LO 2 – amino acids bonds and protein structural classification
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
N terminus
Amino group (-NH3+)
= start
C terminus
Carboxyl group (-COO-)
= end
Primary protein structure
numerous amino acids forming a polypeptide
linear sequence of amino acids
sequence determines higher orders of structures
Polypeptide
many amino acids strung together
Primary protein structure - bonds
covalent peptide bonds linking amino acids - very strong
Which protein structure?
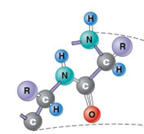
Primary protein structure
Secondary protein structure
backbone of polypeptide
linear sequence folded on itself
contains alpha helix, beta-sheet or random coils
Secondary protein structure - bonds
Peptide and hydrogen bonds
Two variations of Hydrogen bonds in secondary structures
Intermolecular (between CO and NH groups of amino acids on different polypeptides)
Intramolecular (between CO and NH groups of amino acids on same polypeptide)
Which protein structure
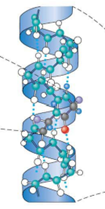
Secondary structure
Alpha helix
spiral shape; coils around itself; flexible, stretchy and dynamic
spiral turn has 3.6 aa (4th aa close to 3rd and 5th aa)
some aa disrupt bonds in alpha helix (known as helix breaker), as lack H, only found in beginning or end
Alpha helix - bonds
Hydrogen bonds (CO group binds to NH group of peptide bond, 4 aa downstream)
Beta Sheet
sheet like conformation
forms when 2 parts of a polypeptide chain lie side by side/across each other
intramolecular, stabilised by H bonds
Motifs
combinations of alpha helices and beta sheets
connected via looped regions of varying length (random coils, sequences of aa together)
Motifs - examples
b-a-b, hairpin loop and helix-turn-helix motifs
Tertiary structures
higher order of folding (polypeptide chain folded back on self to form a condensed structure)
depends on interaction between the side chains
Tertiary structures - bonds
Hydrogen bonds, peptide bonds, disulphide bonds, ionic bonds, London dispersion forces, hydrophobic interactions
Which protein structure?
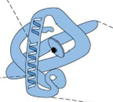
Tertiary structure
Tertiary structures - fibrous
extended and filamentous
repetitive aa sequence
structural (e.g collagen, hair, elastin, silk)
only small amount present in cell
Tertiary structures - globular
compact
different aa sequence
many functions
most proteins in cell
e.g. enzymes, haemoglobin
Peptide bond
linking 2 amino acids
condensation reaction: water removed - O from carboxyl group, 2 H from amino group of another aa. Carboxyl carbon and amino nitrogen: linked by peptide bond
Covalent bond (sharing electrons)
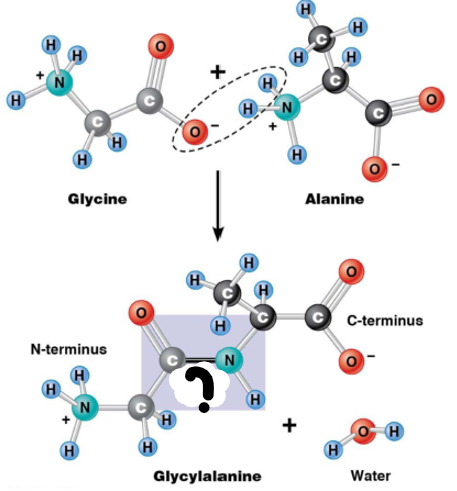
What bond?
peptide bond
Hydrogen bonds
non-covalent (not sharing electrons)
weak bond
donors (amine or hydroxyl group in aa) have H-atom linked to a more electronegative atom of an acceptor (e.g. carbonyl and sulfhydryl groups of aa)
abundant in proteins: one is weak, many is strong
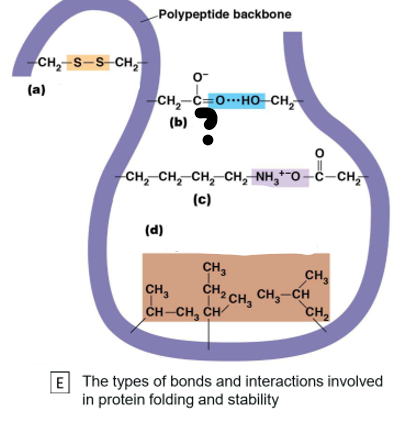
Which bond is (b)
Hydrogen Bond
Disulphide bonds
between sulphur atoms of 2 cysteine aa residues by covalent bonding through oxidation (removal of H atoms)
disruption denatures proteins through reduction (addition of 2 H atoms to regenerate the sulfhydryl groups SH)
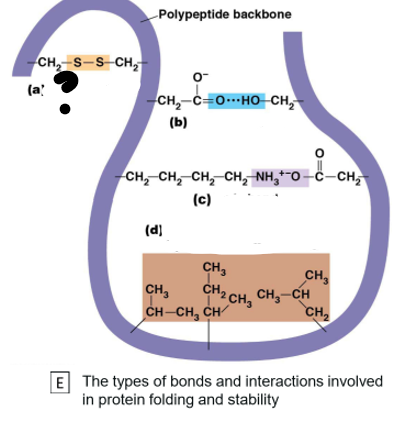
Which bond is (a) ?
Disulphide Bond
Which stage disulphide bond?
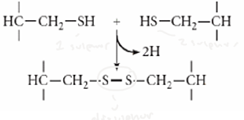
oxidation - formation of bond between sulphur bonds due to removal of H
Ionic bonds
R groups may be +ve or -ve
electrostatic interactions between R groups, +ve and -ve attract, similar charges repel
high/low pH affects ionic bonds, results in denaturation
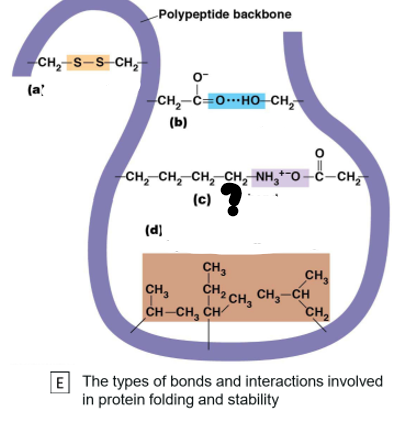
Which bond is c?
Ionic bond
London Dispersion Forces/van der Waals interactions
attraction of 2 non-polar molecules
weak
only effective if molecules are close, not when R group far away
Hydrophobic interactions
hydrophobic molecules excluded from water interaction
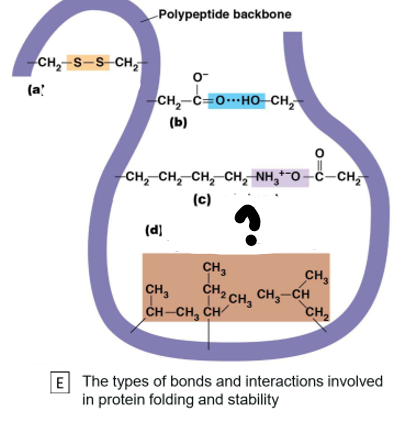
Which bond is (d)?
London dispersion forces, hydrophobic interactions