metabolism, elimination
Buprenorphine has a Kd of 0.18 nM for the u-opioid receptor. Naloxone has a Kd of 1 nM. Both work to reduce dependency on opioids through receptor interaction. Determine Ka values of both substance and suggest which interacts with the u-opiod receptor with higher affinity
buprenorphine: 5.6nM^-1
naloxone: 1 nM^-1
buprenorphine has the highest affinity
Consider the following information about drug X: The dose is 150 mg, the absorptivity is 35% the volume of distribution is 25 L and the half-life is 24 hours with first order kinetics. What is the rate constant?
0.0289 hr^-1
LSD LD50 is approximately 0.1 mg/kg and marijuana Ld50 is 9000 mg/kg. Classify these drugs as with highly toxicity, moderate toxicity or low toxicity. Calculate lethal doses (in mg) for a person that weighs 168 pounds (1 pound=0.454 kg)
LSD : high toxicity, 7.63 mg
marijuana : low toxicity, 686448 mg
Beer-Lambert Law in absorption spectroscopy fails at?
High concentrations of absorbing molecules
GHB may not be detectable by GC analysis due to the removal of carboxylic acid with higher temperatures (decarboxylation). To measure these compounds by GC, what technique might be employed.
Derivatizing the structure by adding different functional groups for detection
An antibody developed for an immunoassay test exhibited one binding constant for the analyte tested Ka-10^8. One might classify this antibody type as?
monocolonal
Chromatographic separations of a mixture are based on?
the interaction of the components with both stationary and mobile phases
What is the primary role of ELISA in forensic toxicology
Screening samples for the presence of drugs or their metabolites
What is the primary purpose of a confirmatory test in forensic toxicology?
To eliminate false positives from initial screening test
A main advantage of using HPLC with UV-Vis detection over GC for thermally labile compound detection is
HPLC analyzes compounds that are nonvolatile and/or are unstable as gases
ESI and MALDI belong to which of the following techniques
Ionization
Alcohol is classified as
Depressant
Medication-assisted treatment (MAT) is approved by the FOOD and Drug Administration for opioids. What is a MAT antagonist?
naltrexone
What is a key step in sample preparation required for GC-MC analysis of amphetamine-like compounds
derivatization
Which of the following drugs act on the GABA A receptors in the brain
diazepam
Which is not an effect of stimulants
sleep
delta-THC is befound in the greatest concentrations in which part of cannabis sativa
plant seed and oil extracts
what is the primary enzyme involved in the metabolism of alcohol in the liver
alcohol dehydrogenase
what does not play a role in the blood alcohol content (BAC)
time of day
Log P values of several opioids are given below . Based on this data, which compound would be the most potent opioid?
Morphine : 0.93
Codeine : 1.20
Hydrocodone: 1.34
Nalorphine : 1.87
oxycodone : 2.03
Amphetamine interacts primarily with which receptors?
Dopamine neurotransmitters
Describe the 3 types of cannabinoids
phytocannabinoids (plant THC, CBD), endocannabinoids, synthetic cannabinoids (man made)
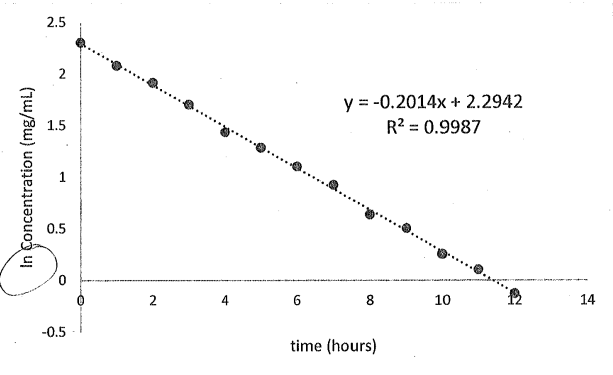
What is the reaction order, rate constant, half-life, and volume of distribution for this plot. The total clearance is 1.25 L/hour
Reaction order : 1st
rate constant:0.02014 hr^-1
half-life:3.44 hrs
Volume distribution : 6.2 L
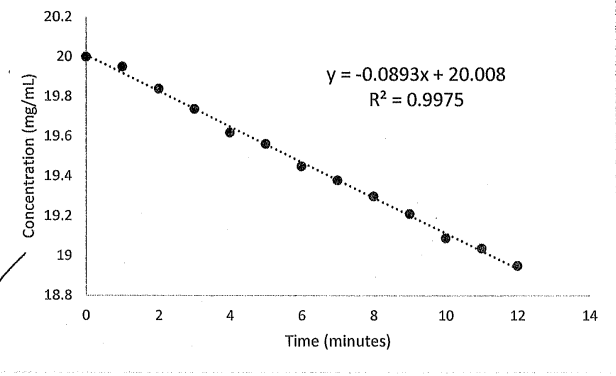
What is the reaction order, rate constant, half-life, and volume of distribution for this plot. The total clearance is 1.25 L/hour
Reaction order : 0th
rate constant: 0.0893 mg/mL*min
half-life: 112 min
Volume distribution : 13.9 L
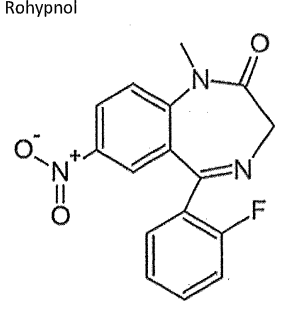
Consider the structure of methadone (Rohypnol) 1) does it have a high number of potential hydrogen bond donors and acceptors? 2) does it have a high tissue distribution 3) does it have a long half-life 4)is it more likely to be eliminated in urine?
1) yes it has 4 HB and HA
2) yes
3) yes
4) No
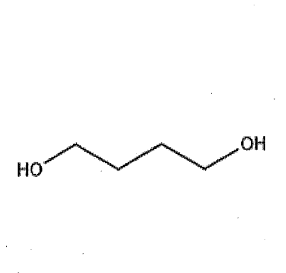
Consider the structure of 1,4-butanediol 1) does it have a high number of potential hydrogen bond donors and acceptors? 2) does it have a high tissue distribution 3) does it have a long half-life 4)is it more likely to be eliminated in urine?
1) No
2) No
3) No
4) Yes
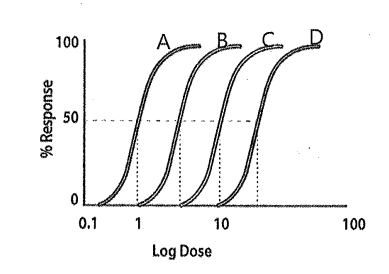
which of the following curves illustrates the lowest potency?
Curve D
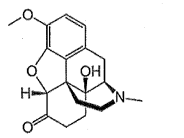
what is the drug class and schedule (if applicable) for this structure?
Opiates and schedule II
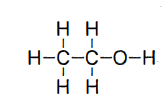
what is the drug class and schedule (if applicable) for this structure?
alcohol and n/a
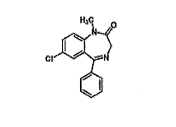
what is the drug class and schedule (if applicable) for this structure?
benzodiazepines and schedule IV
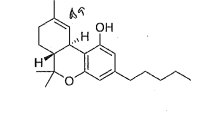
what is the drug class and schedule (if applicable) for this structure?
cannabanoids and schedule I
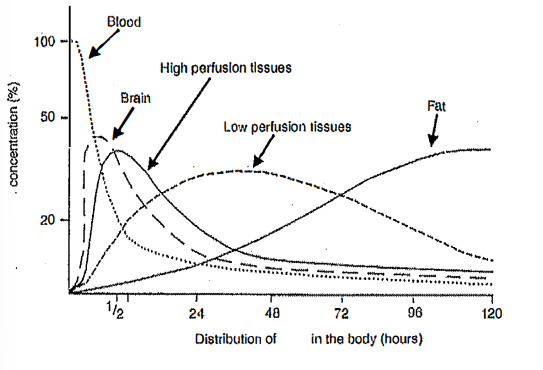
describe the pharmacokinetic terms of half-life, volume of distribution, and bioavailability
1) most of the presence for the longest time is in the tissue and fat
2) short-lived in blood or tissues
3) high volume of distribution
3) bioavailability is high as concentration in blood is high
Pharmacokinetics
the study of the time course of drugs in the body
Bioavailability
amount of available substance in the body as a ratio of the amount taken. It is dependent upon many factors including administration method.
Lipophilic drugs crosses the membrane more?
easily
Distribution pathway depends on?
Ability of a drug to move from absorption site to blood
Volume distribution increases when?
There is more distribution to the tissues and lower in blood
Volume distribution decreases when?
there is less distribution to the tissues and higher blood.
Plasma Clearance
Defined as the removal rate of drugs from plasma
Hepatic Excretion
Clearance by the liver in bile.
Carrier protein
transport of ions and small organic molecules across cell membranes that are not sufficiently lipid soluble
Ion Channel
Gateways in cell membranes
Affinity
refers to the strength of binding between a drug and a recptor
Agonist
drug that binds to receptors and initiates a cellular response and has affinity
Antagonist
drug that binds to receptors but cannot initiate a cellular response but prevent agonist from producing a response
Amniotic Fluid
useful for prenatal drug exposure
Meconium
Long-term window of drug exposure, non-invasive, demands sample prep.
Digestion prep technique
removal of organic matter (ex wet digestion, dry, and microwave assisted decompostion)
Filtration
separation of solid from a liquid
Protein precipitation
removal of protein from sample, protein binding interferes with detection
LLE
involves analyte partition between the aqueous phase and organic solvent
Solid Phase Extractions
based on the principle of separation based on affinity consisting of liquid solid separation
Immunoassay
an analytical technique that uses naturally occurring reagents knwon as antibodies to selectively determine sample compnents
are immunoassays expensive or inexpensive
inexpensive
Forensic Immunoassays pros
screening purposes only, rapid and efficient, and covers a broad range of analytes, high sensitivity and specificity
monoclonal
produced by a single cell line, binding w/ the same affinity, expensive
polyclonal
different lines of antibody-producing cells and binding with a range of affinities
ELISA testing
enzyme linked immunosorbent assay
EMIT testing
enzyme multiplied immunoassay technique
ELISA advantages
high sensitivity, less subject to matrix effects, possibility of automation, shelf life is long
ELISA limitations
cost per sample is usually elevates, it cannot be used in samples stored with sodium azide.
EMIT advantages
easy to automate, shelf life long, labelled drug can be measured without separation from free drug, absorbance change as function of time
sensitivity refers to ?
assays ability to detect low concentrations of the analyte
specificity refers to ?
the assays ability to which it can distinguish between different analytes in a solution
sensitivity formula
TP/TP+FN
Specificity formula
TN/TN+FP
Separation is based on?
sample components residing in stationary and mobile phases
Advantages of Gas Chromatography
Good for volatile samples, can be easily automated for injection and data analysis, compounds that are not decomposed at their vaporization temperature
Gas chromatography gas selection depends on?
Column type and detector
GC column type He
high cost, safe to use, wide optimum linear velocity range
GC column type N2
peak broadening in the column, cheaper, safe, low optimum linear velocity and analysis takes more time.
GC column type H2
alternative to He
GC Detectors- Flame Ionizing Detector (FID)
sample leaving the column exist through H2 air flame burning at the jet tip. They’re ionized
GC Detectors- Electron Capture Detector (ECD)
beta particles ionization w/ low electrical resistance and high current between electrodes, compounds with halogens, CN or NO2 groups are well analyzed by GC-ECD
Liquid Chromatography
Liquid
Mobile phase is liquid
Packed chromatographic column
More expensive than GC'
No limitation with the properties of analyte
Mass Spectrometry Analysis
Measures an analyte that has been converted into an ion in the gas phase
GCMS and LCMS what’s different
it is converted into an ion during its gas phase
Electron Ionization
analytes are exposed to beams and loses a electron
Positive chemical ionization
soft ionization technique that produces less fragmentation in the mass spec.
Derivatization increases?
the number and spectrum complexity