2.6: Shapes of Molecules + IM Forces
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What does the shape of a molecule depend on?
Number of electron pairs in the outer shell, and the Number of these electrons which are bonded and lone pairs
What is the shape, diagram and bond angle in a molecule with 2 bonded pairs and 0 lone pairs?
Linear
180°
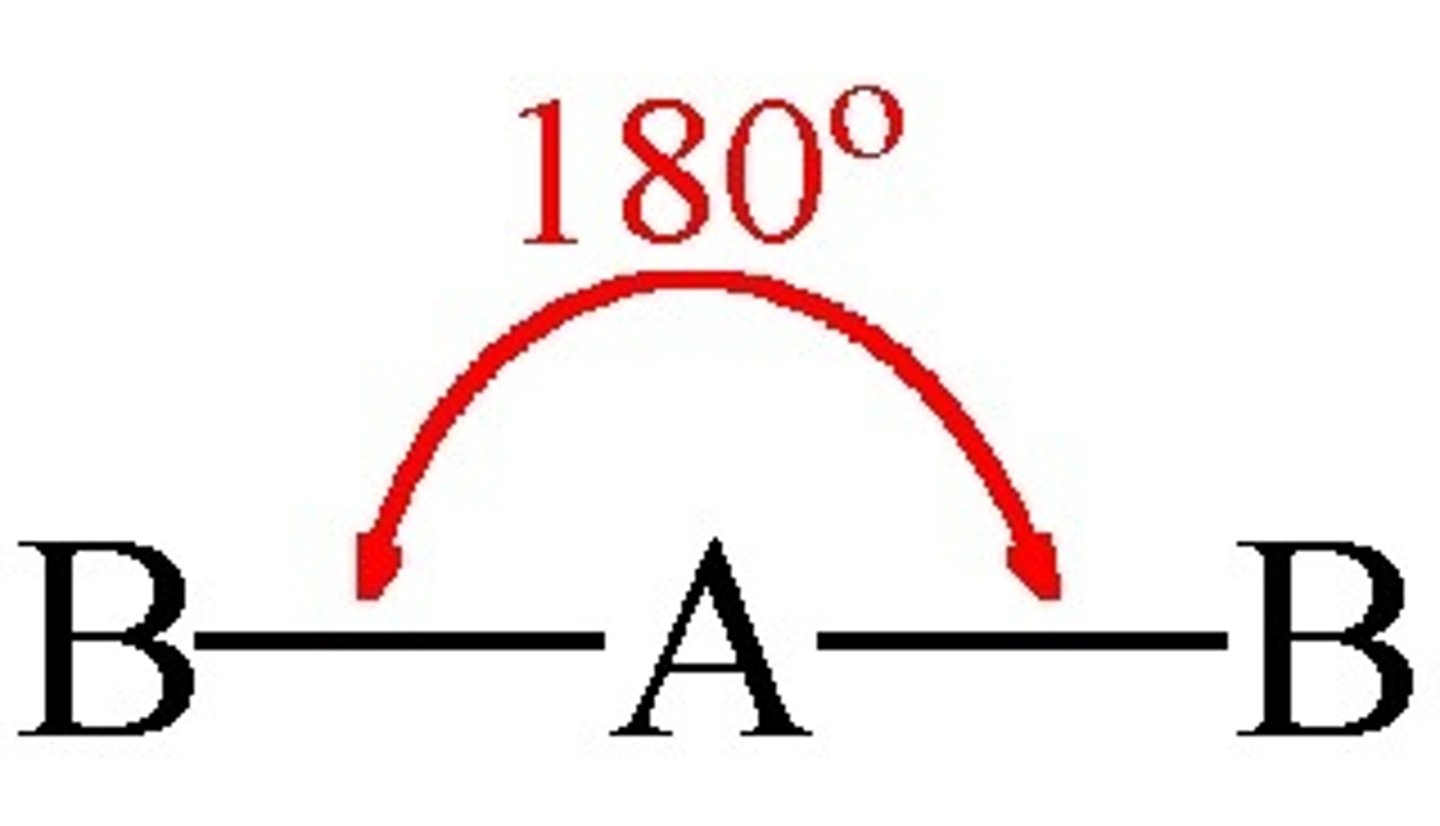
What is the shape, diagram and bond angle in a molecule with 3 bonding pairs and 0 lone pairs?
Trigonal planar
120°
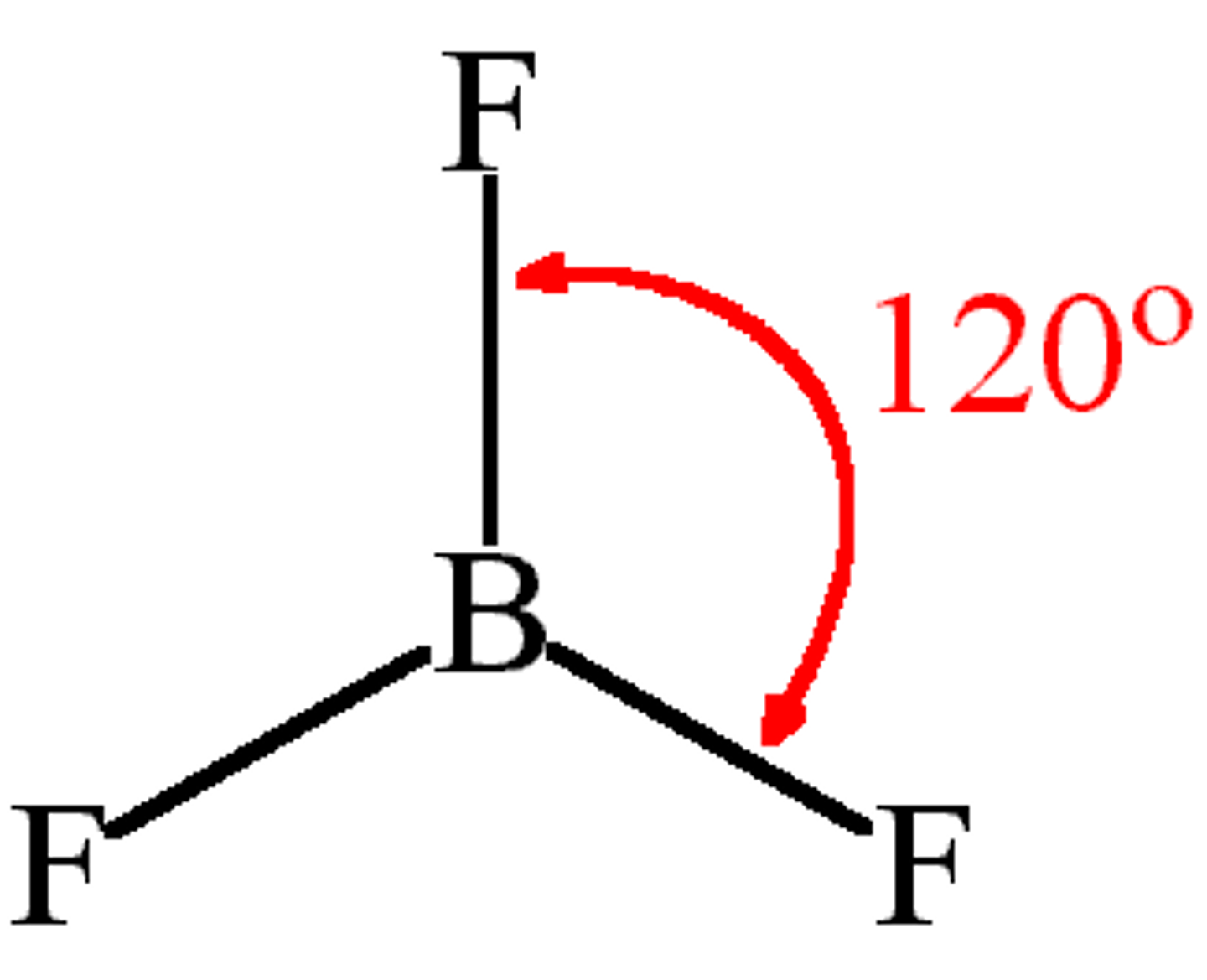
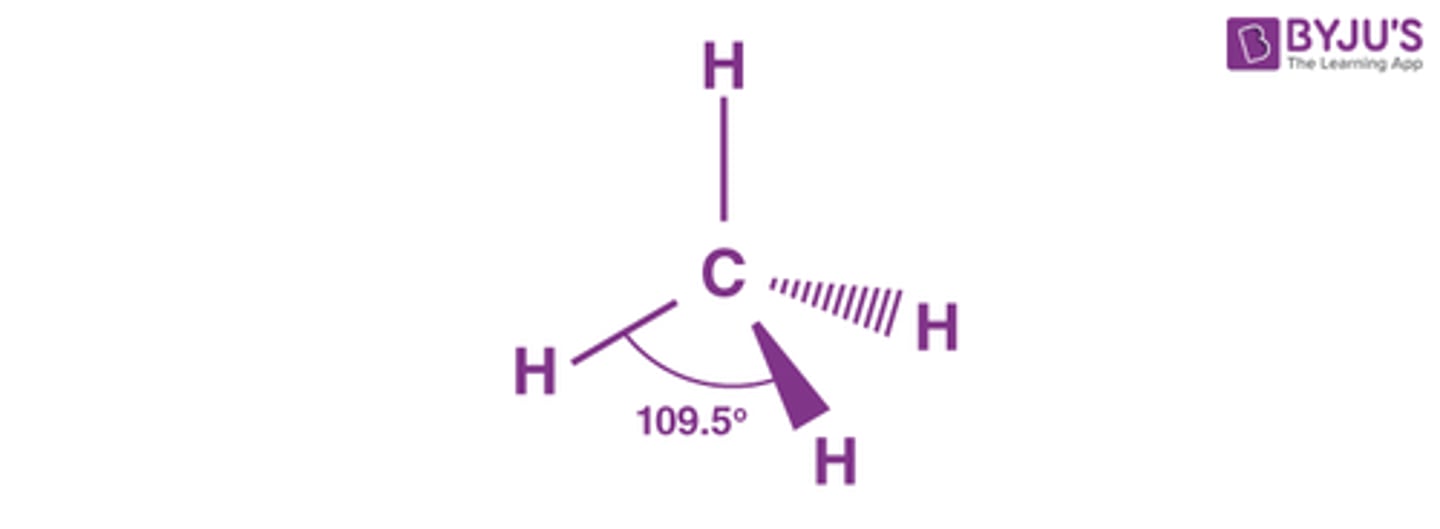
What is the shape, diagram and bond angle in a molecule with 4 bonded pairs and 0 lone pairs?
Tetrahedral
109.5°
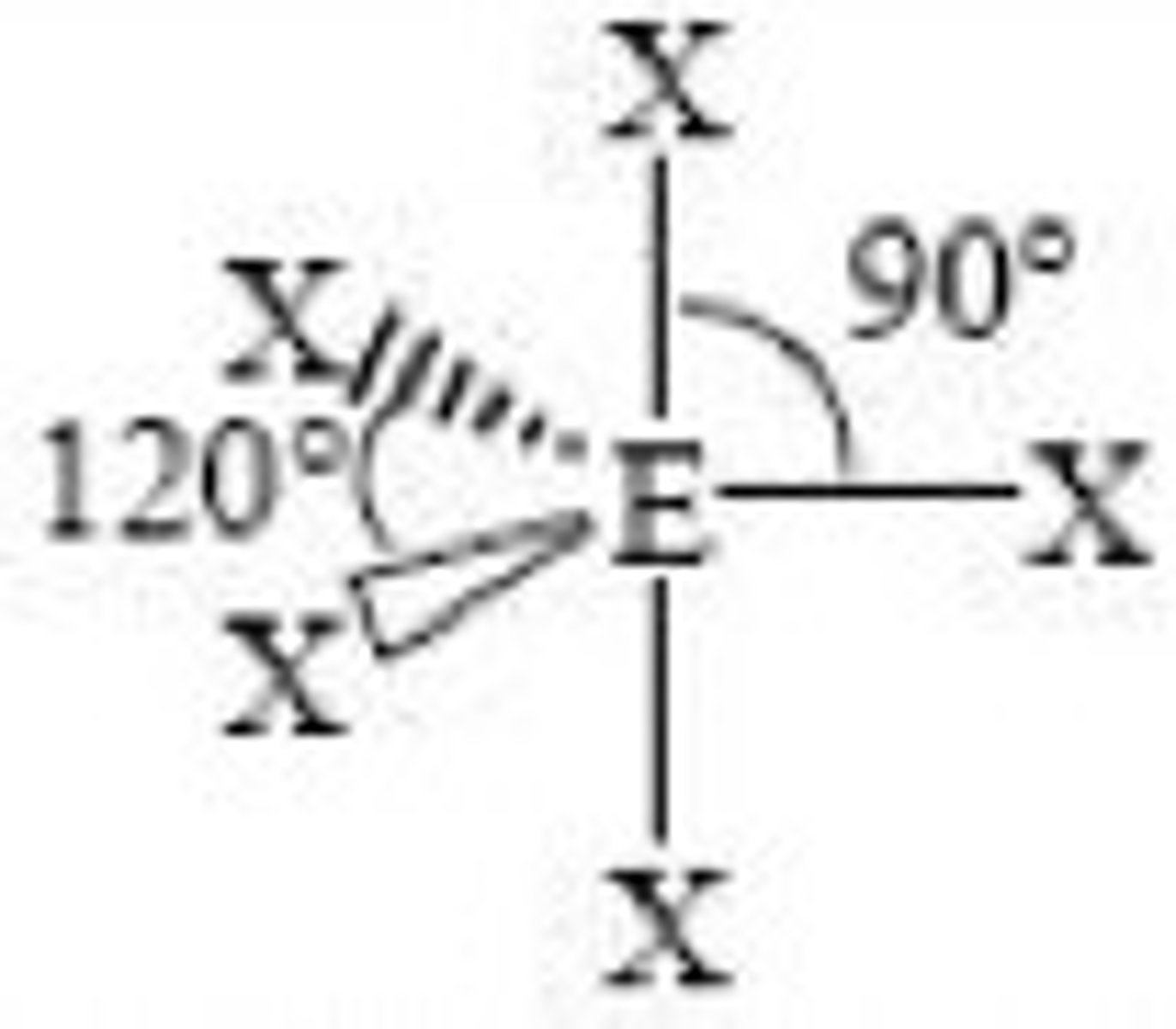
What is the shape, diagram and bond angle in a molecule with 5 bonded pairs and 0 lone pairs?
Trigonal bipyramid
90° and 120°
What is the shape, diagram and bond angle in a molecule with 6 bonded pairs and 0 lone pairs?
Octahedral
90°
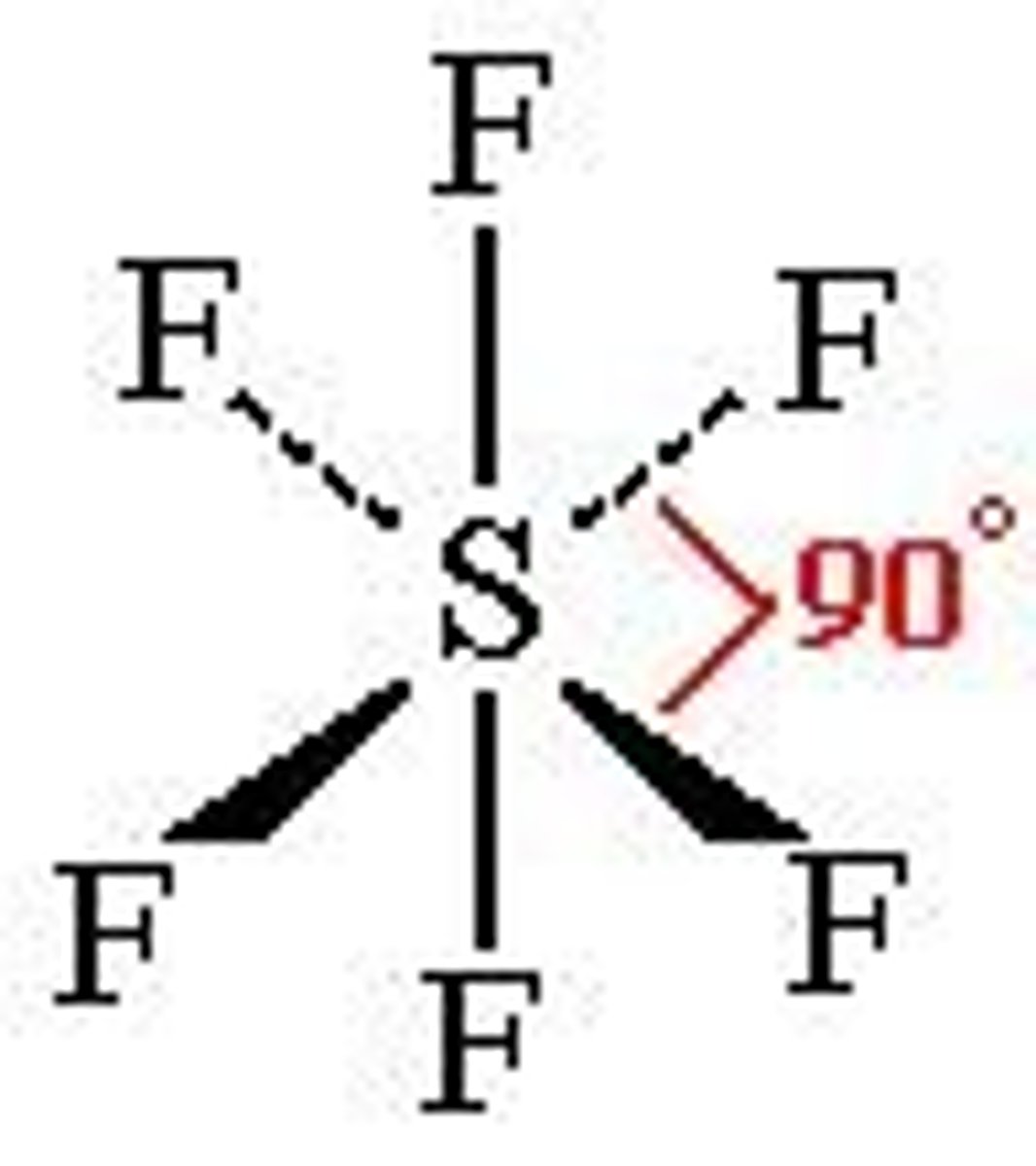
What is the shape, diagram and bond angle in a shape with 3 bonded pairs and 1 lone pairs?
Pyramidal
107°
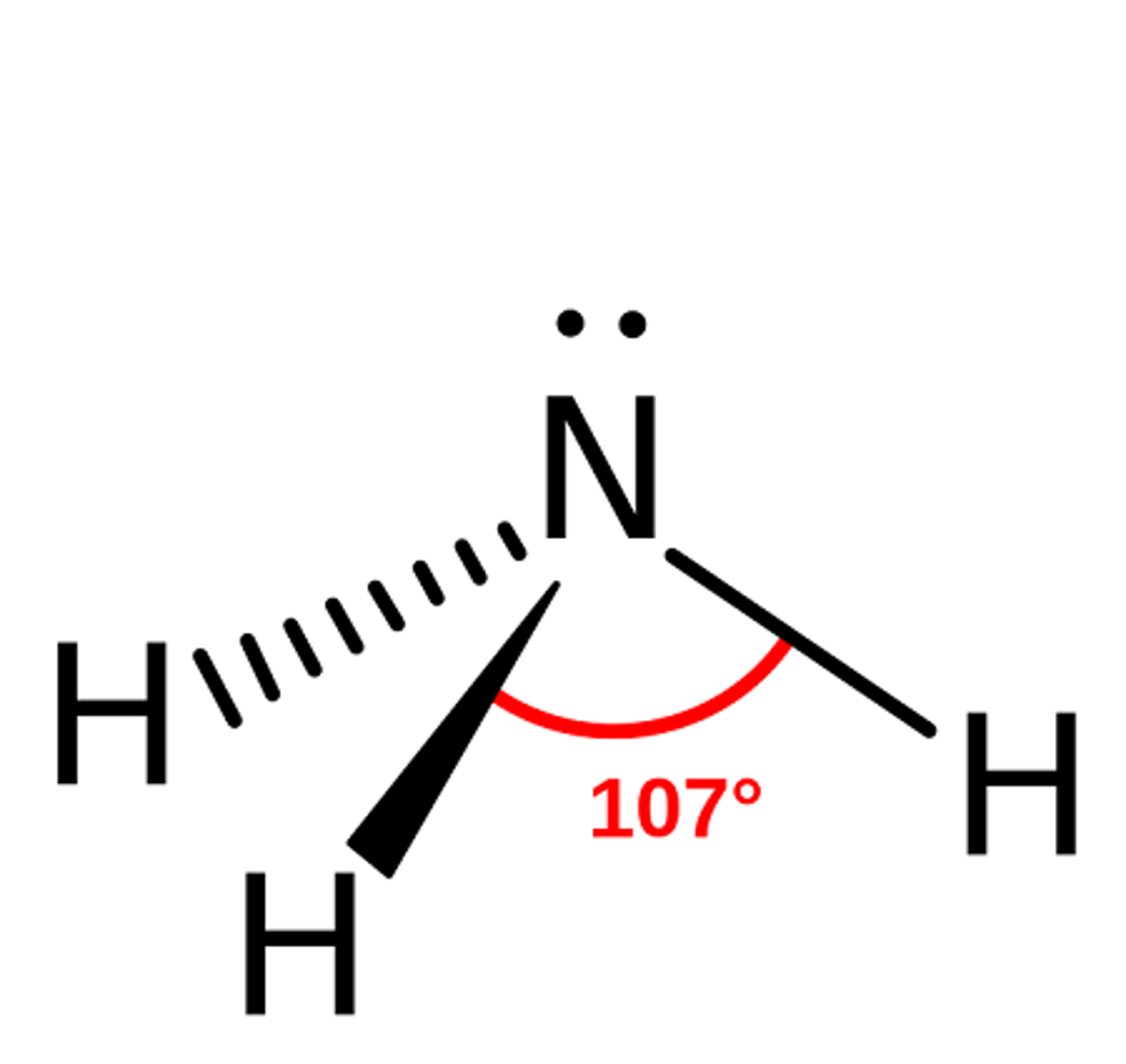
What is the shape, diagram and bond angle in a shape with 2 bonded pairs and 2 lone pairs?
Non linear 104.5°
By how many degrees does each lone pair reduce the bond angle?
2.5°
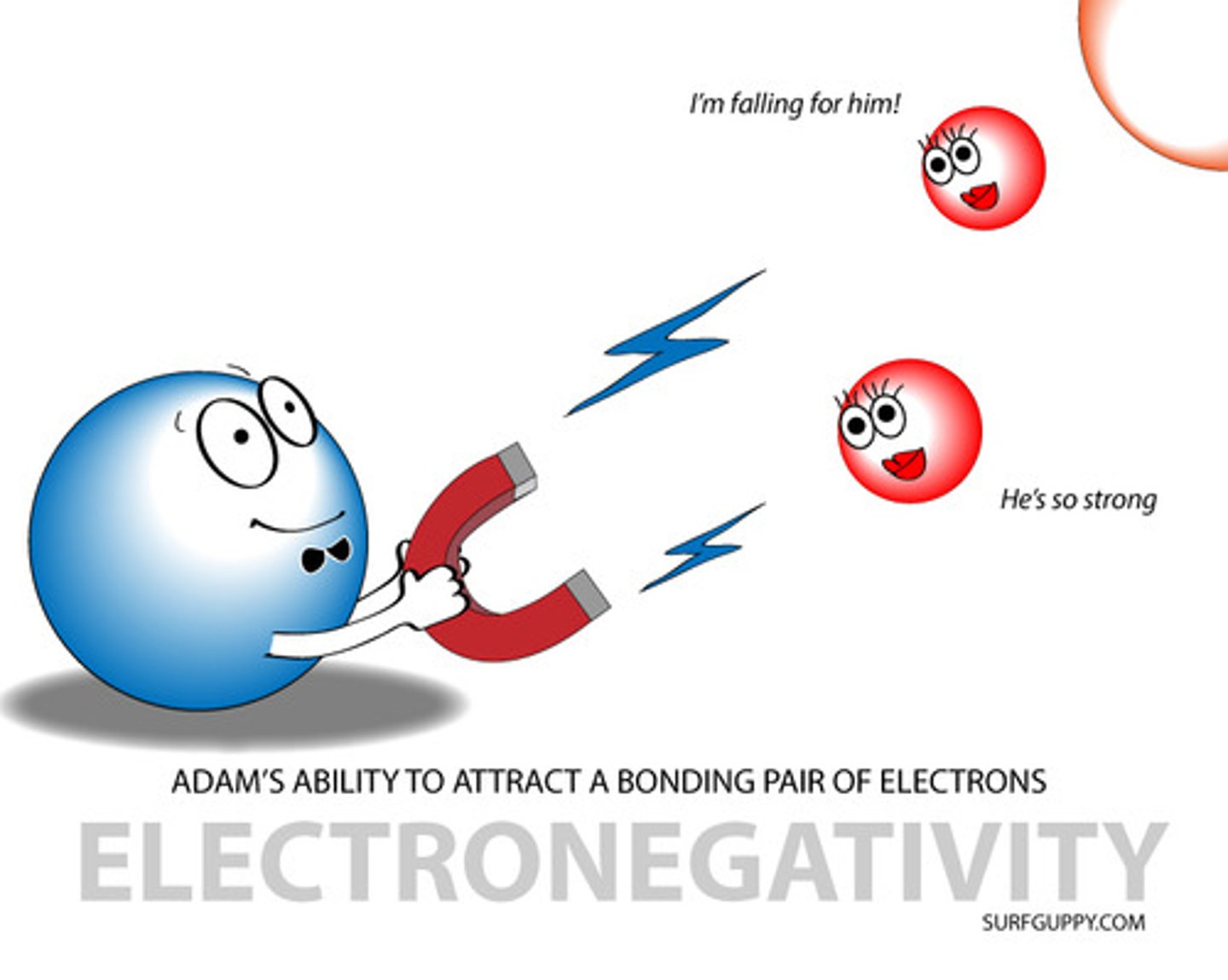
Define electronegativity
The ability of an atom to attract a pair of electrons (the electron density) in a covalent bond
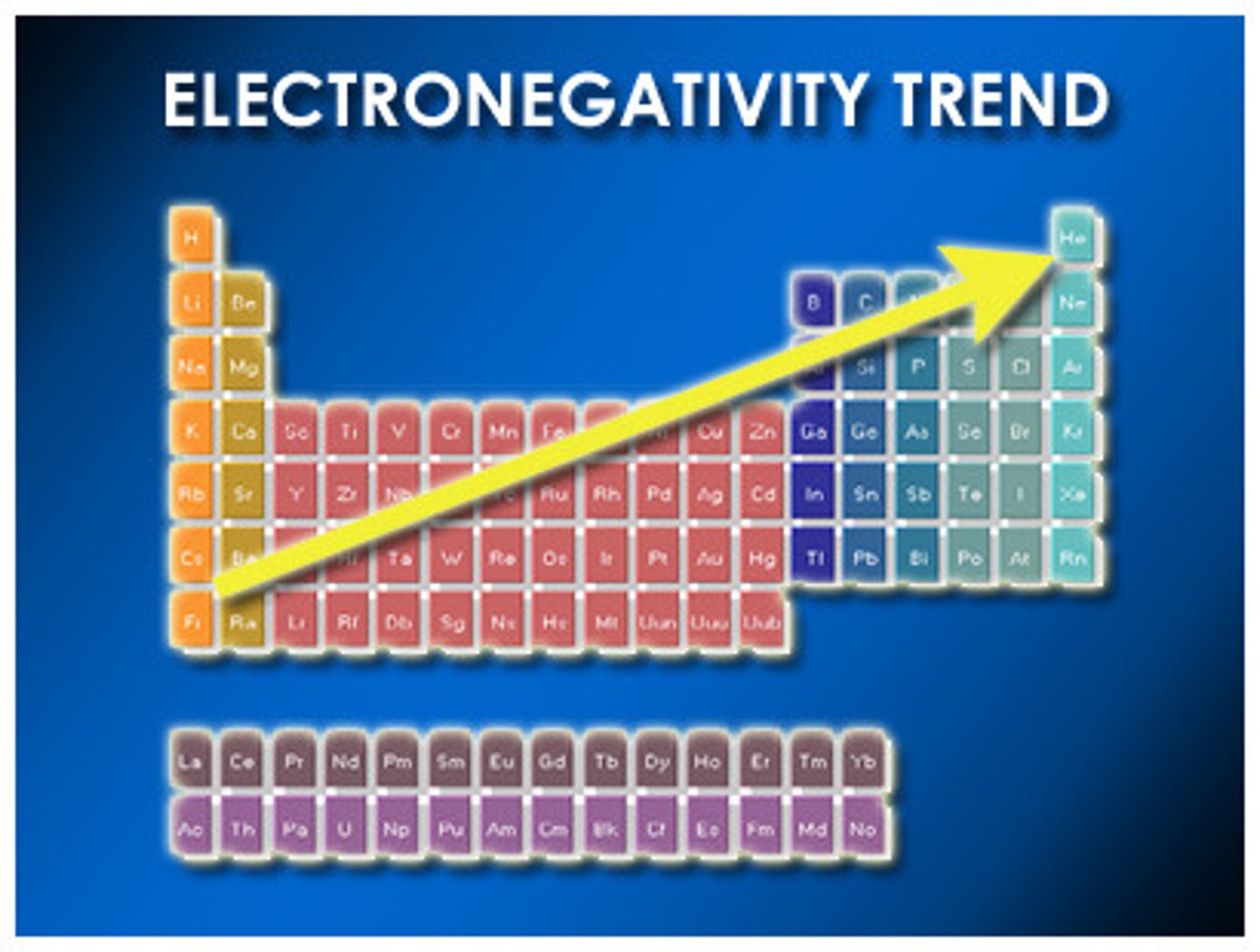
In which direction of the periodic table does electronegativity increase?
Top right, towards fluorine
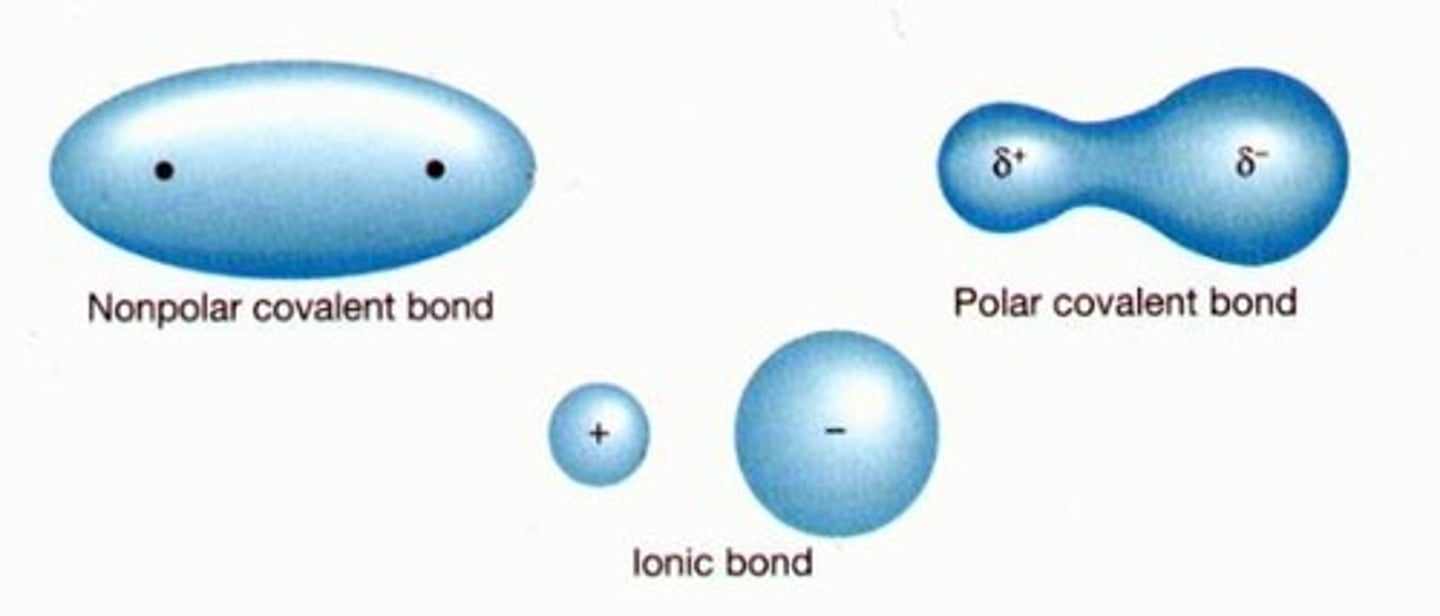
What does it mean when the bond is non-polar?
The electrons in the bond are evenly distributed
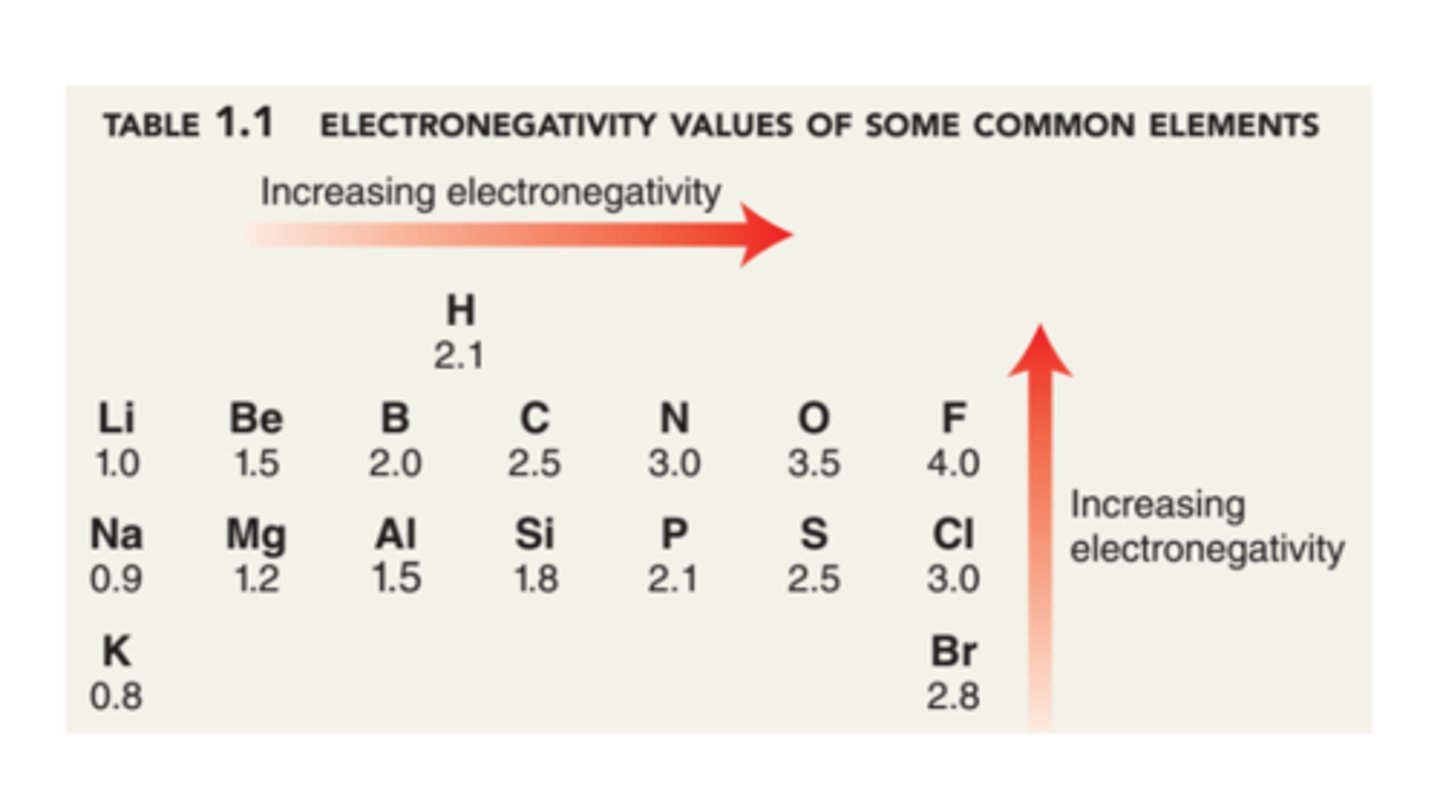
What is the most electronegative element?
Fluorine
How is a polar bond formed?
Bonding atoms have different electronegativities
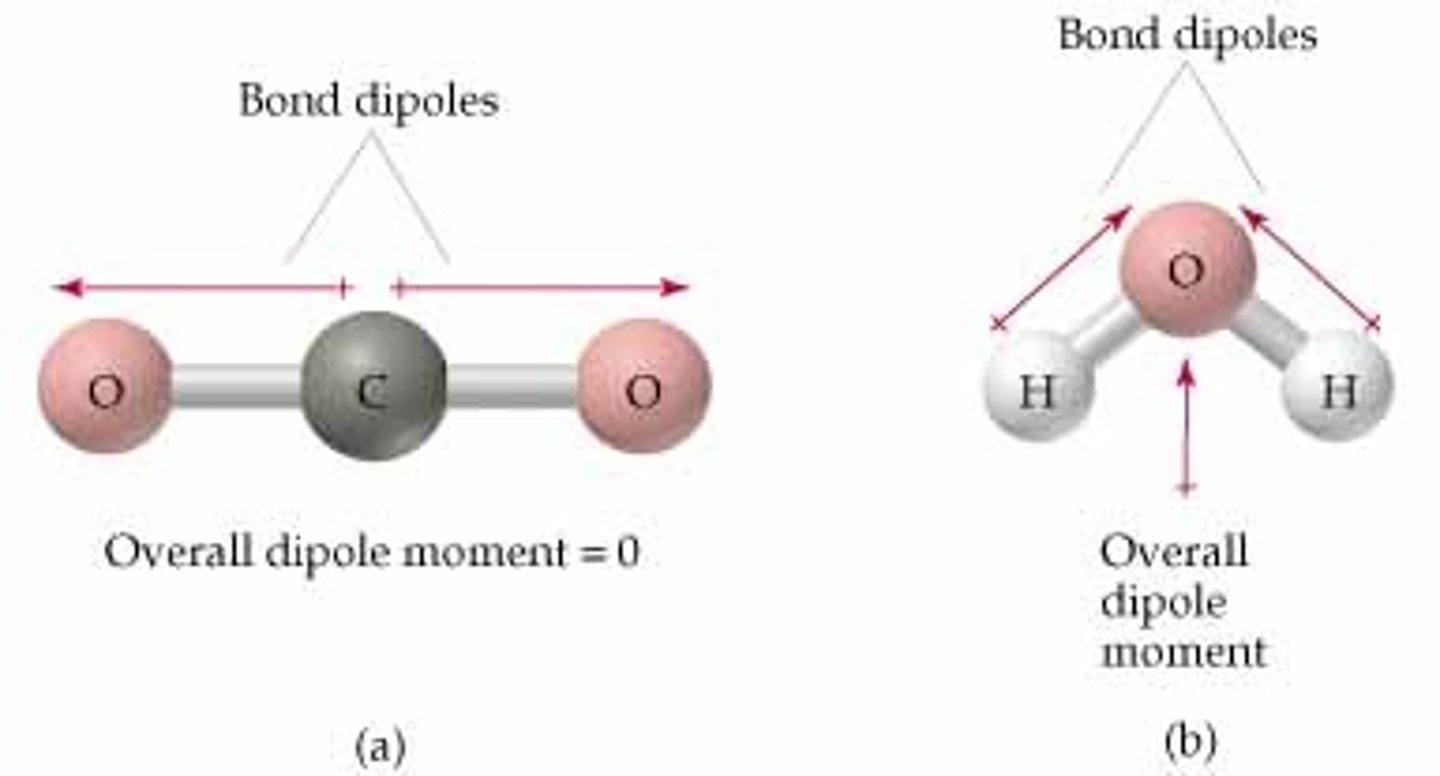
Why is H2O polar, whereas CO2 is nonpolar?
CO2 is a symmetrical molecule, so there is no overall dipole
What is meant by intermolecular force?
Attractive force between neighbouring molecules
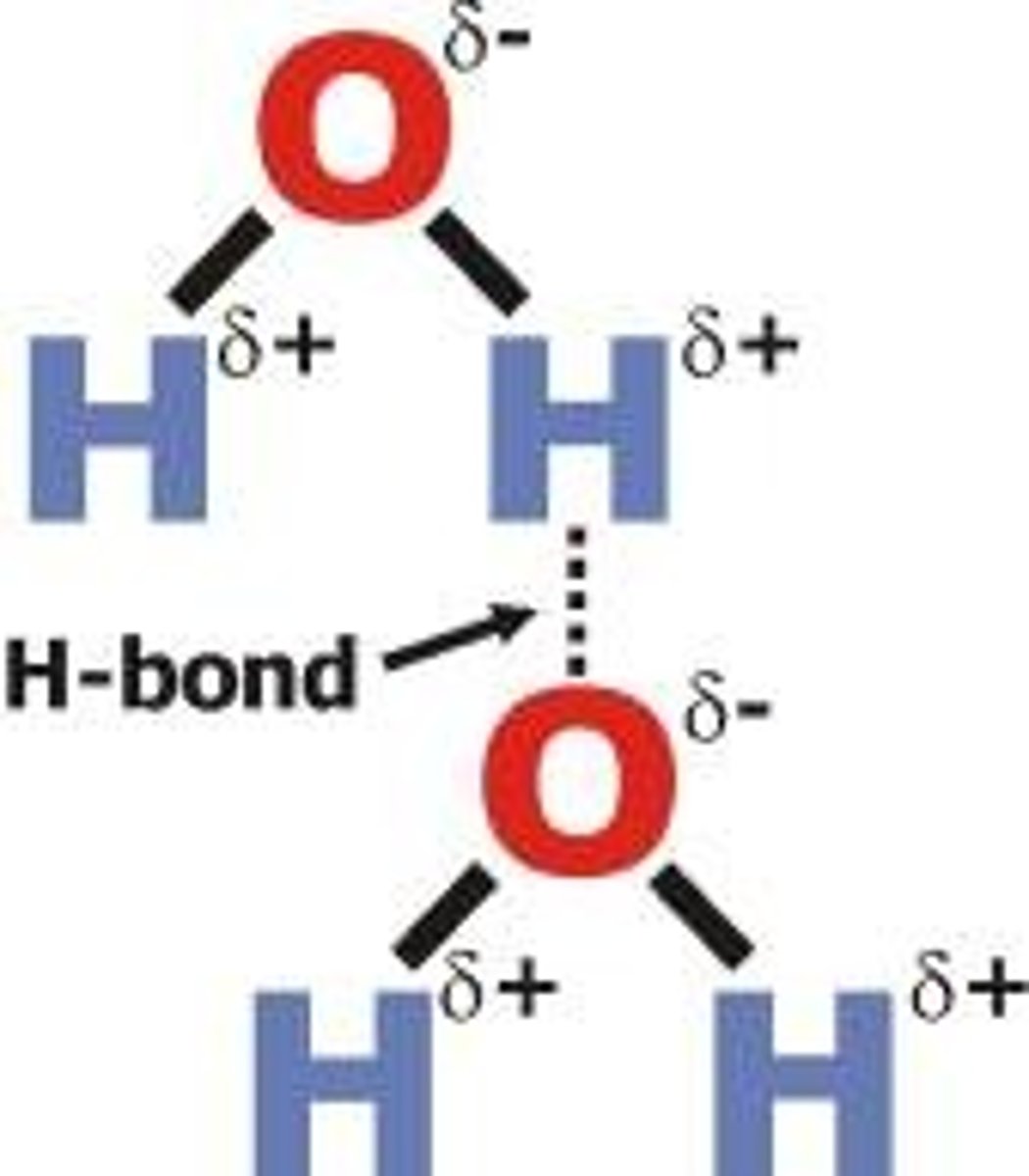
What are the 2 types of intermolecular forces?
● Hydrogen bonding ● Van der Waals' forces
What is the strongest type of intermolecular force?
Hydrogen bonding
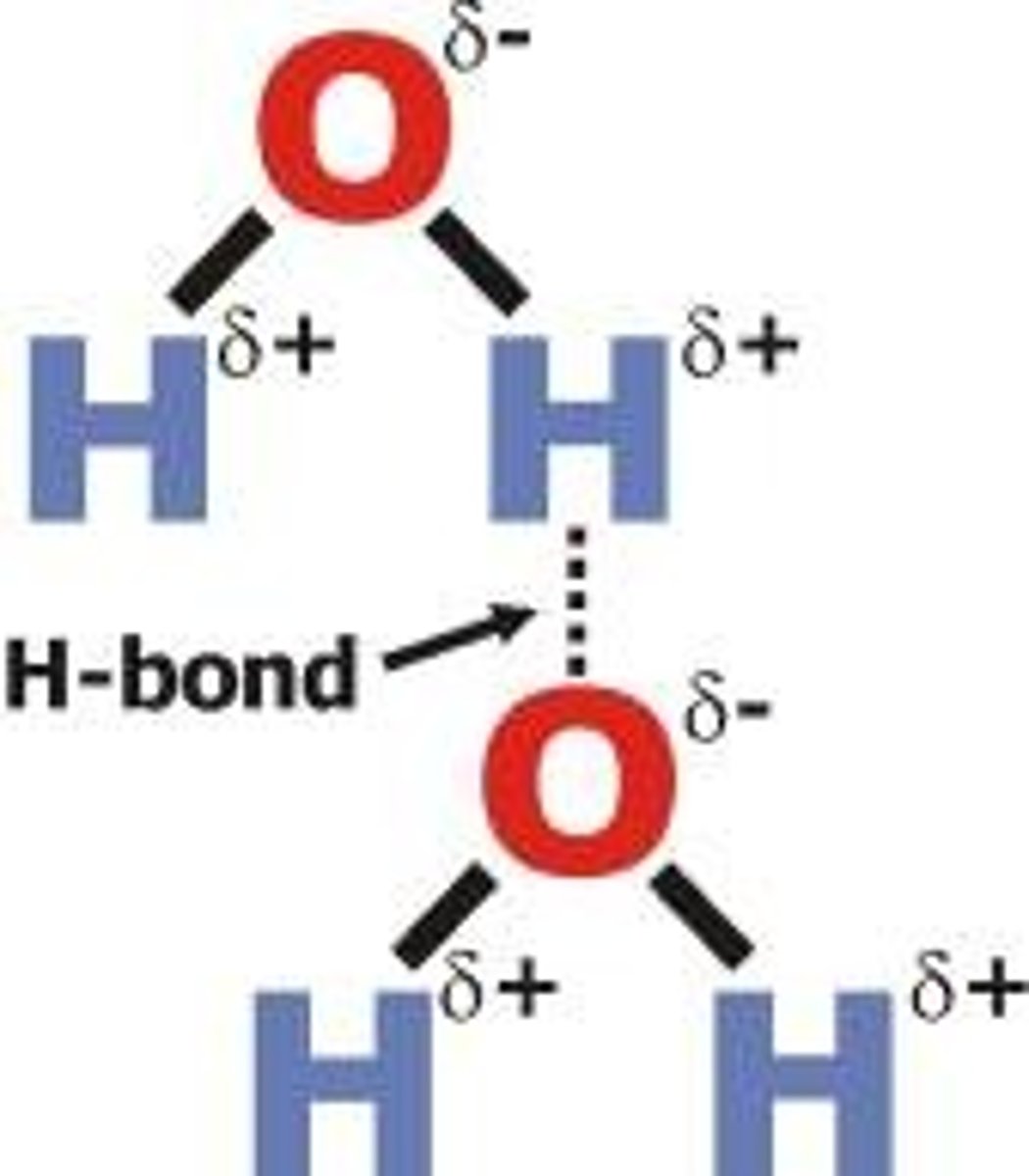
What are the 2 interactions that can be referred as Van der Waals' forces?
● Permanent dipole - induced dipole interaction
● Permanent dipole - permanent dipole interaction
Describe permanent dipole-induced dipole interactions
● When a molecule with a permanent dipole is close to other nonpolar molecules, it causes the nonpolar molecule to become slightly polar, leading to attraction
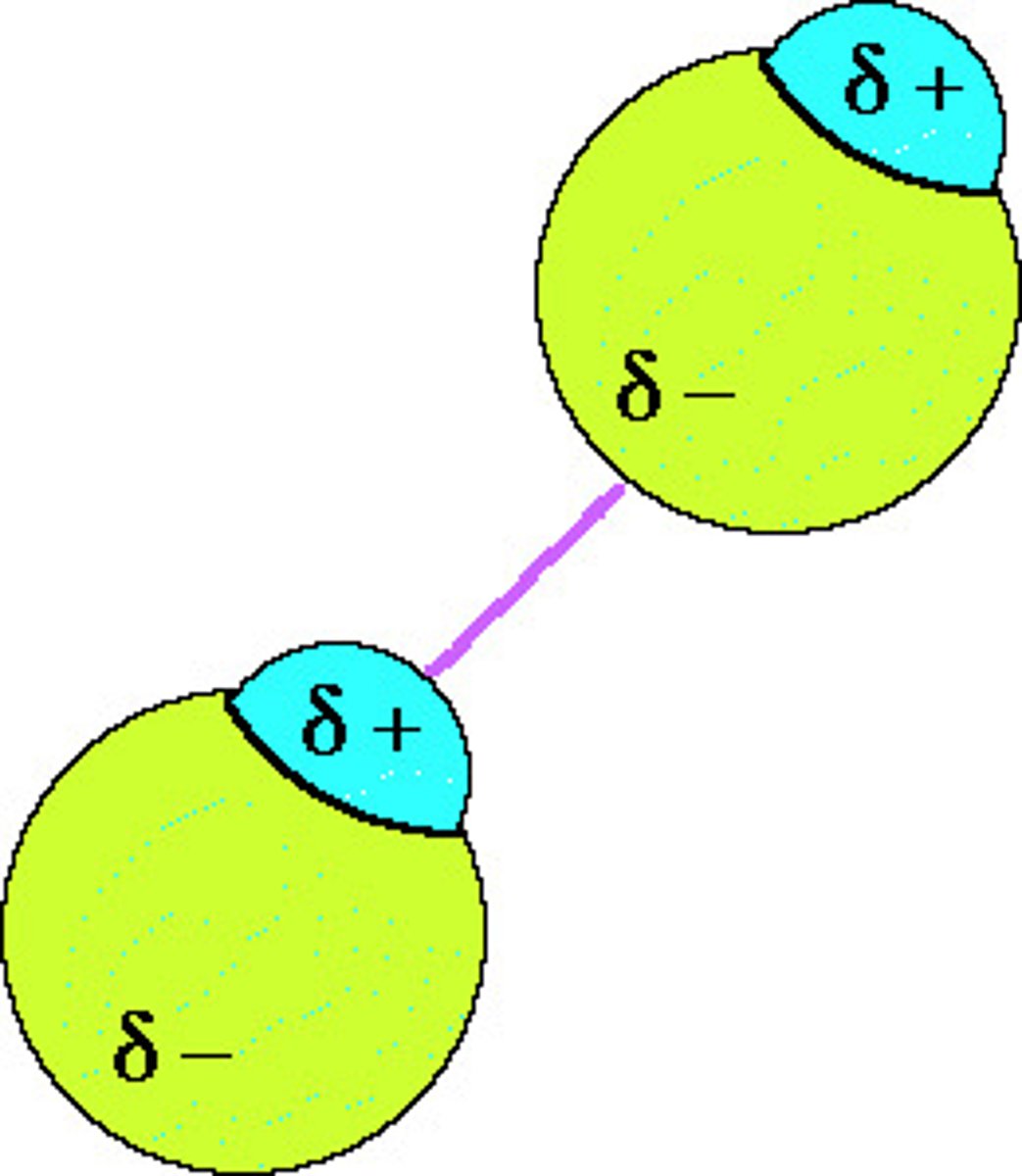
Describe permanent dipole- permanent dipole forces
The force between two molecules which have permanent dipoles
The δ+ end of the dipole in one molecule and the δ- end of the dipole in a neighbouring molecule are attracted towards each other
Describe London forces (Induced dipole-dipole forces)
● caused by random movements of electrons
● This leads to instantaneous dipoles
● Instantaneous dipole induces a dipole in nearby molecules
● Induced dipoles attract one another
Are London forces greater in smaller or larger molecules?
Larger due to more electrons
Does boiling point increase or decrease down the noble gas group? Why?
Boiling point increases because the number of electrons increases and hence the strength of London forces also increases
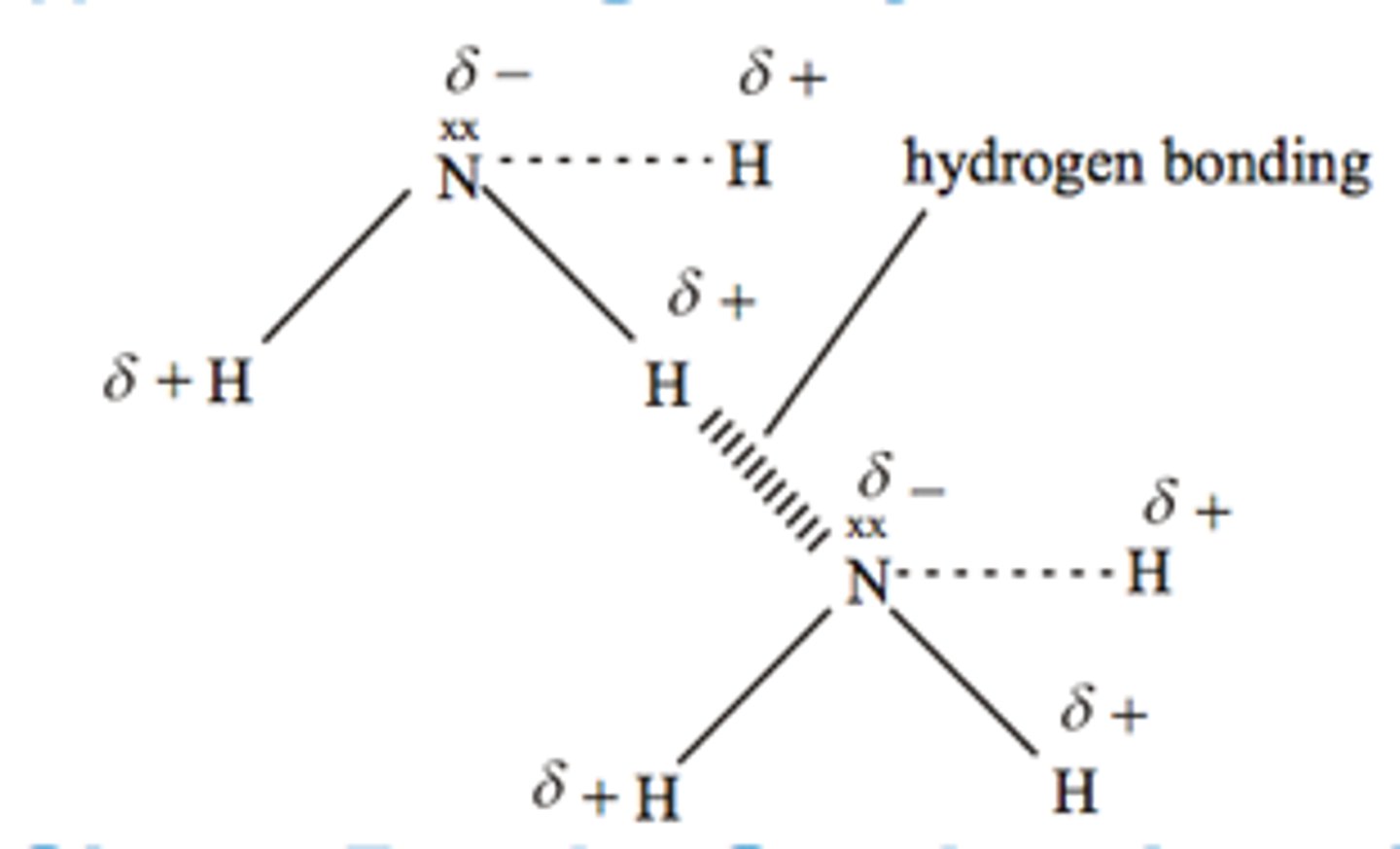
What conditions are needed for hydrogen bonding to occur?
O-H, N-H or F-H bond
lone pair of electrons on O, F, N
Because O, N and F are highly electronegative, H nucleus is left exposed
Strong force of attraction between H nucleus and lone pair of electrons on O, N, F
Draw a diagram of hydrogen bonding
Why is ice less dense than liquid water?
● In ice, the water molecules are arranged in an orderly pattern. It has an open lattice with hydrogen bonds.
● In water, the lattice is collapsed and the molecules are closer together.
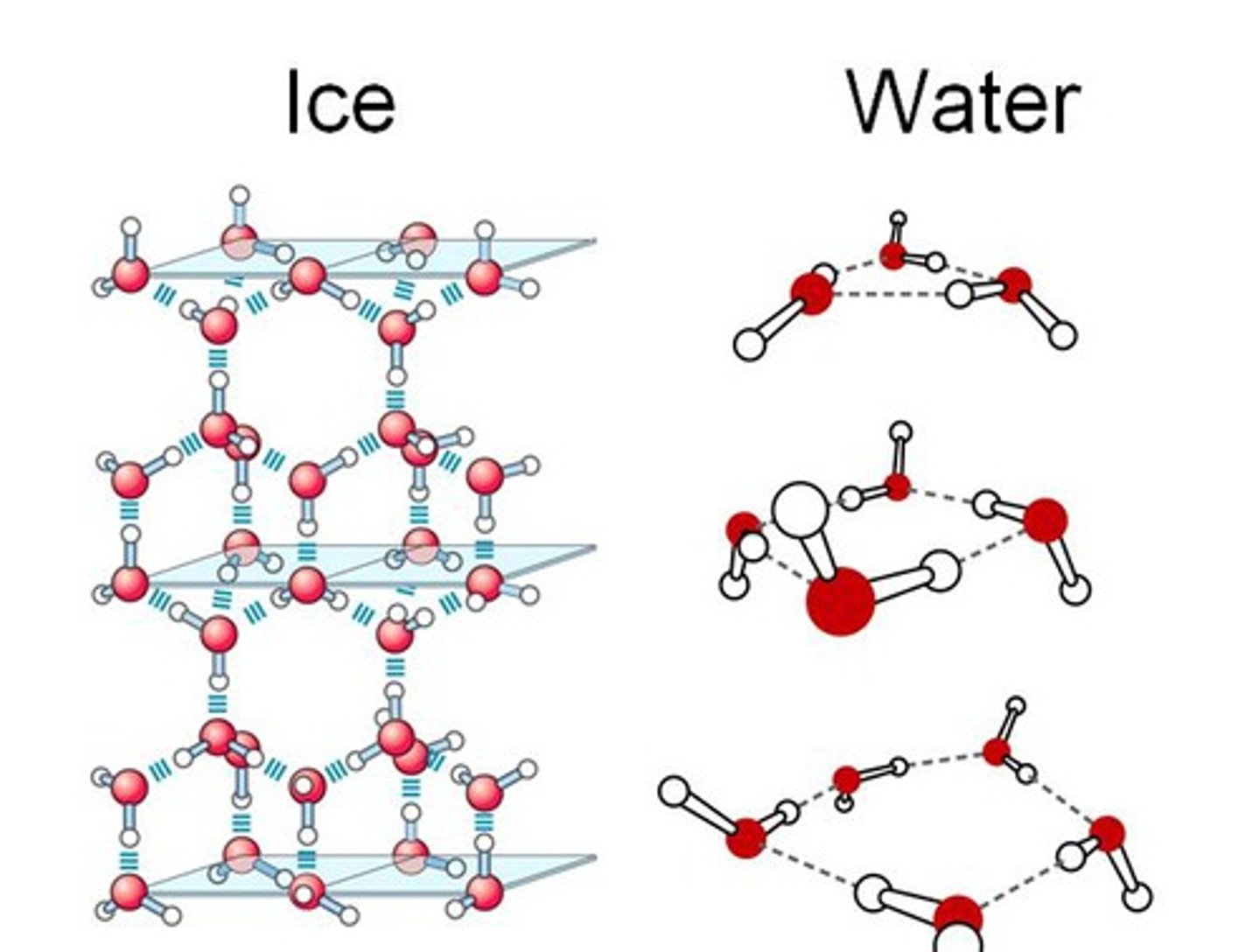
Why does water have a melting/ boiling point higher than expected?
Hydrogen bonds are stronger than other intermolecular forces so extra strength is required to overcome the forces