Peds Ch 48 - ENDO: Pancreas (DIABETES)
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
Pancreas
Dual-function organ:
Endocrine (ductless)
Exocrine (with ducts)
Pancreas - Endocrine Portion (Islets of Langerhans)
Alpha cells → secrete glucagon
Beta cells → secrete insulin
Insulin
Essential for:
Carbohydrate metabolism
Also affects fat and protein metabolism
Produced from amino acids
Normal production:
35–50 units/day in adults (less in children)
Stimulates Insulin Release
↑ Blood glucose (>100 mg/dL)
Hormones:
Gastrin (after eating)
Glucagon
Cortisol
Growth hormone (GH)
Progesterone
Estrogen
Inhibits Insulin Release
Epinephrine
Norepinephrine
Preserve glucose for fight-or-flight response
Key Concept - Insulin
Insulin is released when blood glucose is high and decreases when glucose is low, helping maintain glucose balance (homeostasis)
Common Childhood Disorders
Type 1 Diabetes (T1D)
Type 2 Diabetes (T2D)
Type 1 Diabetes Mellitus (T1D)
Absolute (or near absolute) insulin deficiency
Caused by autoimmune destruction of pancreatic beta cells
Incidence:
~1 in 500 children/adolescents in the U.S.
Affects males and females equally
Common onset:
5–7 years old or puberty
Abrupt onset
Type 1 Diabetes Mellitus (T1D) - Etiology
Autoimmune process destroys islet (beta) cells
Associated with genetic markers:
HLA-DR3 and HLA-DR4
Increased risk:
Family history (siblings)
T1D - Pathophysiology
↓/No insulin
Glucose cannot enter cells
→ Hyperglycemia (↑ blood glucose)
Kidneys respond:
Glucose spills into urine (glycosuria) when > ~160 mg/dL
→ Polyuria (↑ urination)
Fluid loss → Dehydration
→ Polydipsia (↑ thirst)
Cells starved of glucose:
Body breaks down fat & protein
Fat breakdown → Ketones
→ Ketoacidosis (DKA)
Electrolyte shifts:
Loss of potassium & phosphate
T1D - CM
Polyuria (frequent urination; may present as bed-wetting)
Polydipsia (excessive thirst)
Polyphagia (increased hunger)
Weight loss (despite eating)
Fatigue
Blurred vision
Mood/behavior changes
Dehydration
Signs of ketoacidosis (severe cases)
T1D - Complications (Untreated)
Diabetic ketoacidosis (DKA)
Electrolyte imbalance
Severe dehydration
Growth failure (short stature, underweight)
T1D - Management Overview
Insulin therapy (lifelong)
Carbohydrate counting
Blood glucose monitoring
General foot care
Key Features vs Type 2
Requires insulin (oral meds ineffective)
Abrupt onset
Weight loss (not obesity)
Possible “honeymoon period”
Temporary remission (1–12 months after diagnosis)
Type 1 vs Type 2
Category | Type 1 Diabetes | Type 2 Diabetes |
|---|---|---|
Age at Onset | 5–7 years or puberty | 40–65 years (can occur in adolescents—MODY) |
Onset | Abrupt | Gradual |
Weight Changes | Marked weight loss (early sign) | Associated with obesity |
Polydipsia | Present | Present |
Polyphagia | Present | Not typical |
Polyuria | Present (may begin as bed-wetting) | Present |
Fatigue | Present (↓ school performance) | Present |
Blurred Vision | Present | Present |
Mood Changes | Present (behavior issues possible) | Present |
Therapy | Insulin required (oral meds ineffective) | Diet, oral meds, or insulin |
Nutrition | Carb counting + glucose monitoring | Weight control + balanced diet |
Skin/Foot Care | Basic care | Meticulous care required |
Remission | Honeymoon period (1–12 months) | None |
T1D - Assessment (Clinical Signs)
Abrupt onset of symptoms
Polydipsia (increased thirst)
Polyuria (increased urination)
May present as enuresis (bed-wetting) in toilet-trained child
Dehydration
May lead to constipation
In severe cases:
Child may present in coma due to:
Hyperglycemia
Ketoacidosis
T1D - Laboratory Findings
Random plasma glucose > 200 mg/dL
Glycosuria (glucose in urine)
Normal Blood Glucose Ranges (Children with T1D)
Timing | Glucose Level (mg/dL) |
|---|---|
Before meals | 70–110 |
1 hour after meals | 90–180 |
2 hours after meals | 80–150 |
2 a.m. – 4 a.m. | 70–120 |
T1D - Diagnostic Criteria (Confirm on 2 Occasions)
Symptoms + random glucose > 200 mg/dL
Fasting glucose > 126 mg/dL
2-hour GTT glucose > 200 mg/dL
Glucose Tolerance Test (GTT)
Procedure:
8-hour fasting
Drink glucose solution
Blood drawn:
Baseline (fasting)
1 hour
2 hours
GTT - Important Nursing Considerations
GTT can be difficult for children:
Fasting
Sweet solution
Multiple blood draws
Use:
Topical anesthetic (EMLA) to reduce pain
Do NOT draw blood from IV line
IV fluids containing glucose → false high results
T1D - Blood Tests
Acid–Base & Respiratory Status
pH
PCO₂ (partial pressure of carbon dioxide)
Helps assess acidosis (DKA)
Electrolytes
Sodium (Na⁺)
Potassium (K⁺)
Imbalances common in diabetic ketoacidosis
Glycosylated Hemoglobin (HbA1c)
Measures average blood glucose over past 3–4 months
Based on RBC lifespan (~120 days)
Normal (nondiabetic):
1.8–4.0
> 6.0 = chronic hyperglycemia
White Blood Cell (WBC) Count
May be elevated
Even without infection
Due to stress response (DKA)
Still must assess for infection
Common trigger of diabetic crisis
Infection Workup
Nose and throat cultures
To identify possible infection trigger
Electrocardiogram (ECG)
Done if low potassium
Looks for:
T-wave abnormalities
Signs of potassium imbalance
T1D - Overall Treatment Components (5)
Insulin administration
Nutrition regulation
Exercise regulation
Stress management
Blood glucose & urine ketone monitoring
Initial Management (Hospitalization)
~3-day hospital stay
Focus:
Stabilization
Extensive education (child + caregivers)
Initial Insulin Therapy (Emergency Phase)
Child usually presents with:
Hyperglycemia
Ketoacidosis (DKA)
Initial Regulation of Insulin - IV Insulin
Dose:
0.1–0.2 units/kg/hour
Type:
Regular (short-acting) insulin (e.g., Humulin-R)
Reason:
Rapid onset of action
Initial Regulation of Insulin - Response to Treatment
When glucose < 200 mg/dL:
Reduce insulin infusion
Within ~12 hours:
Acidosis improves
Glucose approaches normal range
Initial Regulation of Insulin - Glucose Administration
Even with hyperglycemia:
Glucose may be added to IV fluids
Reason:
Insulin allows cells to use glucose
Without glucose:
Body continues fat/protein breakdown
Acidosis worsens
Transition Phase (After Stabilization)
After ~24 Hours
Switch from:
IV → Oral feeding
Continue insulin therapy
Transition Phase - Short-Term Insulin Plan
Regular insulin (SC):
Given 3–4 times/day initially
Transition Phase - Next Step
Add:
Intermediate-acting insulin
Timing:
Usually day 2, once oral intake begins
T1D - Daily Insulin Requirements
Children:
0.4–0.7 units/kg/day
Adolescents:
Up to 1.2 units/kg/day
Given in 2 divided doses:
Morning (before breakfast): 2/3 total dose
Evening (before dinner): 1/3 total dose
Common Insulin Regimen
Combination of:
Intermediate-acting insulin + Regular (short-acting) insulin
Typical ratio:
2:1 ratio
Example: 0.75 intermediate : 0.33 regular
Often mixed in same syringe (varies per child)
Rapid-acting Insulin
Lispro (Humalog)
Aspart
Lispro (Humalog) - Onset
Immediate
Lispro (Humalog) - Peak
30 min–1 hr
Lispro (Humalog) - Duration
3–4 hr
Aspart - Onset
~15 min
Aspart - Peak
30–40 min
Aspart - Duration
3–5 hr
Short-acting Insulin
Regular (Humulin-R)
Regular (Humulin-R) - Onset
0.5–1 hr
Regular (Humulin-R) - Peak
2–4 hr
Regular (Humulin-R) - Duration
5–7 hr
Long-acting Insulin
Lantus (glargine)
Humulin-U
Lantus (glargine) - Onset
~1 hr
Lantus (glargine) - Peak
Minimal (~5 hr)
Lantus (glargine) - Duration
~24 hr
Humulin-U - Onset
~6 hr
Humulin-U - Peak
16–18 hr
Humulin-U - Duration
36+ hr
Intermediate-acting Insulin
Humulin-N
Humulin-L
Humulin-N - Onset
1–2 hr
Humulin-N - Peak
4–12 hr
Humulin-N - Duration
24+ hr
Humulin-L - Onset
1–3 hr
Humulin-L - Peak
6–14 hr
Humulin-L - Duration
24+ hr
Types of Insulin
Type | Example | Onset | Peak | Duration |
|---|---|---|---|---|
Rapid-acting | Lispro (Humalog) | Immediate | 30 min–1 hr | 3–4 hr |
Rapid-acting | Aspart | ~15 min | 30–40 min | 3–5 hr |
Short-acting | Regular (Humulin-R) | 0.5–1 hr | 2–4 hr | 5–7 hr |
Long-acting | Lantus (glargine) | ~1 hr | Minimal (~5 hr) | ~24 hr |
Intermediate-acting | Humulin-N | 1–2 hr | 4–12 hr | 24+ hr |
Intermediate-acting | Humulin-L | 1–3 hr | 6–14 hr | 24+ hr |
Long-acting | Humulin-U | ~6 hr | 16–18 hr | 36+ hr |
Why Combine Insulins
Different peak times → better glucose control
Covers:
Meals (short-acting)
Baseline needs (intermediate/long-acting)
Peak Times & Hypoglycemia Risk
Short-acting peak (2–4 hr):
~10 a.m.–12 p.m. (after breakfast dose)
Intermediate peak (8–14 hr):
Late afternoon (before dinner)
⚠ These are high-risk times for hypoglycemia
Alternative Regimens
Some children require:
3–4 injections/day
Benefit:
More flexibility with:
Meals
Activity
Dose Adjustment (“Thinking Scales”)
Lag Time (Injection → Meal)
High premeal glucose:
↑ lag time (delay eating)
Low premeal glucose:
↓ lag time (eat sooner)
Adjustments Based on Lifestyle
Large meal:
↑ insulin dose
Increased activity (e.g., sports):
↓ insulin dose
Insulin Glargine (Lantus)
Long-acting (24 hr)
Given via pen injection
Cannot be mixed with other insulins (low pH)
May be used with:
3 doses of short-acting insulin before meals
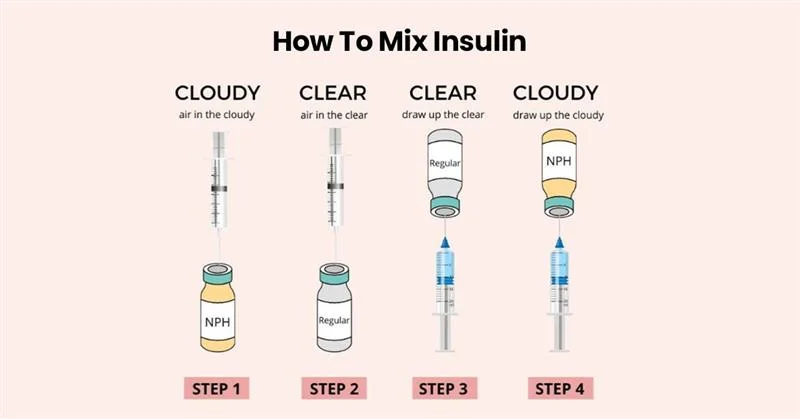
Mixing Insulin
Draw short-acting (regular) insulin FIRST, then intermediate-acting.
Rationale:
Prevents contamination of short-acting insulin.
Maintains its rapid onset, which is critical for emergency use.
Route of Administration
Subcutaneous (SC) is the standard route.
IV administration: REGULAR INSULIN ONLY
Used only in emergencies.
May give ½ of the required dose IV.
Injection Sites
Common sites in children:
Upper outer arms
Outer thighs
Other possible site:
Abdomen (more common in adults)
Often avoided in children due to tenderness
Guidelines:
Avoid:
Area around the navel
Use:
Thigh: 4 inches below hip to 4 inches above knee (adjust for child size)
Site Rotation
Rotate injection sites systematically.
Document site use (especially in hospital settings).
Prevents:
Lipodystrophy (atrophy) → pockmarks
Lipohypertrophy (fat buildup/scar tissue)
Effects of Repeated Use of Same Site
Leads to:
Lipohypertrophy (scar tissue buildup)
Reduced pain → encourages repeated use
Problem:
Poor insulin absorption
Insulin becomes “trapped” in tissue
Risk:
Increased dose required → later injection at new site can cause hypoglycemia (overdose)
Activity Considerations - Insulin
Exercise increases insulin absorption.
Avoid injecting into areas that will be exercised soon:
Example:
Jogging → avoid thigh
Tennis → avoid dominant arm
Insulin Storage & Temperature
Store extra insulin in refrigerator.
Inject at room temperature:
Reduces tissue damage (atrophy)
Improves effectiveness
Injection Technique
Use short needle (< 0.4 inches).
Inject at 90-degree angle:
Still reaches subcutaneous tissue
Easier for children than 45-degree angle
Devices
Options include:
Insulin pens
Jet injectors
Benefits:
Pre-measured doses
Easier to use
Promote independence
Compatible with 90-degree technique
Insulin Pumps (CSII – Continuous Subcutaneous Insulin Infusion)
Small automatic device (≈ size of an iPhone).
Delivers continuous insulin at a constant rate.
Uses:
Regular insulin in a syringe inside the pump.
Thin tubing connected to a subcutaneous needle in the abdomen.
Insulin Pumps - Effectiveness
Provides better glucose regulation than periodic injections.
Improves management of Type 1 Diabetes (T1D).
Insulin Pumps - Benefits
Most children:
Adjust well
Prefer over injections
Psychosocial & Quality of Life Improvements:
Increased self-efficacy in diabetes management
Greater independence (especially adolescents)
Improved quality of life (all age groups)
Reduced:
Parenting stress
Worry about hypoglycemia
Overall disease burden
Inhalation Insulin
Not currently available.
Still in experimental trials.
Challenges
Developing:
Accurate delivery system
Concerns:
Effects of colds/allergies
Nasal membrane swelling (edema) may alter absorption
Insulin-to-Carbohydrate Ratio
Individualized based on:
Age
Activity level
Example (from text):
Ratio: 1 unit insulin : 10 g carbs
Meal: 50 g carbs
Calculation:
50 ÷ 10 = 5 units insulin before meal
Meal Planning
Daily pattern:
3 meals (spaced, high in fiber)
3 snacks:
Midmorning
Midafternoon
Evening
Purpose
Maintain stable carbohydrate levels throughout the day
Self-Monitoring of Blood Glucose (SMBG)
Children (early school age) can learn:
Finger puncture technique
Use of computerized glucose monitors
Tools:
Spring-loaded lancet → minimizes pain
Automatic glucometer → simplifies reading
Adolescents:
Usually able to monitor glucose independently daily
Urine Testing
Not routine
Used when child:
Has acute illness (e.g., nausea, vomiting, diarrhea)
Cannot eat
Purpose
Detect ketonuria
Key Finding
Presence of acetone:
Indicates fat breakdown for energy
May signal developing acidosis
“Honeymoon” Period
Occurs after initial diagnosis and insulin regulation
Characteristics
Temporary phase where:
Little or no insulin is required
Cause
Exogenous insulin stimulates pancreas to produce small amounts of natural insulin
Duration
Can last:
1 month up to 1 year
Important Teaching
Not a cure
Symptoms will return as pancreatic function declines
Parents should be prepared for recurrence
Stress Adjustment
Effect of Stress
Physical or emotional stress → increased insulin needs
Nursing/Assessment Points
Assess:
Blood glucose monitoring difficulties
Injection challenges
Home and school stress
Communication
Interview child separately from parents when possible
School Considerations
May need:
Parent–school collaboration
Support for participation in activities/sports
Privacy:
Some children prefer testing in nurse’s office
Complications
Short-Term
Infection/fever:
Causes insulin resistance
Requires increased insulin
Parental Teaching
Notify provider if child:
Is ill
Has nausea/vomiting
Surgery
Requires:
Careful glucose regulation
Monitoring if oral intake is restricted
Long-Term (Chronic Hyperglycemia Effects)
Arteriosclerosis → poor circulation, kidney disease
Retinal damage → vision problems, blindness
Cataracts
Key Point
Prevention starts in childhood with good glucose control
Pancreas Transplantation
Indication
Severe complications:
Kidney disease
Arteriosclerosis
Procedure
Original pancreas:
Not fully removed (digestive function preserved)
New pancreas:
Enzymes diverted to:
Intestine or bladder
OR ducts blocked (sclerosed)
Donors
Cadaver donors
Living donors:
Can donate up to 45% of pancreas
Post-Transplant Care
Lifelong immunosuppressive therapy:
Antilymphocyte globulin
Cyclosporine
Prednisone
Azathioprine
Rejection treatment:
Monoclonal T-cell antibodies (OKT3)
Limitations
Considered last resort because:
Major surgery
~50% rejection rate
Lifelong immunosuppression required
Possible recurrence of diabetes
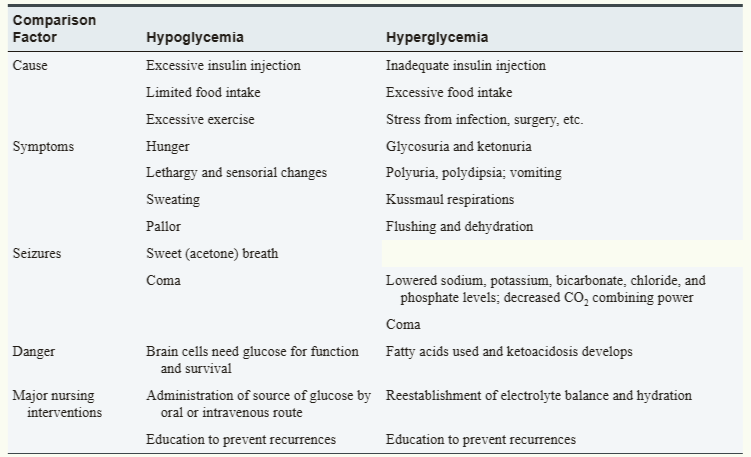
HYPOGLYCEMIA vs HYPERGLYCEMIA
Type 2 Diabetes Mellitus (T2D)
Characterized by diminished insulin secretion
Not autoimmune (unlike T1D)
Pancreas:
Still produces small amounts of insulin
T2D - Management
Most children:
Do NOT require daily insulin
Treatment includes:
Diet (nutrition therapy)
Exercise
Oral hypoglycemic agents
T2D - Medication
Metformin (biguanide)
Decreases glucose production in liver
Increases insulin sensitivity in liver and muscles
T2D - Risk Factors
Obesity (major predisposing factor)
Family history of diabetes
Higher prevalence in:
African, Hispanic, Asian, Native American populations
Lifestyle factors:
High fat and carbohydrate diet
Physical inactivity
Associated condition:
Polycystic Ovary Syndrome (PCOS)
T2D - Onset & Diagnosis
Often appears during puberty
Due to increased insulin resistance from sex hormones
Diagnosis may be difficult initially:
Symptoms overlap with T1D
T2D - CM
Urine findings:
Glucose present
Few or no ketones
Symptoms (often mild):
Less pronounced polyuria (urination)
Less pronounced polydipsia (thirst)
T2D - Key Physical Sign
Acanthosis Nigricans
Present in ~90% of children with T2D
Appearance:
Dark, shiny, thickened skin
Common locations:
Neck (“dirty neck”)
Axilla (armpits)
Between fingers and toes
T2D - Screening Recommendations
Screen children who have:
Family history of T2D
High-risk ethnicity
Signs like acanthosis nigricans or hypertension
Method
Fasting blood glucose test
Timing
At puberty
Every 2 years thereafter
T2D - Complications (Long-Term)
Atherosclerosis → poor circulation
Kidney disease
Poor wound healing
Vision problems/blindness
Cause
Chronic hyperglycemia → blood vessel damage